Abstract
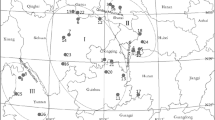
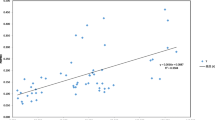
Cordyceps sinensis is one of the most valuable medicinal caterpillar fungi native to China. However, its productivity is extremely limited and the species is becoming endangered. The genetic diversity of eighteen C. sinensis populations across its major distributing regions in China was evaluated by inter-simple sequence repeat (ISSR) markers. A total of 141 markers were produced in 180 individuals from the 18 populations, of which 99.3% were polymorphic. The low average of Shannon (0.104) and Nei index (0.07) of the 18 populations indicates that there are little genetic variations within populations. For all 18 populations, estimates of total gene diversity (HT), gene diversity within populations (HS), coefficient of genetic differentiation (GST), and gene flow (Nm) were 0.170, 0.071, 0.583, and 0.357, respectively. This pattern suggests that the genetic diversity of C. sinensis is low and most of the ISSR variations are found among populations with little gene exchange. The 18 populations are divided into five groups based on the genetic distance and the grouping pattern matches with the geographic distribution along the latitudinal gradient. The five groups show obvious difference in the GST and Nm values. Therefore, the genetic diversification of C. sinensis populations may be determined by geographic isolation and the combined effects of life history characters and the interaction with host insect species. The information illustrated by this study is useful for selecting in situ conservation sites of C. sinensis.
Similar content being viewed by others
References
Belard, Y., N. Chtourou-Ghorbel, and M. Marrakchi. 2006. Genetic diversity within and between populations of Lathyrus genus (Fabaceae) revealed by ISSR markers. Genet. Resour. Crop Evol. 53, 1413–1418.
Cai, Z.J., D.H. Yin, T.F. Huang, S.J. Chen, and Q.S. Li. 2003. Comparison of the mannitol content in Cordyceps from different growing areas. China Pharmacy 14, 505–506.
Cai, Z.J., D.G. Yin, L. Li, and W.J. Xia. 2001. Investigation on quality difference between Cordyceps of Sichuan and those of Xizang. China J. Chinese Material Med. 26, 450–452.
Casu, M., D. Casu, T. Lai, P. Cossu, and M. Curini-Calletti. 2006. Inter-simple sequence repeat markers reveal strong genetic differentiation among populations of the endangered mollusk Patella ferruginea (Gastropoda: Patellidae) from two Sardinian marine protected areas. Marine Biology 149, 1163–1174.
Chen, Y.Q., B. Hu, F. Xu, W.M. Zhang, H. Zhou, and L.H. Qu. 2004. Genetic variation of Cordyceps sinensis, a fruit-body-producing entomopathogenic species from different geographical regions in China. FEMS Microbiol. Lett. 230, 153–158.
Chen, Y.Q., N. Wang, L.H. Qu, T.H. Li, and W.M. Zhang. 2001. Determination of the anamorph of Cordyceps sinensis inferred from the analysis of the ribosomal DNA internal transcribed spacers and 5.8S rDNA. Biochem. Syst. Ecol. 29, 597–607.
Chen, Y.J., W. Wang, and Y.X. Yang. 1997. Genetic divergence of Cordyceps sinensis as estimated by random amplified polymorphic DNA analysis. Acta Genet. Sin. 24, 410–416.
Chen, Y.J., Y.P. Zhang, Y.X. Yang, and D.R. Yang. 1999. Genetic diversity and taxonomic implication of Cordyceps sinensis as revealed by RAPD markers. Biochem. Genet. 37, 201–213.
Cheng, Z., Y. Geng, H.H. Liang, X.L. Yang, S. Li, Y.G. Zhu, G.P. Guo, T.S. Zhou, and J.K. Chen. 2007. Phylogenetic relationships of host insects of Cordyceps sinensis inferred from mitochondrial cytochrome b sequences. Prog. Natur. Sci. 17, 789–797.
Fritsch, P. and L.H. Rieseberg. 1996. The use of random amplified polymorphic DNA (RAPD) in conservation genetics, p. 54–73. In T.B. Smith and R.K. Wayne (eds.), Molecular Genetic Approaches in Conservation. Oxford University Press, London, UK.
Hedrick, P.W., M.E. Ginevan, and E.P. Ewing. 1976. Genetic polymorphism in heterogeneous environments. Ann. Rev. Ecol. Syst. 7, 1–32.
Jiang, Y. and Y.J. Yao. 2003. Anamorphic fungi related to Cordyceps sinensis. Mycosystema 22, 161–176.
Kar, P.K., K. Vijayan, T.P. Mohandas, C.V. Nair, B. Saratchandra, and K. Thangavelu. 2005. Genetic variability and genetic structure of wild and semi-domestic populations of tasar silk worm (Antheraea mylitta) ecorace Daba as revealed through ISSR markers. Genetica 125, 173–183.
Kinjo, N. and M. Zang. 2001. Morphological and phylogenetic studies on Cordyceps sinensis distributed in southwestern China. Mycoscience 42, 567–574.
Li, Z.Z., B. Huang, C.R. Li, and M.Z. Fan. 2000. The evidences of molecular biology of the asexual type of Cordyceps sinensis (Berk.) Sacc. (I) Relationship of Hirsutella sinensis and Cordyceps sinensis (Berk.) Sacc. ACTA Mycol. Sin. 19, 60–64.
Li, S.P. and K.W.K. Tsim. 2002. The biological and pharmacological properties of Cordyceps sinensis, p. 657–683. In L. Packer and C.N. Ong (eds.), Herbal and Traditional Medicine-Molecular Aspects of Health. Marcel Dekker, New York, N.Y., USA.
Liang, H.H., Z. Cheng, X.L. Yang, S. Li, T.S. Zhou, W.J. Zhang, and J.K. Chen. 2005. Genetic variation and affinity of Cordyceps sinensis in Qinghai province based on analysis of morphologic characters and inter-simple sequence repeat markers. Chinese Tradit. Herb. Drugs 36, 1859–1864.
Liu, F., X.L. Wu, and D.H. Yin. 2005. Overview in biological studies of host insects of Cordyceps sinensis. Chongqing J. Research on Chinese Drugs and Herbs (In Chinese) 51, 45–52.
Nei, M. 1973. Analysis of gene diversity in subdivided populations. Proc. Nat. Acad. Sci. 70, 3321–3323.
Nei, M. 1978. Estimation of average heterozygosity and genetic distance from a small number of individuals. Genetics 89, 583–590.
Nevo, E., A. Beiles, and D. Kaplan. 1988. Genetic diversity and environmental associations of wild emmer wheat in Turkey. Heredity 61, 31–45.
Rohlf, F.J. 2000. NTSYS-pc. Numerical Taxonomy and Multivariate Analysis System, Version 2.1. Exeter Software, Setauket, New York, N.Y., USA.
Shimitsu, D. 1978. Cordyceps. Green Book 51, New Science Co., Japan.
Slatkin, M. 1987. Gene flow and the geographic structure of natural populations. Science 236, 787–792.
Spagnuolo, V., L. Mscariello, S. Cozzolino, R.C. Cobianchi, and S. Giordano. 2007. Genetic diversity in ISSR markers between and within populations of the asexually producing moss Pleurochaete squarrosa. Plant Ecol. 188, 91–101.
The State Pharmacopoeia Commission of P. R. China. 2005. Pharmacopoeia of The People’s Republic of China (Volume I), p. 75–76. Chemistry Industry Publishing House, Beijing, China.
Yang, D.R., C.D. Li, C. Shu, and Y.X. Yang. 1996. Studies on the Chinese species of the genus Hepialus and their geographical distribution. Acta Entomol. Sin. 39, 413–422.
Yeh, F.C., R.C. Yang, T.B.J. Boyle, Z.H. Ye, and J.X. Mao. 1999. POPGENE Ver. 1.32, the User-friendly Shareware for Population Genetic Analysis. Molecular biology and biotechnology centre, University of Alberta, Canada.
Zhang, Y.W., Y.J. Chen, F.R. Shen, Y.X. Yang, D.R. Yang, and Y.P. Zhang. 1999. Study of genetic divergence in Cordyceps sinensis and C. crassispora from northwest of Yunnan by using RAPD. Mycosystema 18, 176–183.
Zhu, H.Y. and J.L. Mou. 2006. Removal of chongcao at the source of the three rivers: A craze out of control, p. 291–298. In C.J. Liang (ed.), Crisis and Breakthrough of China’s Environment 2005. Social Sciences Academic Press, Beijing, China.
Zietkiewicz, E., A. Rafalski, and D. Labuda. 1994. Genome fingerprinting by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 20, 176–183.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liang, HH., Cheng, Z., Yang, XL. et al. Genetic diversity and structure of Cordyceps sinensis populations from extensive geographical regions in China as revealed by inter-simple sequence repeat markers. J Microbiol. 46, 549–556 (2008). https://doi.org/10.1007/s12275-008-0107-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-008-0107-1