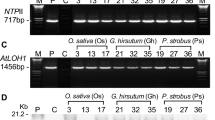
Abstract
ATP-citrate lyase catalyzes cracking citrate to produce acetyl CoA in the cytoplasm and plays an important role in normal growth and development of plants. In this study, SoACLA-1 gene in sugarcane was cloned and characterized. The amino acid sequence of SoACLA-1 showed high homology with other ACLAs families, and they were clustered together in the phylogenetic tree. On transcript level, SoACLA-1 expressed strongly in root and stalk and responded to polyethylene glycol 6000, drought and ABA stresses. The results indicated that SoACLA-1 possibly involves in stress-responsive metabolic process. PCR analysis was performed to confirm the putative transgenic type in T1 tobacco plants. Overexpression of SoACLA-1 enhanced the tolerance of tobacco plants to drought. The sense transgenic tobacco plants were healthier, had longer roots and higher contents of relative water content and chlorophyll and activities of superoxide dismutase, peroxidase and catalase, but lower ion leakage and contents of malondialdehyde (MDA) and H2O2 than the wild-type and antisense transgenic plants under drought stress. In conclusion, SoACLA-1 plays an important role as a positive factor in drought stress response pathways and can be utilized for transformation to improve crop drought tolerance.









Similar content being viewed by others
References
Barrs, H.D., and P.E. Weatherley. 1962. A reexamination of the relative turgidity technique for estimating water deficit in leaves. Australian Journal of Biological Sciences 15: 413–428.
Blokhina, O., E. Virolainen, and K.V. Fagerstedt. 2003. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Annals of Botany 91: 179–194.
Bower, R., and R.G. Birch. 1992. Transgenic sugarcane plants via microprojectile bombardment. Plant Journal 2(3): 409–416.
Chypre, M., N. Zaidi, and K. Smans. 2012. ATP-citrate lyase: A mini-review. Biochemical and Biophysical Research Communications 422: 1–4.
Clouse, S.D. 2002. Arabidopsis mutants reveal multiple roles for sterols in plant development. The Plant Cell 14: 1995–2000.
Dhindsa, R., and W. Matowe. 1981. Drought tolerance in two mosses: Correlated with enzymatic defense against lipid peroxidation. Journal of Experimental Botany 32(1): 79–91.
Elshourbagy, N.A., J.C. Near, P.J. Kmetz, T.N. Wells, P.H. Groot, B.A. Saxty, S.A. Hughes, M. Franklin, and I.S. Gloger. 1992. Cloning and expression of a human ATP-citrate lyase cDNA. European Journal of Biochemistry 204: 491–499.
Farooq, M., A. Wahid, N. Kobayashi, D. Fujita, and S.M.A. Basra. 2009. Plant drought stress: Effects, mechanisms and management. Agronomy for Sustainable Development 29: 185–212.
Fatland, B.L., J. Ke, M.D. Anderson, W.I. Mentzen, L.W. Cui, C.C. Allred, J.L. Johnston, B.J. Nikolau, and E.S. Wurtele. 2002. Molecular characterization of a heteromeric ATP-citrate lyase that generates cytosolic acetyl-coenzyme A in Arabidopsis. Plant Physiology 130: 740–756.
Fatland, B.L., B.J. Nikolau, and E.S. Wurtele. 2005. Reverse genetic characterization of cytosolic acetyl-CoA generation by ATP-citrate lyase in Arabidopsis. The Plant Cell 17: 182–203.
Gurel, S., E. Gurel, R. Kaur, J. Wong, L. Meng, H.Q. Tan, and P.G. Lemaux. 2009. Efficient, reproducible Agrobacterium-mediated transformation of sorghum using heat treatment of immature embryos. Plant Cell Reports 28(3): 429–444.
Heath, R.L., and L. Packer. 1968. Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Archives in Biochemistry and Biophysics 125: 189–198.
Horsch, R.B., J.E. Fry, N.L. Hoffmann, D. Eichholtz, S.C. Rogers, and R.T. Fraley. 1985. A simple and general method for transferring genes into plants. Science 227: 1229–1231.
Hu, H.H., M.Q. Dai, J.L. Yao, B.Z. Xiao, X.H. Li, Q.F. Zhang, and L.Z. Xiong. 2006. Overexpressing a NAM, ATAF, and CUC (NAC) transcription factor enhances drought resistance and salt tolerance in rice. Proceedings of the National Academy of Sciences of the United States of America 103: 12987–12992.
Hu, W., C. Huang, X. Deng, S. Zhou, L. Chen, Y. Li, C. Wang, Z. Ma, Q. Yuan, Y. Wang, R. Cai, X. Liang, G. Yang, and G. He. 2013. TaASR1, a transcription factor gene in wheat, confers drought stress tolerance in transgenic tobacco. Plant, Cell and Environment 36(8): 1449–1464.
Hu, X.M., C.Y. Shi, X. Liu, L.F. Jin, Y.Z. Liu, and S.A. Peng. 2015. Genome-wide identification of citrus ATP-citrate lyase genes and their transcript analysis in fruits reveals their possible role in citrate utilization. Molecular Genetics and Genomics 290: 29–38.
Jaleel, C.A., K. Riadh, R. Gopi, P. Manivannan, J. Inès, H.J. Al-Juburi, C.X. Zhao, H.B. Shao, and R. Panneerselvam. 2009. Antioxidant defense response: Physiological plasticity in higher plants under abiotic constraints. Acta Physiologiae Plantarum 31: 427–436.
Jiang, M.Y., and J.H. Zhang. 2001. Effect of abscisic acid on active oxygen species, antioxidative defense system and oxidative damage in leaves of maize seedlings. Plant and Cell Physiology 42: 1265–1273.
Koziol, C. 2002. Overexpression of ATP citrate lyase in Arabidopsis thaliana. Under the direction of the Wurtele Lab, Iowa State University—Department of Botany, Summer 2002 NSF REU Report in Molecular Biotechnology.
Krause, G., and E. Weis. 1991. Chlorophyll fluorescence and photosynthesis: The basics. Annual Review of Plant Physiology and Plant Molecular Biology 42(1): 313–349.
Langlade, N.B., G. Messerli, L. Weisskopf, S. Plaza, N. Tomasi, J. Smutny, G. Neumann, E. Martinoia, and A. Massonneau. 2002. ATP citrate lyase: Cloning, heterologous expression and possible implication in root organic acid metabolism and excretion. Plant, Cell and Environment 25: 1561–1569.
Li, C.N., N. Qian, Q.L. Tan, K.S. Manoj, L.T. Yang, and Y.R. Li. 2012. Cloning and expression analysis of ATP-citrate lyase genes from sugarcane. Acta Agronamica Sinica 38: 2024–2033.
Li, C.N., M.K. Srivastava, Q. Nong, and Y.R. Li. 2010. Mechanism of tolerance to drought in sugarcane plant enhanced by foliage dressing of abscisic acid under water stress. Acta Agronamica Sinica 36(5): 863–870.
Li, G.W., M.H. Zhang, W.M. Cai, W.N. Sun, and W.A. Su. 2008. Characterization of OsPIP2;7, a water channel protein in rice. Plant and Cell Physiology 49: 1851–1858.
Liu, G., and I.D. Godwin. 2012. Highly efficient sorghum transformation. Plant Cell Reports 31: 999–1007.
Liu, J.Y., Y.R. Li, and L.T. Yang. 2014. Advances in ATP-citrate lyase research. Journal of Southern Agriculture 45(2): 204–208.
Livak, K.J., and T.D. Schmittgen. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 25: 402–408.
Miller, G., N. Suzuki, S. Ciftci-Yilmaz, and R. Mittler. 2010. Reactive oxygen species homeostasis and signaling during drought and salinity stresses. Plant, Cell and Environment 33: 453–457.
Mittler, R., S. Vanderauwera, M. Gollery, and F. Van Breusegem. 2004. Reactive oxygen gene network of plants. Trends in Plant Science 9: 490–498.
Moore, K., and L.J. Roberts. 1998. Measurement of lipid peroxidation. Free Radical Research 28: 659–671.
Murashige, T., and F. Skoog. 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiologia Plantarum 15(3): 473–497.
Nelson, D.R., and R.W. Rinne. 1975. Citrate cleavage enzymes from developing soybean cotyledons incorporation of citrate carbon into fatty acids. Plant Physiology 55: 69–72.
Nikolau, B.J., J.B. Ohlrogge, and E.S. Wurtele. 2003. Plant biotincontaining carboxylases. Archives of Biochemistry and Biophysics 414: 211–222.
Niu, J.Q., A.Q. Wang, J.L. Huang, H. Zhu, Y.R. Li, and L.T. Yang. 2013. Cloning and expression analysis of a soluble acid invertase Gene (SoSAI1) of Sugarcane. Scientia Agricultura Sinica 46(24): 5248–5260.
Oliver, S.N., E.S. Dennis, and R. Dolferus. 2007. ABA regulates apoplastic sugar transport and is a potential signal for cold-induced pollen sterility in rice. Plant and Cell Physiology 48: 1319–1330.
Pang, X.B., X.G. Mao, R.L. Jing, J.F. Shi, T. Gao, X.P. Chang, and Y.F. Li. 2007. Analysis of gene expression profile responses to water stress in wheat (Triticum aestivum L.) seedlings. Acta Agronomica Sinica 33: 333–336.
Polle, A. 2001. Dissection the superoxide dismutase–ascorbate–glutathione pathway by metabolic modeling: Computer analysis as a step towards flux analysis. Plant Physiology 126: 445–462.
Polle, A., T. Otter, and F. Seifert. 1994. Apoplastic peroxidases and lignification in needles of Norway spruce (Picea abies L.). Plant Physiology 106: 53–60.
Rangasamy, D., and C. Ratledge. 2000. Compartmentation of ATP: Citrate lyase in plants. Plant Physiology 122: 1225–1230.
Rogers, S., and A. Bendich. 1985. Extraction of DNA from milligram amounts of fresh, herbarium and mummified plant tissues. Plant Molecular Biology 5(2): 69–76.
Seki, M., M. Narusaka, H. Abe, M. Kasuga, K. Yamaguchi-Shinozaki, P. Carninci, Y. Hayashizaki, and K. Shinozaki. 2001. Monitoring the expression pattern of 1300 Arabidopsis genes under drought and cold stresses by using a full-length cDNA microarray. The Plant Cell 13: 61–72.
Seki, M., M. Narusaka, J. Ishida, T. Nanjo, M. Fujita, Y. Oono, A. Kamiya, M. Nakajima, A. Enju, T. Sakurai, M. Satou, K. Akiyama, T. Taji, K. Yamaguchi-Shinozaki, P. Carninci, J. Kawai, Y. Hayashizaki, and K. Shinozaki. 2002. Monitoring the expression profiles of 7000 Arabidopsis genes under drought, cold, and high-salinity stresses using a full-length cDNA microarray. Plant Journal 31: 279–292.
Shalit, M., I. Guterman, H. Volpin, E. Bar, T. Tamari, N. Menda, Z. Adam, D. Zamir, A. Vainstein, D. Weiss, E. Pichersky, and E. Lewinsohn. 2003. Volatile ester formation in roses: Identification of an acetyl-coenzyme A geraniol/citronellol acetyltransferase in developing rose petals. Plant Physiology 131: 1868–1876.
Shinozaki, K., and K. Yamaguchi-Shinozaki. 1997. Gene expression and signal transduction in water-stress response. Plant Physiology 115: 327–334.
Shinozaki, K., K. Yamaguchi-Shinozaki, and M. Seki. 2003. Regulatory network of gene expression in the drought and cold stress responses. Current Opinion in Plant Biology 6: 410–417.
Sinha, A.K., and M.G. Hofmann. 2002. Metabolizable and non-metabolizable sugars activate different signal transduction pathways in tomato. Plant Physiology 128: 1480–1489.
Souter, M., J. Topping, M. Pullen, J. Friml, K. Palme, R. Hackett, D. Grierson, and K. Lindsey. 2002. Hydra mutants of Arabidopsis are defective in sterol profiles and auxin and ethylene signaling. The Plant Cell 14: 1017–1031.
Suh, M.C. 2001. Pathogen-induced expression of plant ATP:citrate lyase. FEBS Letters 488: 211–212.
Tamura, K., G. Stecher, D. Peterson, A. Filipski, and S. Kumar. 2013. MEGA6: Molecular evolutionary genetics analysis version 6.0. Molecular Biology and Evolution 30(12): 2725–2729.
Xing, S., N. van Deenen, P. Magliano, L. Frahm, E. Forestier, C. Nawrath, H. Schaller, C.S. Gronover, D. Prüfer, and Y. Poirier. 2014. ATP citrate lyase activity is post-translationally regulated by sink strength and impacts the wax, cutin and rubber biosynthetic pathways. Plant Journal 79: 270–284.
Zaidi, N., J.V. Swinnen, and K. Smans. 2012. ATP-citrate lyase: A key player in cancer metabolism. Cancer Research 72: 3709–3714.
Zhang, H., X. Mao, R. Jing, X. Chang, and H. Xie. 2011. Characterization of a common wheat (Triticum aestivum L.) TaSnRK2.7 gene involved in abiotic stress responses. Journal of Experimental Botany 62(3): 975–988.
Zhang, H., X. Mao, C. Wang, and R. Jing. 2010. Overexpression of a common wheat gene TaSnRK2.8 enhances tolerance to drought, salt and low temperature in Arabidopsis. PLoS One 5(12): e16041.
Acknowledgments
This work was supported by the grants from the National High Technology Research and Development Program (“863” Program) of China (2013AA102604), Natural Science Foundation of China (31360293), International Scientific Cooperation Program of China (2013DFA31600), Guangxi Special Funds for Bagui Scholars and Distinguished Experts (2013) and Guangxi Natural Science Foundation (2011GXNSFF018002, 2012GXNSFDA053011).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material. ξ
Rights and permissions
About this article
Cite this article
Phan, TT., Li, J., Sun, B. et al. ATP-Citrate Lyase Gene (SoACLA-1), a Novel ACLA Gene in Sugarcane, and Its Overexpression Enhance Drought Tolerance of Transgenic Tobacco. Sugar Tech 19, 258–269 (2017). https://doi.org/10.1007/s12355-016-0464-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12355-016-0464-8