Abstract
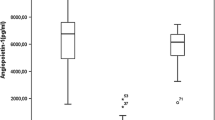
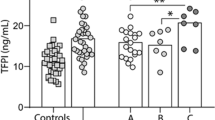
In actuality, chronic lymphocytic leukaemia (CLL) remains an incurable haematopoietic malignancy of high prevalence amongst elderly populations in the West. Malignant CLL cells characteristically accumulate in the peripheral blood, bone marrow, lymph nodes, and spleen of CLL patients. There is evidence that CLL cells express Ang2 and Tie1, two central components of the Ang-Tie2 pro-angiogenic pathway. Central to blood vessel development and maintenance, at present it remains unclear how the Ang-Tie2 pathway modulates CLL pathophysiology. Here we evaluate the status of the Ang-Tie2 pathway in CLL cells and assess Ang1 levels in plasma/cell medium from CLL samples. To understand how angiopoietins in the microenvironment regulate the components of Ang-Tie2 pathway, survival, migration, and metabolic fitness of CLL cells, we exposed CLL cells to recombinant angiopoietins. CLL plasma and CLL cells in culture present significant lower levels of Ang1. CLL cells simultaneously express Ang1, Ang2, and Tie1 mRNA, but lack that of Tie2 and its regulator, VE-PTP. Exposure to Ang1 confers survival advantage in the long-term, whereas Ang2 and trebananib, an angiopoietin blocker, proved detrimental. Angiopoietins differentially modulate expression of Ang1, Ang2, and Tie1 transcripts. Ang2, but not Ang1, induces the concomitant and transient expression of Tie2 and VE-PTP mRNA. Both angiopoietins, particularly Ang2, increase CLL-Tie1 expression and Ang1 clearly induces chemotaxis and transendothelial-like migration of CLL cells. Besides, changes in caspase and ATP content corroborate the sensitivity of CLL cells to angiopoietin exposure. Altogether, this work shows that angiopoietins regulate the fate of CLL cells in a Tie2-independent manner and highlights the potential of the Ang-Tie2 pathway as a therapeutic target in CLL research.










Similar content being viewed by others
Abbreviations
- Ang1/Ang2:
-
Angiopoetin-1 and angiopoietin -2
- bFGF:
-
Basic fibroblast growth factor
- CLL:
-
Chronic lymphocytic leukaemia
- PBMC:
-
Peripheral blood mononuclear cells
- Tie1/Tie2:
-
Tyrosine kinase with immunoglobulin and endothelial growth factor domain-1 and -2
- VEGF:
-
Vascular endothelia growth factor
- VE-PTP:
-
Vascular endothelial-protein tyrosine phosphatase
References
Folkman J (2001) Angiogenesis. In: Brenner S, Miller JH (eds) Encyclopedia of genetics. Academic, New York, pp 66–73
Kay NE, Bone ND, Tschumper RC et al (2002) B-CLL cells are capable of synthesis and secretion of both pro- and anti-angiogenic molecules. Leukemia 16(5):911–919
Hallek M (2013) Chronic lymphocytic leukemia: 2013 update on diagnosis, risk stratification and treatment. Am J Hematol 88(9):803–816
Mauro FR, Foa R, Giannarelli D et al (1999) Clinical characteristics and outcome of young chronic lymphocytic leukemia patients: a single institution study of 204 cases. Blood 94(2):448–454
Peterson L, Kini AR, Kay N (2001) Angiogenesis is increased in B-cell chronic lymphocytic leukemia. Blood 97(8):2529–2530
Maffei R, Martinelli S, Santachiara R et al (2010) Angiopoietin-2 plasma dosage predicts time to first treatment and overall survival in chronic lymphocytic leukemia. Blood 116(4):584–592
Maffei R, Martinelli S, Castelli I et al (2010) Increased angiogenesis induced by chronic lymphocytic leukemia B cells is mediated by leukemia-derived Ang2 and VEGF. Leuk Res 34(3):312–321
Martinelli S, Kanduri M, Maffei R et al (2013) ANGPT2 promoter methylation is strongly associated with gene expression and prognosis in chronic lymphocytic leukemia. Epigenetics 8(7)
Aguayo A, Kantarjian H, Manshouri T et al (2000) Angiogenesis in acute and chronic leukemias and myelodysplastic syndromes. Blood 96(6):2240–2245
Augustin HG, Young Koh G, Thurston G, Alitalo K (2009) Control of vascular morphogenesis and homeostasis through the angiopoietin–Tie system. Nat Rev Mol Cell Biol 10(3):165–177
Seegar TCM, Eller B, Tzvetkova-Robev D et al (2010) Tie1-Tie2 interactions mediate functional differences between angiopoietin ligands. Mol Cell 37(5):643–655
Papapetropoulos A, García-Cardeña G, Dengler TJ, Maisonpierre PC, Yancopoulos GD, Sessa WC (1999) Direct actions of Angiopoietin-1 on human endothelium: evidence for network stabilization, cell survival, and interaction with other angiogenic growth factors. Lab Invest J Tech Methods Pathol 79(2):213–223
Jeansson M, Gawlik A, Anderson G et al (2011) Angiopoietin-1 is essential in mouse vasculature during development and in response to injury. J Clin Invest 121(6):2278–2289
Huang H, Bhat A, Woodnutt G, Lappe R (2010) Targeting the ANGPT–TIE2 pathway in malignancy. Nat Rev Cancer 10(8):575–585
Yancopoulos GD, Davis S, Gale NW, Rudge JS, Wiegand SJ, Holash J (2000) Vascular-specific growth factors and blood vessel formation. Nature 407(6801):242–248
Kawaguchi M, Sugaya M, Suga H et al (2013) Serum levels of Angiopoietin-2, but not Angiopoietin-1, are Elevated in Patients with Erythrodermic Cutaneous T-cell Lymphoma. Acta Derm Venereol
Moss A (2013) The angiopoietin:Tie 2 interaction: a potential target for future therapies in human vascular disease. Cytokine Growth Factor Rev
Maisonpierre PC, Suri C, Jones PF et al (1997) Angiopoietin-2, a natural antagonist for Tie2 that disrupts in vivo angiogenesis. Science 277(5322):55–60
Barton WA, Tzvetkova-Robev D, Miranda EP et al (2006) Crystal structures of the Tie2 receptor ectodomain and the angiopoietin-2–Tie2 complex. Nat Struct Mol Biol 13(6):524–532
Reiss Y, Droste J, Heil M et al (2007) Angiopoietin-2 impairs revascularization after limb ischemia. Circ Res 101(1):88–96
Barton WA, Tzvetkova D, Nikolov DB (2005) Structure of the Angiopoietin-2 receptor binding domain and identification of surfaces involved in Tie2 recognition. Structure 13(5):825–832
Yu X, Seegar TCM, Dalton AC et al (2013) Structural basis for Angiopoietin-1–mediated signaling initiation. Proc Natl Acad Sci 110(18):7205–7210
Winderlich M, Keller L, Cagna G et al (2009) VE-PTP controls blood vessel development by balancing Tie-2 activity. J Cell Biol 185(4):657–671
Fachinger G, Deutsch U, Risau W (1999) Functional interaction of vascular endothelial-protein-tyrosine phosphatase with the angiopoietin receptor Tie-2. Oncogene 18(43):5948–5953
Nawroth R, Poell G, Ranft A et al (2002) VE-PTP and VE-cadherin ectodomains interact to facilitate regulation of phosphorylation and cell contacts. EMBO J 21(18):4885–4895
Shen J, Frye M, Lee BL et al (2014) Targeting VE-PTP activates TIE2 and stabilizes the ocular vasculature. J Clin Invest 124(10):4564–4576
Nottebaum AF, Cagna G, Winderlich M et al (2008) VE-PTP maintains the endothelial barrier via plakoglobin and becomes dissociated from VE-cadherin by leukocytes and by VEGF. J Exp Med 205(12):2929–2945
Martinelli S, Maffei R, Castelli I et al (2008) Increased expression of Angiopoietin-2 characterizes early B-cell chronic lymphocytic leukemia with poor prognosis. Leuk Res 32(4):593–597
Aguayo A, Manshouri T, O’Brien S et al (2001) Clinical relevance of Flt1 and Tie1 angiogenesis receptors expression in B-cell chronic lymphocytic leukemia (CLL). Leuk Res 25(4):279–285
Kay NE, Hamblin TJ, Jelinek DF et al (2002) Chronic lymphocytic leukemia. ASH Educ Program Book 2002(1):193–213
Parikh SA, Kay NE, Shanafelt TD (2013) Monoclonal B-cell lymphocytosis: update on diagnosis, clinical outcome, and counseling. Clin Adv Hematol Oncol 11(11):720–729
Venneri MA, Palma MD, Ponzoni M et al (2007) Identification of proangiogenic TIE2-expressing monocytes (TEMs) in human peripheral blood and cancer. Blood 109(12):5276–5285
Goon PKY, Lip GYH, Boos CJ, Stonelake PS, Blann AD (2006) Circulating endothelial cells, endothelial progenitor cells, and endothelial microparticles in cancer. Neoplasia 8(2):79–88
Horner A, Bord S, Kelsall AW, Coleman N, Compston JE (2001) Tie2 ligands Angiopoietin-1 and Angiopoietin-2 are coexpressed with vascular endothelial cell growth factor in growing human bone. Bone 28(1):65–71
Carlson TR, Feng Y, Maisonpierre PC, Mrksich M, Morla AO (2001) Direct cell adhesion to the angiopoietins mediated by integrins. J Biol Chem 276(28):26516–26525
Maffei R, Bulgarelli J, Fiorcari S et al (2013) The monocytic population in chronic lymphocytic leukemia shows altered composition and deregulation of genes involved in phagocytosis and inflammation. Haematologica 98(7):1115–1123
Stapor PC, Sweat RS, Dashti DC, Betancourt AM, Murfee WL (2014) Pericyte dynamics during angiogenesis: new insights from new identities. J Vasc Res 51(3):163–174
Cao Y, Sonveaux P, Liu S et al (2007) Systemic overexpression of Angiopoietin-2 promotes tumor microvessel regression and inhibits angiogenesis and tumor growth. Cancer Res 67(8):3835–3844
ten Hacken E, Burger JA (2014) Molecular pathways: targeting the microenvironment in chronic lymphocytic leukemia—focus on the B-cell receptor. Clin Cancer Res 20(3):548–556
Morrison SJ, Scadden DT (2014) The bone marrow niche for haematopoietic stem cells. Nature 505(7483):327–334
Eikesdal HP, Kalluri R (2009) Drug resistance associated with antiangiogenesis therapy. Semin Cancer Biol 19(5):310–317
Woo KV, Baldwin HS (2011) Role of Tie1 in shear stress and atherosclerosis. Trends Cardiovasc Med 21(4):118–123
Fukuhara S, Sako K, Minami T et al (2008) Differential function of Tie2 at cell–cell contacts and cell–substratum contacts regulated by Angiopoietin-1. Nat Cell Biol 10(5):513–526
Prezma T, Shteinfer A, Admoni L et al (2013) VDAC1-based peptides: novel pro-apoptotic agents and potential therapeutics for B-cell chronic lymphocytic leukemia. Cell Death Dis 4(9), e809
Kasahara A, Scorrano L (2014) Mitochondria: from cell death executioners to regulators of cell differentiation. Trends Cell Biol
Tao Z, Chen B, Tan X et al (2011) Coexpression of VEGF and Angiopoietin-1 promotes angiogenesis and cardiomyocyte proliferation reduces apoptosis in porcine myocardial infarction (MI) heart. Proc Natl Acad Sci 108(5):2064–2069
Dallabrida SM, Ismail N, Oberle JR, Himes BE, Rupnick MA (2005) Angiopoietin-1 promotes cardiac and skeletal myocyte survival through integrins. Circ Res 96(4):e8–e24
Coxon A, Bready J, Min H et al (2010) Context-dependent role of Angiopoietin-1 inhibition in the suppression of angiogenesis and tumor growth: implications for AMG 386, an Angiopoietin-1/2-neutralizing peptibody. Mol Cancer Ther 9(10):2641–2651
Herbst RS, Hong D, Chap L et al (2009) Safety, pharmacokinetics, and antitumor activity of AMG 386, a selective angiopoietin inhibitor, in adult patients with advanced solid tumors. J Clin Oncol 27(21):3557–3565
Acknowledgments
We would like to thank Keri Elizabeth Parsons Mattaliano, MLIS, for kindly revising the manuscript. We would like to thank Thomas Landwehr and Lukas Heukamp from the biobank, Center for Integrated Oncology, for their kind support in providing CLL specimens.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interests
The authors declare that they have no conflicts of interest. In addition, the authors declare that they have no disclosures to make.
Funding
This work was made possible by a grant (No. 2011.101.02) from to the Wilhelm Sander-Foundation, Munich, Germany.
Rights and permissions
About this article
Cite this article
Aguirre Palma, L., Flamme, H., Gerke, I. et al. Angiopoietins Modulate Survival, Migration, and the Components of the Ang-Tie2 Pathway of Chronic Lymphocytic Leukaemia (CLL) Cells In Vitro. Cancer Microenvironment 9, 13–26 (2016). https://doi.org/10.1007/s12307-016-0180-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12307-016-0180-7