Abstract
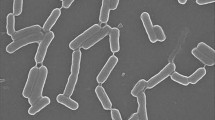
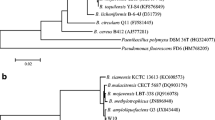
One isolate among the various bacterial soil isolates screened in our study showed broad spectrum and profound antifungal activity and was characterized by biochemical and molecular approaches. Sequence analysis of the 16S rRNA gene indicated more than 98 % sequence similarity with Bacillus amyloliquefaciens ATCC 23350T and its variant Bacillus velezensis BCRC 17467T. However, the biochemical characteristics of the isolated strain, such as cellular fatty acid composition, differed significantly from those of these two Bacillus strains, with the isolated strain containing anteiso-C15:0 (terminally branched saturated fatty acids) as a predominant fatty acid. The band pattern of the pulsed field gel electrophoresis profile of AvrII-digested genomic DNA of the isolate also showed significant variation from those of the two closely related Bacillus type strains. Based on these differences, the isolate was considered to be a new strain of B. amyloliquefaciens and deposited as Bacillus amyloliquefaciens strain fiply 3A in the Deutsche Sammlung von Mikroorganismen und Zellkulturen GmbH (DSMZ) culture collection (DSM 22646) and in MTCC, IMTECH, Chandigarh (DSM 22646). Purification of the extracellular antifungal compound produced by the isolate by HPLC and its analysis by LC-ESI-MS revealed it to be a bacillomycin D-like cyclic lipopeptide. The antifungal activity of the compound was found to be due to its inhibitory effect on β-1,3-glucan biosynthesis, a major fungal cell-wall component. Thus, we describe here the identification and characterization of a novel strain of Bacillus amyloliquefaciens (fiply 3A) which produces antifungal lipopeptide and the deciphering of its mechanism of action.


Similar content being viewed by others
References
Albert RA, Archambault J, Rosselló-Mora R, Tindall BJ, Mathen M (2005) Bacillus acidicola sp. nov., a novel mesophilic, acidophilic species isolated from acidic Sphagnum peat bogs in Wisconsin. Int J Syst Evol Microbiol 55:2125–2130
Barchiesi F, Colombo AL, McGough DA, Rinaldi MG (1994) Comparative study of broth macrodilution and microdilution techniques for in vitro antifungal susceptibility testing of yeasts by using the National Committee for Clinical Laboratory proposed standard. J Clin Microbiol 32(10):2494–2500
Basurto-Cadena MGL, Vázquez-Arista M, García-Jiménez J, Salcedo-Hernández R, Bideshi DK, Barboza-Corona JE (2012) Isolation of a new Mexican strain of Bacillus subtilis with antifungal and antibacterial activities. Sci World J 2012:7. doi:10.1100/2012/384978
Bowman SM, Free SJ (2006) The structure and synthesis of the fungal cell wall. BioEssays 28:799–808
Carrillo C, Teruel JA, Aranda FJ, Ortiz A (2003) Molecular mechanism of membrane permeabilization by the peptide antibiotic surfactin. Biochim Biophys Acta 1611:91–97
Chen XH, Koumoutsi A, Scholz R, Eisenreich A, Schneider K, Heinemeyer I, Morgenstern B, Voss B, Hess WR, Reva O, Junge H, Voigt B, Jungblut PR, Vater J, Süssmuth R, Liesegang H, Strittmatter A, Gottschalk G, Borriss R (2007) Comparative analysis of the complete genome sequence of the plant growth–promoting bacterium Bacillus amyloliquefaciens FZB42. Nat Biotechnol 25(9):1007–1014
Chen LL, Wang N, Wang XM, Hu JC, Wang SJ (2010) Characterization of two anti-fungal lipopeptides produced by Bacillus amyloliquefaciens SH-B10. Bioresour Technol 101:8822–8827
Cui T-B, Chai H-Y, Jiang L-X (2012) Isolation and partial characterization of an antifungal protein produced by Bacillus licheniformis BS-3. Molecules 17:7336–7347
Ellis JG, Dodds PN, Lawrence GJ (2007) The role of secreted proteins in diseases of plants caused by rust, powdery mildew and smut fungi. Curr Opin Microbiol 10:326–331
Free S (2013) Fungal cell wall organization and biosynthesis. Adv Genet 81:33–82
Gong Q, Zhang C, Lu F, Zhao H, Bie X, Lu Z (2014) Identification of bacillomycin D from Bacillus subtilis fmbj and its inhibition effects against Aspergillus flavus. Food Control 36:8–14
Hajare SN, Subramanian M, Gautam S, Sharma A (2013) Induction of apoptosis in human cancer cells by a Bacillus lipopeptide bacillomycin D. Biochimie 95(9):1722–1731
Hassan MN, Osborn AM, Hafeez FY (2010) Molecular and biochemical characterization of surfactin producing Bacillus species antagonistic to Colletotrichum falcatum Went causing sugarcane red rot. Afr J Microbiol Res 4(20):2137–2142
Huang A, Edwards F, Bernard EM, Armstrong D, Schmitt HJ (1990) In vitro activity of the new semi-synthetic polypeptide cilofungin (LY121019) against Aspergillus and Candida species. Eur J Clin Microbiol Infect Dis 9:697–699
Janisiewicz WJ, Korsten L (2002) Biological control of postharvest diseases of fruits. Annu Rev Phytopathol 40:411–441
Kabak B (2010) Prevention and Management of Mycotoxins in Food and Feed. In: Rai M, Varma A (eds) Mycotoxins in food, feed and bioweapons. Springer, Berlin, Heidelberg
Kim PI, Bai H, Bai D, Chae H, Chung S, Kim Y, Park R, Chi Y-T (2004) Purification and characterization of a lipopeptide produced by Bacillus thuringiensis CMB26. J Appl Microbiol 97:942–949
Kumar SN, Sreekala SR, Chandrasekaran D, Nambisan B, Anto RJ (2014) Biocontrol of Aspergillus species on peanut kernels by antifungal diketopiperazine producing Bacillus cereus associated with entomopathogenic nematode. PLoS One 9(8): e106041. doi:10.1371/journal.pone.0106041
Kurtz MB, Heath IB, Marrinan J, Dreikorn S, Onishi J, Douglas C (1994) Morphological effects of lipopeptides against Aspergillus fumigatus correlate with activities against (1,3)-beta-D-glucan synthase. Antimicrob Agents Chemother 38:1480–1489
Lee HA, Kim JH (2012) Isolation of Bacillus amyloliquefaciens strains with antifungal activities from Meju. Prev Nutr Food Sci 17:64–70
Leelasuphakul W, Hemmanee P, Chuenchitt S (2008) Growth inhibitory properties of Bacillus subtilis strains and their metabolites against the green mold pathogen (Penicillium digitatum Sacc.) of citrus fruit. Postharvest Biol Technol 48:113–121
Martinez-Absalon SC, del Orozco-Mosqueda MC, Martinez-Pacheco MM, Farias-Rodriguez R, Govindappa M, Santoyo G (2012) Isolation and molecular characterization of a novel strain of Bacillus with antifungal activity from the sorghum rhizosphere. Genet Mol Res 11(3):2665–2673
Mercier J, Jiménez JI (2004) Control of fungal decay of apples and peaches by the biofumigant fungus Muscodor albus. Postharvest Biol Technol 31:1–8
Momany M, Lindsey R, Hill TW, Richardson EA, Momany C, Pedreira M, Guest GM, Fisher JF, Hessler RB, Roberts KA (2004) The Aspergillus fumigatus cell wall is organized in domains that are remodelled during polarity establishment. Microbiology 150:3261–3268
Peypoux F, Bonmatin JM, Wallach J (1999) Recent trends in the biochemistry of surfactin. Appl Microbiol Biotechnol 51:553–563
Pujol I, Guarro J, Llop C, Soler L, Fernandez-Ballart J (1996) Comparison study of broth macrodilution and microdilution antifungal susceptibility tests for the filamentous fungi. Antimicrob Agents Chemother 40(9):2106–2110
Quiroga EN, Sampietro AR, Vattuone MA (2001) Screening antifungal activities of selected medicinal plants. J Ethnopharmacol 74:89–96
Ramage G, Roberston SN, Williams C (2014) Strength in numbers: antifungal strategies against fungal biofilms. Int J Antimicrob Agents 43(2):114–120
Romano A, Vitullo D, Senatore M, Lima G, Lanzotti V (2013) Antifungal cyclic lipopeptides from Bacillus amyloliquefaciens strain BO5A. J Nat Prod 76:2019–2025
Ruiz-García C, Béjar V, Martínez-Checa F, Llamas I, Quesada E (2005) Bacillus velezensis sp. nov., a surfactant-producing bacterium isolated from the river Vélez in Málaga, southern Spain. Int J Syst Evol Microbiol 55:191–195
Souto GI, Correa OS, Montecchia MS, Kerber NL, Pucheu NL, Bachur M, Garcia AF (2004) Genetic and functional characterization of a Bacillus sp. strain excreting surfactin and antifungal metabolites partially identified as iturin-like compounds. J Appl Microbiol 97:1247–1256
Tabbene O, Kalai L, Ben Slimene I, Karkouch I, Elkahoui S, Gharbi A, Cosette P, Mangoni ML, Jouenne T, Limam F (2011) Anti-candida effect of bacillomycin D-like lipopeptides from Bacillus subtilis B38. FEMS Microbiol Lett 316(2):108–114
Thasana N, Prapagdee B, Rangkadilok N, Sallabhan R, Aye SL, Ruchirawat S, Loprasert S (2010) Bacillus subtilis SSE4 produces subtulene A, a new lipopeptide antibiotic possessing an unusual C15 unsaturated beta-amino acid. FEBS Lett 584:3209–3214
Van den Velde S, Lagrou K, Desmet K, Wauters G, Verhaegen J (2006) Species identification of Corynebacteria by cellular fatty acid analysis. Diagn Microbiol Infect Dis 54(2):99–104
Velasco J (1975) Fluorometric measurement of aflatoxin adsorbed on florisil in minicolumns. J Assoc Off Anal Chem 58:757–763
Wan T-C, Cheng F-Y, Liu Y-T, Wang C-M, Shyu C-L, Chen C-M, Lin L-C, Sakata R (2008) Identification of a novel Bacillus species isolated from Calculus bovis. Anim Sci J 79:693–698
Wang L-T, Lee F-L, Tai C-J, Kuo H-P (2008) Bacillus velezensis is a later heterotypic synonym of Bacillus amyloliquefaciens. Int J Syst Evol Microbiol 58:671–675
Wingard JR, Leather H (2004) A new era of antifungal therapy. Biol Blood Marrow Transplant 10:73–90
Yang L, Quan X, Xue B, Goodwin PH, Lu S, Wang J, Wei D, Wu C (2015) Isolation and identification of Bacillus subtilis strain YB-05 and its antifungal substances showing antagonism against Gaeumannomyces graminis var. tritici. Biol Control 85:52–58
Yavuz E, Gunes H, Harsa S, Bulut C, Yenidunya AF (2004) Optimization of pulsed field gel electrophoresis (PFGE) conditions for thermophilic bacilli. World J Microb Biot 20:871–874
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 27000 kb)
Rights and permissions
About this article
Cite this article
Hajare, S.N., Gautam, S. & Sharma, A. A novel strain of Bacillus amyloliquefaciens displaying broad spectrum antifungal activity and its underlying mechanism. Ann Microbiol 66, 407–416 (2016). https://doi.org/10.1007/s13213-015-1123-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-015-1123-0