Abstract
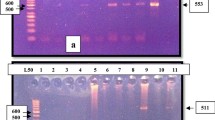
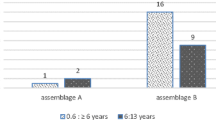
Giardiasis is a prevailing intestinal disease in children. This study aimed to determine molecular prevalence of Giardia intestinalis in children attending Cairo University Pediatrics Hospitals, using copro-PCR assays, conventional methods and to evaluate diagnostic effectiveness of used tests. 229 fecal samples were collected from children suffering from gastrointestinal symptoms and examined for Giardia by microscopy, Immuno-chromatographic test (ICT), copro-DNA using two PCR assays targeting tpi [nested-PCR (nPCR)] and 18S [conventional-PCR (cPCR)] genes. Out of 229 samples assessed, Giardia was diagnosed in 13.9, 17, 17.9, 4.8 % of cases using microscopy, ICT, nPCR (tpi) and cPCR (18S), respectively. Nominating both PCR assays as composite reference standard, microscopy and ICT were of reliable specificity (100 and 96.9 %) and accuracy (95.6 and 93.6 %) but of limited sensitivity (78.6 and 76.2 %). Kappa agreement showed, there was substantial agreement of ICT (0.776) and almost perfect agreement of microscopy (0.839) with PCR assays. Giardia showed a molecular prevalence of 18.3 % (42/229). ICT assay for Giardia surpassed microscopy but both couldn’t be used as a consistent single detection method due to their lowered sensitivities. nPCR targeting tpi is a reliable diagnostic test aiding to determine true prevalence of Giardia.

Similar content being viewed by others
References
Alonzo TA, Pepe MS (1999) Using a combination of reference tests to assess the accuracy of a new diagnostic test. Stat Med 18:2987–3003
Al-Saeed AT, Issa SH (2010) Detection of Giardia lamblia antigen in stool specimens using enzyme-linked immunosorbent assay. East Med Health J 16(4):362–364
Amar CF, Dear PH, Pedaza-Diaz S, Looker N, Linnane E, McLauchlin J (2002) Sensitive PCR-RFLP assay for detection and genotyping of Giardia duodenalis in human feces. Clin Microbiol J 40(2):446–452
Amjad IA, Hussein AB, Yamaguchi T, Nakamoto K, Motohiro Iseki M, Tokoro M (2009) Multiple-subgenotype infections of Giardia intestinalis detected in Palestinian clinical cases using a subcloning approach. Parasitol Internat 58:258–262
Bertrand I, Albertini L, Schwartzbord A (2005) Comparison of two target genes for detection and genotyping of Giardia lamblia in human feces by PCR and PCR-restriction fragment length polymorphism. J Clin Microbiol 43(12):5940–5944
Elsafi SH, Al-Maqati TN, Hussein MI, Adam AA, Hassan MM, Al Zahrani EM (2013) Comparison of microscopy, rapid immunoassay, and molecular techniques for the detection of Giardia lamblia and Cryptosporidium parvum. Parasitol Res 112(4):1641–1646
Farthing MJ, Mata L, Urrutia JJ, Kronmal RA (1986) Natural history of Giardia infection of infants and children in rural Guatemala and its impact on physical growth. Am J Clin Nutr 43(3):395–405
Fawzi M, El-Sahn AA, Ibrahim HF, Shehata AI (2004) Vegetable transmitted parasites among inhabitants of El-Prince, Alexandria and its relation to housewives’ knowledge and practices. J Egypt Public Health Assoc 79:13–29
Foronda PM, Argues N, Abreu-Acosta MV, Periago MA, Valero BV, Mas-coma S (2008) Identification of genotypes of Giardia intestinalis of human isolates in Egypt. Parasitol Res 103:1177–1181
Gelanew T, Lalle M, Hailu A, Pozio E, Caccio SM (2007) Molecular characterization of human isolates of Giardia duodenalis from Ethiopia. Acta Trop 102:92–99
Goñi P, Martín B, Villacampa M, García A, Seral C, Castillo FJ, Clavel A (2012) Evaluation of an immunochromatographic dip strip test for simultaneous detection of Cryptosporidium spp, Giardia duodenalis, and Entamoeba histolytica antigens in human faecal samples. Eur J Clin Microbiol Infect Dis 31(8):2077–2082
Haque R (2007) Human intestinal parasites. J Health Popul Nutr 25:387–391
Helmy MM, Abdel-Fattah HS, Rashed L (2009) Realtime PCR-RFLP assay to detect Giardia intestinalis genotypes in human isolates with diarrhea in Egypt. J Parasitol 95(4):1–5
Helmy YA, Krücken J, Nöckler K, Samson-Himmelstjerna GV, Zessin KH (2014) Comparison between two commercially available serological tests and polymerase chain reaction in the diagnosis of Cryptosporidium in animals and diarrhoeic children. Parasitol Res 113(1):211–216 (See comment in PubMed Commons below)
Ignatius R, Gahutu JB, Klotz C, Musemakweri A, Aebischer T, Mockenhaupt FP (2014) Detection of Giardia duodenalis assemblage A and B isolates by immunochromatography in stool samples from Rwandan children. Clin Microbiol Infect 1111(10)/1469-0691
Lalle M, Bruschi F, Castagna B, Campa M, Pozio E, Cacciò SM (2009) High genetic polymorphism among Giardia duodenalis isolates from Sahrawi children. Trans R Soc Trop Med Hyg 103(8):834–838
Levy DA, Bens MS, Craun GF, Calderon RL, Herwaldt BL (1998) Surveillance for waterborne-disease outbreaks United States, 1995–1996. CDC Surveill Summ MMWR 47(5):1–34
Mank TG, Zaat JM, Deelder AM, Eijk JThMV, Polderman AM (1997) Sensitivity of microscopy versus enzyme immunoassay in the laboratory diagnosis of Giardiasis. Eur J Clin Microbiol Infect 16:615–619
Monis PT, Andrews RH, Mayrhofer G, Ey PL (1999) Molecular systematic of the parasitic protozoan Giardia intestinalis. Mol Biol Evol 16:1135–1144
Nahavandi KH, Fallah E, Asgharzadeh M, Mirsamadi N, Mahdavipour B (2011) Glutamate dehydrogenase and triose-phosphate-isomerase coding genes for detection and genetic characterization of Giardia lamblia in human feces by PCR and PCR-RFLP. Turk J Med Sci 41(2):283–289
Natividad FF, Buerano CC, Lago CB, Mapua CA, de Guzman BB, Seraspe EB, Samentar LP, Endo T (2008) Prevalence rates of Giardia and Cryptosporidium among diarrheic patients in the Philippines. Southeast Asian J Trop 39(6):991–999
Norhayati M, Fatmah MS, Yusof S (2003) Intestinal parasitic infections in man. A review. Med J Malays 58:2–10
Sadek GS, El-Settawy MA, Soha A, Nasr SA (2013) Genotypic characterization of Giardia duodenalis in children in Menoufiya and Sharkiya governorates. Egypt Life Sci J 10(1):4006–4015
Sulaiman IM, Fayer R, Bern C, Gilman RH, Trout JM, Schantz PM (2003) Triosephosphate isomerase gene characterization and potential zoonotic transmission of Giardia duodenalis. Emerg Infect Dis 9(11):1444–1452
Thompson RC, Smith A (2011) Zoonotic enteric protozoa. Vet Parasitol 182:70–78
Thompson RC, Hopkins RM, Homan WL (2000) Nomenclature and genetic groupings of Giardia infecting mammals. Parasitol Today 16:210–213
Verweij JJ, Blangé RA, Templeton K, Schinkel J, Brienen EA, Van Rooyen MA (2004) Simultaneous detection of Entamoeba histolytica Giardia lamblia and Cryptosporidium parvum in fecal samples by using multiplex real-time PCR. J Clin Microbiol 42(3):1220–1223
Volotão AC, Costa-Macedo LM, Haddad FS, Brandão A, Peralta JM, Fernades O (2007) Genotyping of Giardia duodenalis from human and animal samples from Brazil using β-giardin gene a phylogenetic analysis. Acta Trop 102(1):258–262
Zheng G, Alsarakibi M, Liu Y, Hu W, Luo Q, Tan L, Li G (2014) Genotyping of Giardia duodenalis isolates from dogs in Guangdong, China based on multi-locus sequence. Korean J Parasitol 52(3):299–304
Acknowledgments
We are grateful to the Scientific Research Developing Unit, Bani-suef University for granting and funding the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no reported conflicts of interest.
Author contribution
All manuscript authors contributed to every activity of it; idea of paper, study design, collection of materials, methodology, writing the paper and revising it.
Rights and permissions
About this article
Cite this article
Ghieth, M.A., Kotb, M.A., Abu-Sarea, E.Y. et al. Molecular detection of giardiasis among children at Cairo University Pediatrics Hospitals. J Parasit Dis 40, 1470–1474 (2016). https://doi.org/10.1007/s12639-015-0714-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-015-0714-9