Abstract
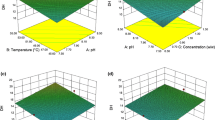
Shrimp waste is an important source of astaxanthin, which occur as a complex with proteins, and protein isolates as well as carotenoids are known to possess antioxidant activity. Investigations were carried out to optimize hydrolysis of shrimp waste using a bacterial protease to obtain antioxidant activity rich protein isolate. The effect of three process variables namely enzyme concentration to waste, incubation temperature and time on carotenoid recovery, protein content, trichloro acetic acid (TCA) soluble peptide content and DiPhenyl Picryl Hydrazylchloride (DPPH) scavenging activity was evaluated using a fractionally factorial design. A high correlation coefficient (>0.90) between the observed and the predicted values indicated the appropriateness of the design employed. Maximum carotenoid recovery was obtained by hydrolysing the shrimp waste with 0.3 % enzyme for 4 h. DPPH radical scavenging activity of carotenoprotein isolate was markedly affected by enzyme concentration, temperature and time of hydrolysis. The study indicated that in order to obtain the carotenoprotein from shrimp waste with higher carotenoid content hydrolysing with an enzyme concentration of 0.2–0.4 %, at lower temperature of 25–30° upto 4 h is ideal. However, in order to obtain the protein isolate with increased antioxidant activity hydrolysing at higher temperature of 50 °C, with higher enzyme concentration of 0.5 % for shorter duration is more ideal.





Similar content being viewed by others
References
Aoki H, Ahsan M, Matsuo K, Hagiwara T, Watabe S (2004) Partial Purification of proteases that are generated by processing of the Northern shrimp Pandalus borealis and which can tenderize meat. Int J Food Sci Technol 39:471–480
Armenta RE, Guerrero-Legarreta I (2009) Amino acid profile and enhancement of the enzymatic hydrolysis of fermented shrimp carotenoproteins. Food Chem 112:310–315
Bhaskar N, Suresh PV, Sakhare PZ, Lalitha RG, Sachindra NM (2010) Yield and chemical composition of fractions obtained from fermented shrimp biowaste. Waste Manag Res 28:64–70
Binsan W, Benjakul S, Visessanguan W, Roytrakul S, Tanaka M, Kisimura H (2008) Antioxidative activity of mungoong an extract paste from the cephalothorax of white shrimp (Litopenaeus vannamei). Food Chem 106:185–193
Cano-Lopez A, Simpson BK, Haard NF (1987) Extraction of carotenoprotein from shrimp process wastes with the aid of trypsin from Atlantic cod. J Food Sci 52:503–506
Cao W, Zhang C, Hong P, Ji H (2008) Response surface methodology for autolysis parameters optimization of shrimp head and amino acids released during autolysis. Food Chem 109:176–183
Cao W, Zhang C, Hong P, Ji H, Hao J, Zhang J (2009) Autolysis of shrimp head by gradual temperature and nutritional quality of the resulting hydrolysate. LWT Food Sci Technol 42:244–249
Duan XJ, Zhang WW, Li XM, Wang BG (2006) Evaluation of antioxidant property of extract and fractions obtained from red alga Polysiphonia urceolata. Food Chem 95:37–43
Ghidalia W (1985) Structural and biological aspects of pigments. In: Bliss DE, Mantel LH (eds) The biology of crustacea, vol 9, Integument pigment and hormonal processes. Academic, New York, pp 301–394
Heu MS, Kim JS, Shahidi F, Jeong YH, Jeon YJ (2003) Characteristics of protease from shrimp processing discards. J Food Biochem 27:221–236
Holanda HD, Netto FM (2006) Recovery of components from shrimp (Xiphopenaeus kroyeri) processing waste by enzymatic hydrolysis. J Food Sci 71:298–303
Huang G, Zhao J, Jiang J (2011) Effect of defatting and enzyme type on antioxidative activity of shrimp processing byproducts hydrolysate. Food Sci Biotechnol 20:651–657
Klomklao S, Benjakul S, Visessanguan W, Kishimura H, Simpson BK (2009) Extraction of carotenoprotein from black tiger shrimp shells with the aid of bluefish trypsin. J Food Biochem 33:201–217
Klompong V, Benjakul S, Kanatachote D, Shahidi F (2006) Antioxidative activity and functional properties of protein hydrolysate of yellow stripe trevally (Selaroides leptolepis) as influenced by the degree of hydrolysis and enzyme type. Food Chem 102:1317–1327
Kristinsson HG, Rasco BA (2000) Fish protein hydrolysates: production biochemical and functional properties. Crit Rev Food Sci Nutr 40:43–81
Lowry OH, Fan AL, Randall RJ, Rosebrough NJ (1951) Protein measurement with Folin Phenol reagent. J Biol Chem 193:265–275
Manni L, Ghorbel-Bellaaj O, Jellouli K, Younes I, Nasri M (2010) Extraction and characterization of chitin chitosan and protein hydrolysates prepared from shrimp waste by treatment with crude protease from Bacillus cereus SV1. Appl Biochem Biotechnol 162:345–357
Manu-Tawai W, Haard NF (1987) Recovery of carotenoprotein from the exoskeleton of snow crab Chinocetes opilio. Can Inst Food Sci Technol J 20:31–33
Marcuse R (1962) The effect of some amino acids on the oxidation of linoleic acid and its methyl ester. J Am Oil Chem Soc 39:97–103
Meenata K, Sowmya R, Rathinaraj K, Sachindra NM (2011) Antioxidant activity of carotenoprotein isolate from shrimp processing discards. J Aquac Food Prod Technol 20:209–221
Sachindra NM, Bhaskar N (2008) In-vitro antioxidant activity of liquor from fermented shrimp biowaste. Bioresour Technol 99:9013–9016
Sachindra NM, Mahendrakar NS (2005) Extractability of carotenoids from shrimp waste in vegetable oils and process optimization. Bioresour Technol 96:1195–1200
Sachindra NM, Mahendrakar NS (2010) Stability of carotenoids recovered from shrimp waste and their use as colorant in fish sausage. J Food Sci Technol 47:77–83
Sachindra NM, Mahendrakar NS (2011) Effect of protease treatment on oil extractability of carotenoids from shrimp waste. J Aquac Food Prod Technol 20:22–31
Sachindra NM, Bhaskar N, Mahendrakar NS (2005) Carotenoids in different body components of Indian shrimps. J Sci Food Agric 85:167–172
Sachindra NM, Bhaskar N, Mahendrakar NS (2006a) Carotenoids in Solonocera indica and Aristeus alcocki, deep-sea shrimps from Indian waters. J Aquac Food Prod Technol 15:5–16
Sachindra NM, Bhaskar N, Mahendrakar NS (2006b) Recovery of carotenoids from shrimp waste in organic solvents. Waste Manage 26:1092–1098
Sachindra NM, Bhaskar N, Siddegowda GS, Sathisha AD, Suresh PV (2007a) Recovery of carotenoids from ensiled shrimp waste. Bioresour Technol 98:1642–1646
Sachindra NM, Hosokawa M, Miyashita K (2007b) Biofunctions of marine carotenoids. In: Hou CT, Shaw J (eds) Biocatalysis and biotechnology for functional foods and industrial products. CRC Press, NewYork, pp 91–110
Seymour TA, Li SJ, Morrissey MT (1996) Characterisation of a natural antioxidant from shrimp shell waste. J Agri Food Chem 44:682–685
Shahidi F, Metusalach, Brown JA (1998) Carotenoid pigments in seafoods and aquaculture. CRC Crit Rev Food Sci 38:1–67
Simpson BK, Haard NF (1985) The use of enzymes to extract carotenoprotein from shrimp waste. J Appl Biochem 7:212–222
Sowmya R, Rathinaraj K, Sachindra NM (2011) An autolytic process for recovery of antioxidant activity rich carotenoprotein from shrimp heads. Mar Biotechnol 13:918–927
Statsoft (1999) Statistics for windows. Statsoft Inc, Tulsa
Suetsuna K (2000) Antioxidant peptides from the protease digest of prawn (Penaeus japonicus) muscle. Mar Biotechnol 2:5–10
Wu HC, Chen HM, Shiau CY (2003) Free amino acids and peptides as related to antioxidant properties in protein hydrolysates of mackeral (Scomber austriasicus). Food Res Int 36:949–957
Zagalsky PF (1985) Invertebrate carotenoproteins. Methods Enzymol 111:216–247
Zagalsky PF, Eliopoulos EE, Findlay JBC (1990) The architecture of invertebrate carotenoproteins. Comp Biochem Physiol 97B:1–18
Acknowledgments
Authors wish to thank Director, CFTRI for his encouragement and for the facilities provided. This study formed a part of the project funded by Department of Biotechnology, Govt. of India.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sowmya, R., Ravikumar, T.M., Vivek, R. et al. Optimization of enzymatic hydrolysis of shrimp waste for recovery of antioxidant activity rich protein isolate. J Food Sci Technol 51, 3199–3207 (2014). https://doi.org/10.1007/s13197-012-0815-8
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-012-0815-8