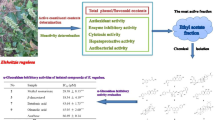
Abstract
Diabetes mellitus (DM) and Alzheimer’s disease (AD) constitute two global health issues. DM is an ever-increasing epidemic affecting millions of elderly people worldwide, causing major repercussions on patients’ daily lives, mostly due to chronic complications. Complications from DM can affect the brain, thereby characterizing DM as a risk factor for AD. In the present study, we examined the inhibitory activity of methanol extracts of different parts of 12 Angelica species against α-glucosidase, protein tyrosine phosphatase 1B (PTP1B), acetylcholinesterase (AChE), and butyrylcholinesterase (BChE). The methanol extract of Angelica decursiva exhibited the highest inhibitory activities against α-glucosidase, PTP1B, AChE, and BChE and so was selected for further investigation. Repeated column chromatography based on bioactivity-guided fractionation yielded seven compounds (1–7). Among these compounds, nodakenin (1), nodakenetin (2), umbelliferone (3), cis-3′-acetyl-4′-angeloylkhellactone (4), 3′(R)-O-acetyl-4′(S)-O-tigloylkhellactone (5), isorutarine (6), and para-hydroxybenzoic acid (7) exhibited potent inhibitory activities against α-glucosidase, PTP1B, rat lens aldose reductase (RLAR), AChE, BChE, and β-site amyloid precursor protein cleaving enzyme 1 (BACE1). Our results clearly indicate the potential inhibition of α-glucosidase, PTP1B, RLAR, AChE, BChE, and BACE1 by A. decursiva as well as its isolated constituents, which could be further explored to develop therapeutic modalities for the treatment of DM and AD.

Similar content being viewed by others
References
Association, Alzheimer’s. 2014. Alzheimer’s disease facts and figures. Alzheimers Dementia 10: 47–92.
Butterfield, D.A., T. Reed, S.F. Newman, and R. Sultana. 2007. Roles of amyloid β peptide associated oxidative stress and brain protein modifications in the pathogenesis of Alzheimer’s disease and mild cognitive impairment. Free Radical Biology & Medicine 43: 658–677.
Cai, H., Y. Wang, D. McCarthy, H. Wen, D.R. Borchelt, D.L. Price, and P.C. Wong. 2001. BACE1 is the major beta-secretase for generation of Aβ peptides by neurons. Nature Neuroscience 4: 233–234.
Campbell, R.K. 2011. Clarifying the role of incretin-based therapies in the treatment of type 2 diabetes mellitus. Clinical Therapeutics 33: 511–527.
Chen, Y.C., P.Y. Chen, C.C. Wu, I.L. Tsai, and I.S. Chen. 2008. Chemical constituents and anti-platelet aggregation activity from the root of peucedanum formosanum. Journal of Food and Drug Analysis 16: 15–25.
Ciro, A., J. Park, G. Burkhard, N. Yan, and C. Geula. 2012. Biochemical differentiation of cholinesterases from normal and Alzheimer’s disease cortex. Current Alzheimer Research 9: 138–143.
Citron, M. 2004. Strategies for disease modification in Alzheimer’s disease. Nature Reviews Neuroscience 5: 677–685.
Clapham, A.R., T.G. Tutin, and E.F. Warburg. 1952. Flora of the British Isles. London: The University Press Cambridge.
Cui, L., M. Na, H. Oh, E.Y. Bae, D.G. Jeong, S.E. Ryu, B.Y. Kim, W.K. Oh, and J.S. Ahn. 2006. Protein tyrosine phosphatase 1B inhibitors from Morus root bark. Bioorganic & Medicinal Chemistry Letters 16: 1426–1429.
Darvesh, S., D. Hopkins, and C. Geula. 2003. Neurobiology of butyrylcholinesterase. Nature Reviews Neuroscience 4: 131–138.
de la Monte, S.M. 2009. Insulin resistance and Alzheimer’s disease. BMB Reports 42: 475–481.
Ellman, G.L., K.D. Courtney, V.J. Andres, and R.M. Featherstone. 1961. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochemical Pharmacology 7: 88–95.
Elosta, A., T. Ghous, and N. Ahmed. 2012. Natural products as antiglycation agents: Possible therapeutic potential for diabetic complications. Current Diabetes Review 8: 92–108.
Eurich, D.T., F.A. McAlister, and D.F. Blackburn. 2007. Benefits and harms of antidiabetic agents in patients with diabetes and heart failure: Systematic review. BMJ 7618: 497–501.
Fernandez, J., L. Rojo, R.O. Kuljis, and R.B. Maccioni. 2008. The danger signals hypothesis of Alzheimer s disease. Journal of Alzheimer’s Disease 14: 329–333.
Francis, P.T., A.M. Palmer, and M. Snape. 1999. The cholinergic hypothesis of Alzheimer’s disease: A review of progress. Journal of Neurology, Neurosurgery and Psychiatry 54: 137–147.
Gonzalez, R.G., P. Barnett, J. Aguayo, H.M. Cheng, and L.T.J. Chylack. 1984. Direct measurement of polyol pathway activity in the ocular lens. Diabetes 33: 196–199.
Hata, K., and K. Sano. 1969. Studies on coumarins from the root of Angelica decursiva Fr. et Sav I, The structure of decursin and decursidin. Yakugaku Zasshi 89: 549–557.
Hayman, S., and J.H. Kinoshita. 1965. Isolation and properties of lens aldose reductase. Journal of Biological Chemistry 240: 877–882.
Imam, K. 2012. Clinical features diagnostic criteria and pathogenesis of diabetes mellitus. Advances in Experimental Medicine and Biology 771: 340–355.
Johnson, T.O., J. Ermolieff, and M.R. Jirousek. 2002. Protein tyrosine phosphatase 1B inhibitors for diabetes. Nature Reviews Drug Discovery 1: 696–709.
Jung, H.A., Y.J. Jung, S.K. Hyun, B.S. Min, D.W. Kim, J.H. Jung, and J.S. Choi. 2010a. Selective cholinesterase inhibitory activities of a new monoterpene diglycoside and other constituents from Nelumbo nucifera stamens. Biological & Pharmaceutical Bulletin 33: 267–272.
Jung, H.A., S.H. Oh, and J.S. Choi. 2010b. Molecular docking studies of phlorotannins from Eisenia bicyclis with BACE1 inhibitory activity. Bioorganic & Medicinal Chemistry Letters 20: 3211–3215.
Kang, S.Y., K.Y. Lee, S.H. Sung, M.J. Park, and Y.C. Kim. 2001. Coumarins isolated from Angelica gigas inhibit acetylcholinesterase: Structure–activity relationships. Journal of Natural Products 64: 683–685.
Kahn, C.R., and R. Suzuki. 2010. Insulin action in the brain and the pathogenesis of Alzheimer’s disease. Research and Perspectives in Alzheimer’s Disease 27: 1–20.
Karimi, G., M. Iranshahi, F. Hosseinalizadeh, B. Riahi, and A. Sahebkar. 2010. Screening of acetylcholinesterase inhibitory activity of terpenoid and coumarin derivatives from the genus Ferula. Pharmacologyonline 1: 566–574.
Kim, D.H., D.Y. Kim, Y.C. Kim, J.W. Jung, S.J. Lee, B.H. Yoon, J.H. Cheong, Y.S. Kim, S.S. Kang, K.H. Ko, and J.H. Ryu. 2007. Nodakenin, a coumarin compound, ameliorates scopolamine-induced memory disruption in mice. Life Sciences 80: 1944–1950.
Kinoshita, J.H., P. Kador, and M. Catiles. 1981. Aldose reductase in diabetic cataract. JAMA Journal 246: 257–261.
Kwon, S.H., H.K. Lee, J.A. Kim, S.I. Hong, H.C. Kim, and T.H. Jo. 2010. Neuroprotective effects of chlorogenic acid on scopolamine-induced amnesia via antiacetylcholinesterase and anti-oxidative activities in mice. European Journal of Pharmacology 649: 210–217.
Laird, F.M., H. Cai, A.V. Savonenko, M.H. Farah, K. He, T. Melnikova, H. Wen, H.C. Chiang, G. Xu, V.E. Koliatsos, D.R. Borchelt, D.L. Price, H.K. Lee, and P.C. Wong. 2005. BACE1, a major determinant of selective vulnerability of the brain to Aβ amyloidogenesis, is essential for cognitive, emotional, and synaptic functions. Journal of Neuroscience 25: 11693–11709.
Lee, S.W., C.S. Kim, S.H. Cho, H.S. Chun, J.K. Kim, and D.K. Kim. 2009. The effects of Angelica decursiva extract in the inhibition of cell proliferation and in the induction of apoptosis in osteogenic sarcoma cells. Journal of Medicinal Plants Research 3: 241–245.
Li, T., X.D. Zhang, Y.W. Song, and J.W. Liu. 2005. A microplate-based screening method for α-glucosidase inhibitors. Chinese Journal of Clinical Pharmacology and Therapeutics 10: 1128–1134.
Liu, R., Q. Sun, Y. Shi, and L. Kong. 2005. Isolation and purification of coumarin compounds from the root of peucedanum decursivum (Miq.) maxim by high-speed counter-current chromatography. Journal of Chromatography A 1076: 127–132.
Lou, H.X., L.R. Sun, W.T. Yu, P.H. Fan, L. Cui, Y.H. Gao, M. Ma, D.M. Ren, and M. Ji. 2004. Absolute configuration determination of angular dihydrocoumarins from peucedanum praeruptorum. Journal of Asian Natural Products Research 6: 177–184.
Morrison, A.D., R.S. Clements, S.B. Travis, F. Oski, and A.I. Winegrad. 1970. Glucose utilization by the polyol pathway in human erythrocytes. Biochemical and Biophysical Research Communications 40: 199–205.
Moreira, P.I., M.S. Santos, R. Seica, and C.R. Oliveira. 2007. Brain mitochondrial dysfunction as a link between Alzheimer’s disease and diabetes. Journal of the Neurological Sciences 257: 206–214.
Neumann, K.F., L. Rojo, L.P. Navarrete, G. Farias, P. Reyes, and R.B. Maccioni. 2008. Insulin resistance and Alzheimer’s disease: Molecular links and clinical implications. Current Alzheimer Research 5: 438–447.
Ohno, M., E.A. Sametsky, L.H. Younkin, H. Oakley, S.G. Younkin, M. Citron, R. Vassar, and J.F. Disterhoft. 2004. BACE1 deficiency rescues memory deficits and cholinergic dysfunction in a mouse model of Alzheimer’s disease. Neuron 41: 27–33.
Okuyama, T., M. Takata, and S. Shibata. 1989. Structure of linear furano and simple-coumarin glycosides of Bai-Hua Qian-Hu. Planta Medica 55: 64–67.
Ozturk, M. 2012. Anticholinesterase and antioxidant activities of Savory (Satureja thymbra L.) with identified major terpenes of the essential oil. Food Chemistry 134: 48–54.
Panunti, B., A.A. Jawa, and V.A. Fonseca. 2004. Mechanisms and therapeutic targets in type 2 diabetes mellitus. Drug Discovery Today: Disease Mechanisms 1: 151–157.
Pari, L., and N. Rajarajeswari. 2009. Efficacy of coumarin on hepatic key enzymes of glucose metabolism in chemical induced type 2 diabetic rats. Chemico-Biological Interactions 181: 292–296.
Piazzi, L., A. Cavalli, F. Colizzi, F. Belluti, M. Bartolini, F. Mancini, M. Recanatini, V. Andrisano, and A. Rampa. 2008. Multi-target-directed coumarin derivatives: hAChE and BACE1 inhibitors as potential anti-Alzheimer compounds. Bioorganic & Medicinal Chemistry Letters 18: 423–426.
Ramesh, B., P. Viswanathan, and K.V. Pugalendi. 2007. Protective effect of umbelliferone on membranous fatty acid composition in streptozotocin-induced diabetic rats. European Journal of Pharmacology 566: 231–239.
Sharma, R., and V. Arya. 2011. A review on fruits having anti-diabetic potential. Journal of Chemical and Pharmaceutical Research 3: 204–212.
Song, Y.L., Q.W. Zhang, Y.P. Li, R. Yan, and Y.T. Wang. 2012. Enantioseparation and absolute configuration determination of angular-type pyranocoumarins from peucedani radix using enzymatic hydrolysis and chiral HPLC-MS/MS analysis. Molecules 17: 4236–4251.
Vassar, R., D.M. Kovacs, R. Yan, and P.C. Wong. 2009. The γ-secretase enzyme BACE in health and Alzheimer’s disease: Regulation, cell biology, function, and therapeutic potential. Journal of Neuroscience 29: 12787–12794.
Vukovic, N., S. Sukdolak, S. Solujic, and N. Niciforovic. 2010. An efficient synthesis and antioxidant properties of novel imino and amino derivatives of 4-hydroxy coumarins. Archives of Pharmaceutical Research 33: 5–15.
Walsh, D.M., and D.J. Selkoe. 2007. Aβ oligomers a decade of discovery. Journal of Neurochemistry 101: 1172–1184.
Zhang, Z. 2002. Protein tyrosine phosphatases: Structure and function, substrate specificity, and inhibitor development. Annual Review of Pharmacology and Toxicology 42: 209–234.
Zhao, D., M.N. Islam, B.R. Ahn, H.A. Jung, B.W. Kim, and J.S. Choi. 2012. In vitro antioxidant and anti-inflammatory activities of Angelica decursiva. Archives of Pharmaceutical Research 2012(35): 179–192.
Acknowledgments
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Science, ICT & Future Planning (2014R1A1A3051684 and 2012R1A6A1028677).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Rights and permissions
About this article
Cite this article
Yousof Ali, M., Jung, H.A. & Choi, J.S. Anti-diabetic and anti-Alzheimer’s disease activities of Angelica decursiva . Arch. Pharm. Res. 38, 2216–2227 (2015). https://doi.org/10.1007/s12272-015-0629-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-015-0629-0