Abstract
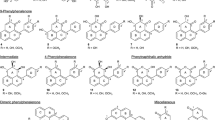
Sugarcane secrets a reddish pigment presumed to contain phytoalexin compounds in the stalk tissues in response to infection of Colletotrichum falcatum, causing red rot. To identify the phytoalexin compounds involved in red rot resistance, detailed HPLC analyses were conducted using the phytoalexin standards in a set of sugarcane cultivars varying in red rot resistance, after pathogen inoculation. We found nine different 3-deoxyanthocyanidin phytoalexin compounds, which are differentially induced upon C. falcatum infection in sugarcane varieties with varying degrees of disease resistance. Among the nine compounds, four compounds were identified as luteolinidin, 5-methoxy luteolinidin, apigeninidin and arabinosyl-5-O-apigeninidin. Multifold induction of luteolinidin, apigeninidin and arabinosyl-5-O-apigeninidinin were recorded in the stalk tissues in disease resistant cvs Co 93009, BO 91 and Baragua, respectively after pathogen inoculation as compared to the susceptible cv CoC 671. In addition to the known compounds one to three unknown compounds were detected in the resistant cultivars. The susceptible cultivars exhibited a placid induction of phytoalexin compounds after pathogen infections. The results very clearly demonstrated a specific induction of 3-deoxyanthocyanidins in resistant cultivars at a higher level as compared to susceptible cultivars after C. falcatum infection in sugarcane. Total extracts of the induced phytoalexins in the calorimetric assays, also revealed similar results. Probably induction of either luteolinidin or apigeninidin compounds alone or in combination at higher concentrations in the resistant cultivars may enable the effective arrest of pathogen invasion and further development inside the stalk tissues.





Similar content being viewed by others
References
Aida, Y., S. Tamogami, O. Kodama, and T. Tsukiboshi. 1996. Synthesis of apigeninidin 7-methylether and its fungicidal activity against. Gloeocercospora sorghi. Bioscience Biotechnology and Biochemistry 60: 1495–1496.
Boddu, J., C. Svabek, R. Sekhon, A. Gevens, R.L. Nicholson, D.A. Jones, J.F. Pedersen, D.L. Gustine, and S. Chopra. 2004. Expression of a putative flavonoid 3′-hydroxylase in sorghum mesocotyls synthesizing 3-deoxyanthocyanidin phytoalexins. Physiological and Molecular Plant Pathology 65: 101–113.
Hammerschmidt, R. 1999. Phytoalexins: What have we learned after 60 years? Annual Review Phytopathology 37: 285–306.
Knogge, W., E. Kombrink, E. Schmelzer, and K. Hahlbrock. 1987. Occurrence of phytoalexins and other putative defense-related substances in uninfected parsley plants. Planta 171: 279–287.
Kodama, O., J. Miyakawa, T. Akatsuka, and S. Kiyosawa. 1992. Sakuranetin, a flavanone phytoalexin from ultraviolet-irradiated rice leaves. Phytochemistry 31: 3807–3813.
Liu, H., Y. Du, H. Chu, C.H. Shih, Y.W. Wong, M. Wang, I.K. Chu, Y. Tao, and C. Lo. 2010. Molecular dissection of the pathogen-inducible 3-deoxyanthocyanidin biosynthesis pathway in sorghum. Plant Cell Physiology 51: 1173–1185.
Lo, S.-C., I. Weiergang, C. Bonham, J. Hipskind, K. Wood, and R.L. Nicholson. 1996. Phytoalexin accumulation in sorghum: Identification of a methyl ether of luteolinidin. Physiological and Molecular Plant Pathology 49: 21–31.
Lo, S.-C.C., K. de Verdier, and R.L. Nicholson. 1999. Accumulation of 3-deoxyanthocyanidin phytoalexins and resistance to Colletotrichum sublineolum in sorghum. Physiological and Molecular Plant Pathology 55: 263–273.
Malathi, P., R. Viswanathan, P. Padmanaban, D. Mohanraj, V. Ganesh Kumar, and K.P. Salin. 2008. Differential accumulation of 3-deoxy anthocyanidin phytoalexins in sugarcane varieties varying in red rot resistance in response to Colletotrichum falcatum infection. Sugar Tech 10: 154–157.
Mansfield, J.W. 1999. Antimicrobial compounds and resistance: the role of phytoalexins and anti anticipins. In Mechanisms of resistance to plant diseases, ed. A.J. Slusarenko, R.S.S. Fraser, and L.C. VanLoon, 325–370. Amsterdam: Kluwer.
Nicholson, R.L., S.S. Kollipara, J.R. Vincent, P.C. Lyons, and G. Cadena-Gomez. 1987. Phytoalexin synthesis by the sorghum mesocotyl in response to infection by pathogenic and nonpathogenic fungi. Proceedings of the National Academy of Sciences USA 84: 5520–5524.
Snyder, B.A., and R.L. Nicholson. 1990. Synthesis of phytoalexins in sorghum as a site specific response to fungal ingress. Science 248: 1637–1639.
Snyder, B.A., B. Leite, J. Hipskind, L.G. Butler, and R.L. Nicholson. 1991. Accumulation of sorghum phytoalexins induced by Colletotrichum graminicola at the infection site. Physiological and Molecular Plant Pathology 39: 463–470.
Tenkouano, A., F.R. Miller, G.E. Hart, R.A. Frederiksen, and R.L. Nicholson. 1993. Phytoalexin assay in juvenile sorghum: An aid to breeding for anthracnose resistance. Crop Science 33: 243–248.
Tsuji, J., E.P. Jackson, D.A. Gage, R. Hammerschmidt, and S.C. Somerville. 1992. Phytoalexin accumulation in Arabidopsis thaliana during the hypersensitive reaction to Pseudomonas syringae pv. syringae. Plant Physiology 98: 1304–1309.
Van Etten, H.D., P.S. Matthews, K.J. Tegtmeier, M.F. Dietert, and J.I. Stein. 1980. The association of pisatin tolerance and demethylation with virulence on pea in Nectria haematococca. Physiological Plant Pathology 16: 257–268.
Van Etten, H.D., J.W. Mansfield, J. Bailey, and E.E. Farmer. 1994. Letter to the editor: Two classes of plant antibiotics: Phytoalexins versus phytoanticipins. Plant Cell 6: 1191–1192.
Viswanathan, R. 2010. Plant disease: red rot of sugarcane. New Delhi: Anmol Publications Pvt. Ltd.
Viswanathan, R., D. Mohanraj, and P. Padmanaban. 1996a. Role of red rot pigments in relation to red rot resistance in sugarcane. Indian Journal of Sugarcane Technology 11: 151–154.
Viswanathan, R., D. Mohanraj, P. Padmanaban, and K.C. Alexander. 1996b. Synthesis of phytoalexins in sugarcane in response to infection by Colletotrichum falcatum Went. Acta Phytopathologica Entomologica Hungarica 31: 229–237.
Wharton, P.S., and R.L. Nicholson. 2000. Temporal synthesis and radiolabelling of the sorghum 3-deoxyanthocyanidin phytoalexins and the anthocyanin, cyanidin3-dimalonyl glucoside. New Phytologist 145: 457–469.
Acknowledgments
Authors are thankful to Dr. N. Vijayan Nair, Director of the Institute for the support and encouragement. The research work was supported by Sugar Development Fund, Ministry of Consumer Affairs, Food and Public Distribution, New Delhi.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ganesh Kumar, V., Viswanathan, R., Malathi, P. et al. Differential Induction of 3-deoxyanthocyanidin Phytoalexins in Relation to Colletotrichum falcatum Resistance in Sugarcane. Sugar Tech 17, 314–321 (2015). https://doi.org/10.1007/s12355-014-0334-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12355-014-0334-1