Abstract
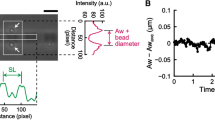
SPOC (spontaneous oscillatory contraction) is a characteristic state of the contractile system of striated (skeletal and cardiac) muscle that exists between the states of relaxation and contraction. For example, Ca-SPOCs occur at physiological Ca2+ levels (pCa ∼6.0), whereas ADP-SPOC occurs in the virtual absence of Ca2+ (pCa ≥ 8; relaxing conditions in the presence of MgATP), but in the presence of inorganic phosphate (Pi) and a high concentration of MgADP. The concentration of Mg-ADP necessary for SPOC is nearly equal to or greater than the MgATP concentration for cardiac muscle and is several times higher for skeletal muscle. Thus, the cellular conditions for SPOC are broader in cardiac muscle than in skeletal muscle. During these SPOCs, each sarcomere in a myofibril undergoes length oscillation that has a saw-tooth waveform consisting of a rapid lengthening and a slow shortening phase. The lengthening phase of one half of a sarcomere is transmitted to the adjacent half of the sarcomere successively, forming a propagating wave (termed a SPOC wave). The SPOC waves are synchronized across the cardiomyocytes resulting in a visible wave of successive contractions and relaxations termed the SPOC wave. Experimentally, the SPOC period (and therefore the velocity of SPOC wave) is observed in demembranated cardiomyocytes and can be prepared from a wide range of animal hearts. These periods correlate well with the resting heartbeats of a wide range of mammals (rat, rabbit, dog, pig and cow). Preliminary experiments showed that the SPOC properties of human cardiomyocytes are similar to the heartbeat of a large dog or a pig. This correlation suggests that SPOCs may play a fundamental role in the heart. Here, we briefly summarize a range of SPOC parameters obtained experimentally, and relate them to a theoretical model to explain those characteristics. Finally, we discuss the possible significance of these SPOC properties in each and every heartbeat.





Similar content being viewed by others
References
Anazawa T, Yasuda K, Ishiwata S (1992) Spontaneous oscillation of tension and sarcomere length in skeletal myofibrils. Microscopic measurement and analysis. Biophys J 61:1099–1108
Barany M (1967) ATPase activity of myosin correlated with speed of muscle shortening. J Gen Physiol 50:197–218
Bers DM (2001) In Excitation-contraction coupling and cardiac contractile force. 2nd. Kluwer-Academic, Dordrecht
Bremel RD, Weber A (1972) Cooperation within actin filament in vertebrate skeletal muscle. Nat New Biol 238:97–101
Brenner B (1979) An indirect proof of stretch-induced Ca2+ release from the sarcoplasmic reticulum in glycerinated skeletal and heart muscle preparations. Basic Res Cardiol 74:177–202
Cooke R, Pate E (1985) The effects of ADP and phosphate on the contraction of muscle fibers. Biophys J 48:789–798
Ebashi S, Endo M (1968) Calcium ions and muscle contraction. Prog Biophys Mol Biol 18:123–183
Ebashi S, Endo M, Ohtsuki I (1969) Control of muscle contraction. Q Rev Biophys 2:351–384
Endo M (1972a) Stretch-induced increase in activation of skinned muscle fibers by calcium. Nat New Biol 237:211–213
Endo M (1972b) Length dependence of activation of skinned muscle fibers by calcium. Cold Spring Harb Symp Quant Biol 37:505–510
Fabiato A, Fabiato F (1978) Myofilament-generated tension oscillations during partial calcium activation and activation dependence of the sarcomere length-tension relation of skinned cardiac cells. J Gen Physiol 72:667–699
Fuchs F, Wang YP (1996) Sarcomere length versus interfilament spacing as determinants of cardiac myofilament Ca2+ sensitivity and Ca2+ binding. J Mol Cell Cardiol 28:1375–1383
Fujita H, Ishiwata S (1998) Spontaneous oscillatory contraction without regulatory proteins in actin filament-reconstituted fibers. Biophys J 75:1439–1445
Fujita H, Yasuda K, Niitsu S, Funatsu T, Ishiwata S (1996) Structural and functional reconstitution of thin filaments in the contractile apparatus of cardiac muscle. Biophys J 71:2307–2318
Fukuda N, Ishiwata S (1999) Effects of pH on spontaneous tension oscillation in skinned bovine cardiac muscle. Pflugers Arch 438:125–132
Fukuda N, Fujita H, Fujita T, Ishiwata S (1996) Spontaneous tension oscillation in skinned bovine cardiac muscle. Pflugers Arch 433:1–8
Fukuda N, Fujita H, Fujita T, Ishiwata S (1998) Regulatory roles of MgADP and calcium in tension development of skinned cardiac muscle. J Muscle Res Cell Motil 19:909–921
Fukuda N, Sasaki D, Ishiwata S, Kurihara S (2001) Length dependence of tension generation in rat skinned cardiac muscle. Role of titin in the Frank–Starling mechanism of the heart. Circulation 104:1639–1645
Fukuda N, Granzier HL, Ishiwata S, Kurihara S (2008) Physiological functions of the giant elastic protein titin in mammalian striated muscle. J Physiol Sci 58:151–159
Funatsu T, Higuchi H, Ishiwata S (1990) Elastic filaments in skeletal muscle revealed by selective removal of thin filaments with plasma gelsolin. J Cell Biol 110:53–62
Funatsu T, Kono E, Higuchi H, Kimura S, Ishiwata S, Yoshioka T, Maruyama K, Tsukita S (1993) Elastic filaments in situ in cardiac muscle: deep-etch replica analysis in combination with selective removal of actin and myosin filaments. J Cell Biol 120:711–724
Günther S, Kruse K (2007) Spontaneous sarcomere dynamics. Chaos 20:045122
Huxley AF (1957) Muscle structure and theories of contraction. Prog Biophys Biophys Chem 7:255–318
Huxley HE, Hanson J (1954) Changes in the cross-striations of muscle during contraction and stretch and their structural interpretation. Nature 173:973–977
Huxley AF, Niedergerke R (1954) Structural changes in muscle during contraction: Interference microscopy of living muscle fibres. Nature 173:971–973
Inoue T, Kobirumaki-Shimozawa F, Kagemoto T, Fujii T, Terui T, Kusakari Y, Hongo K, Morimoto S, Ohtsuki I, Hashimoto K, Fukuda N (2013) Depressed Frank-Starling mechanism in the left ventricular muscle of the knock-in mouse model of dilated cardiomyopathy with troponin T deletion mutation ΔK210. J Mol Cell Cardiol 63:69–78
Ishiwata S, Yasuda K (1993) Mechano-chemical coupling in spontaneous oscillatory contraction of muscle. Phase Transit 45:105–136
Ishiwata S, Yasuda K, Shindo Y, Fujita H (1996) Microscopic analysis of the elastic properties of connectin/titin and nebulin in myofibrils. Adv Biophys 33:135–142
Ishiwata S, Shimamoto Y, Suzuki M (2010) Molecular motors as an auto-oscillator. HFSP J 4:100–104
Ishiwata S, Shimamoto Y, Fukuda N (2011) Contractile system of muscle as an auto-oscillator. Prog Biophys Mol Biol 105:187–198. This is the most detailed review summarizing the characteristics of SPOC mainly experimentally obtained until 2011.
Jülicher F, Prost J (1997) Spontaneous oscillations of collective motor units. Phys Rev Lett 78:4510–4513
Kobirumaki-Shimozawa F, Oyama K, Serizawa T, Mizuno A, Kagemoto T, Shimozawa T, Ishiwata S, Kurihara S, Fukuda N (2012) Sarcomere imaging by quantum dots for the study of cardiac muscle physiology. J Biomed Biotechnol 2012:1–7
Kobirumaki-Shimozawa F, Inoue T, Shintani SA, Oyama K, Terui T, Minamisawa S, Ishiwata S, Fukuda N (2014) Cardiac thin filament regulation and the Frank–Starling mechanism. J Physiol Sci 64:221–232
Linke WA, Bartoo ML, Pollack GH (1993) Spontaneous sarcomeric oscillations at intermediate activation levels in single isolated cardiac myofibrils. Circ Res 73:724–734
Malmqvist UP, Aronshtam A, Lowey S (2004) Cardiac myosin isoforms from different species have unique enzymatic and mechanical properties. Biochemistry 43:15058–15065
Maruyama K, Natori R, Nonomura Y (1976) New elastic protein from muscle. Nature 262:58–60
Okamura N, Ishiwata S (1988) Spontaneous oscillatory contraction of sarcomeres in skeletal myofibrils. J Muscle Res Cell Motil 9:111–119
Sasaki D, Fujita H, Fukuda N, Kurihara S, Ishiwata S (2005) Auto-oscillations of skinned myocardium correlating with heartbeat. J Muscle Res Cell Motil 26:93–101
Sasaki D, Fukuda N, Ishiwata S (2006) Myocardial sarcomeres spontaneously oscillate with the period of heartbeat under physiological conditions. Biochem Biophys Res Commun 343:1146–1152
Sato K, Ohtaki M, Shimamoto Y, Ishiwata S (2011) A theory on auto-oscillation and contraction in striated muscle. Prog Biophys Mol Biol 105:199–207. This paper proposed the concrete model that has succeeded in explaining almost all the SPOC properties, i.e., the oscillation waveform and the phase diagram, on a single sarcomere (termed a unit-model).
Sato K, Kuramoto Y, Ohtaki M, Shimamoto Y, Ishiwata S (2013) Locally and globally coupled oscillators in muscle. Phys Rev Lett 111:108104. This is a paper that presented the model to explain the SPOC properties, i.e., the oscillation pattern (SPOC wave), on a single myofibril (termed a connected model). This model is an extension of the unit model that was visco-elastically connected in series (Sato et al., 2011).
Serizawa T, Terui T, Kagemoto T, Mizuno A, Shimozawa T, Kobirumaki F, Ishiwata S, Kurihara S, Fukuda N (2011) Real-time measurement of the length of a single sarcomere in rat ventricular myocytes: a novel analysis with quantum dots. Am J Physiol Cell Physiol 301:C1116–C1127
Shimamoto Y, Kono F, Suzuki M, Ishiwata S (2007) Non-linear force-length relationship in the ADP-induced contraction of skeletal myofibrils. Biophys J 93:4330–4341
Shimamoto Y, Suzuki M, Ishiwata S (2008) Length-dependent activation and auto-oscillation in skeletal myofibrils at partial activation by Ca2+. Biochem Biophys Res Commun 366:233–238
Shimamoto Y, Suzuki M, Mikhailenko SV, Yasuda K, Ishiwata S (2009) Inter-sarcomere coordination in muscle revealed through individual sarcomere response to quick stretch. Proc Natl Acad Sci U S A 106:11954–11959
Shimizu H, Fujita T, Ishiwata S (1992) Regulation of tension development by MgADP and Pi without Ca2+. Role in spontaneous tension oscillation of skeletal muscle. Biophys J 61:1087–1098
Shintani SA, Oyama K, Kobirumaki-Shimozawa F, Ohki T, Ishiwata S, Fukuda N (2014) Sarcomere length nanometry in rat neonatal cardiomyocytes expressed with α-actinin-AcGFP in Z-discs. J Gen Physiol 143:513–524
Smith DA, Stephenson DG (2009) The mechanism of spontaneous oscillatory contractions in skeletal muscle. Biophys J 96:3682–3691
Stehle R, Krüger M, Pfitzer G (2002) Force kinetics and individual sarcomere dynamics in cardiac myofibrils after rapid Ca2+ changes. Biophys J 83:2152–2161
Sweitzer NK, Moss RL (1990) The effect of altered temperature on Ca2+-sensitive force in permeabilized myocardium and skeletal muscle. Evidence for force dependence of thin filament activation. J Gen Physiol 96:1221–1245
Terui T, Sodnomtseren M, Matsuba D, Udaka J, Ishiwata S, Ohtsuki I, Kurihara S, Fukuda N (2008) Troponin and titin coordinately regulate length-dependent activation in skinned porcine ventricular muscle. J Gen Physiol 131:275–283
Terui T, Shimamoto Y, Yamane M, Kobirumaki F, Ohtsuki I, Ishiwata S, Kurihara S, Fukuda N (2010) Regulatory mechanism of length-dependent activation in skinned porcine ventricular muscle: role of thin filament cooperative activation in the Frank-Starling relation. J Gen Physiol 136:469–482
Vilfan A, Duke T (2003) Synchronization of active mechanical oscillators by an inertial lode. Phys Rev Lett 91:114101
Wang K, Ramirez-Mitchell R, Palter D (1984) Titin is an extraordinarily long, flexible, and slender myofibrillar protein. Proc Natl Acad Sci USA 81: 3685-3689
Wolfe JE, Ishiwata S, Braet F, Whan R, Su Y, Lal S, dos Remedios CG (2011) SPontaneous Oscillatory Contraction (SPOC): auto-oscillations observed in striated muscle at partial activation. Biophys Rev 3:53–62
Yasuda K, Shindo Y, Ishiwata S (1996) Synchronous behavior of spontaneous oscillations of sarcomeres in skeletal myofibrils under isotonic conditions. Biophys J 70:1823–1829
Compliance with Ethical Standards
ᅟ
Funding
This work was supported in part by Grants-in-Aid for Specially Promoted Research and Scientific Research (S) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan (to SI) and from the National Heart Foundation of Australia (to LA).
Conflict of interest
Tatsuya Kagemoto, Amy Li, Cris dos Remedio, and Shin’ichi Ishiwata declare that they have no conflict of interest.
Ethical approval
The human heart was provided by Sydney Heart Bank directed by Cris dos Remedios, which has been approved by Human Research Ethics Committee of The University of Sydney. This research has also been approved by Human Ethics Committee at Waseda University.
Author contributions
Tatsuya Kagemoto, Amy Li, Cris dos Remedio, and Shin’ichi Ishiwata discussed all contents of this review and prepared the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Special Issue: Biophysics of Human Heart Failure
Rights and permissions
About this article
Cite this article
Kagemoto, T., Li, A., dos Remedios, C. et al. Spontaneous oscillatory contraction (SPOC) in cardiomyocytes. Biophys Rev 7, 15–24 (2015). https://doi.org/10.1007/s12551-015-0165-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-015-0165-7