Abstract
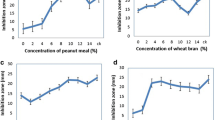
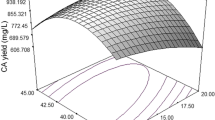
Response surface methodology was used to optimize the medium for antifungal active substance production from Streptomyces lydicus E12 in flask cultivation. Initially, the component factors, which influence antifungal substance production, were studied by varying one factor at a time. Starch, soybean cake powder, K2HPO4·3H2O and MgSO4·7H2O were found to have a significant effect on the production of antifungal substances by the traditional design. Then, a Box–Behnken design was applied for further optimization. A quadratic model was found to fit antifungal active substance production. The analysis revealed that the optimum values of the tested variable were starch 84.96 g/L, soybean cake powder 4.13 g/L, glucose 5 g/L, MgSO4·7H2O 1.23 g/L, K2HPO4·3H2O 2.14 g/L and NaCl 0.5 g/L. The test result of 67.44% antifungal inhibition agreed with the prediction and increased by 14.28% in comparison with the basal medium.






Similar content being viewed by others
References
Maestro B, Sanz JM (2007) Novel approaches to fight Streptococcus pneumoniae. Recent Patents Antiinfect Drug Discov 2(3):188–196
Kitouni M, Boudemagh A, Oulmi L et al (2005) Isolation of actinobacteria producing bioactive substances from water, soil and tree bark samples of the north-east of Algeria. J Med Mycol 15(1):45–51
Leiva PS, Yáñez SM, Zaror CL et al (2004) Actividad antimicrobiana de actinobacteria aislados desde ambientes acuáticos del sur de Chile. Revista Médica de Chile 132(2):151–159
Thomson RH (1997) Naturally occurring quinones IV. Blackie Academic and Professional, Chapman and Hall, London
Malviya MK, Pandey A, Trivedi P et al (2009) Chitinolytic activity of cold tolerant antagonistic species of Streptomyces isolated from glacial sites of Indian himalaya. Curr Microbiol 59(5):502–508
János B (2005) Bioactive microbial metabolites. J Antibiot 58(1):1–26
Singh SB, Genilloud O, Pelaez F (2010) NP structural diversity II—secondary metabolite sources, evolution and selected molecular structures: terrestrial micro-organisms—bacteria. Elsevier, UK, pp 109–140
Braña AF, Fiedler HP, Nava H et al (2015) Two Streptomyces species producing antibiotic, antitumor, and anti-inflammatory compounds are widespread among intertidal macroalgae and deep-sea coral reef invertebrates from the central cantabrian sea. Microb Ecol 69(3):512–524
Komatsua M, Uchiyama T, Omura S et al (2010) Genome-minimized Streptomyces host for the heterologous expression of secondary metabolism. Proc Natl Acad Sci 107(6):2646–2651
Balachandran C, Arun Y, Duraipandiyan V et al (2014) Antimicrobial and cytotoxicity properties of 2,3-dihydroxy-9,10-anthraquinone isolated from Streptomyces galbus (ERINLG-127). Appl Biochem Biotechnol 172(7):3513–3528
Balachandran C, Duraipandiyan V, Emi N et al (2015) Antimicrobial and cytotoxic properties of Streptomyces sp. (ERINLG-51) isolated from Southern Western Ghats. South Indian J Biol Sci 1(5):7–14
Balachandran C, Duraipandiyan V, Arasu MV et al (2014) Antimicrobial, antioxidant and cytotoxic properties of Streptomyces sp. (ERINLG-01) isolated from Southern Western Ghats. Int J Pharm Pharm Sci 6(2):189–196
Cheng JS, Lv XM, Yuan YJ (2012) Investigation of proteomic responses of Streptomyces lydicus to pitching ratios for improving streptolydigin production. Biotechnol Bioprocess Eng 17(5):997–1007
Cheng JS, Cui SF, Ding MZ et al (2013) Insights into the roles of exogenous glutamate and prolinein improving streptolydigin production of Streptomyces lydicus with metabolomic analysis. J Ind Microbiol Biotechnol 40(11):1303–1314
Cheng JS, Liang YQ, Ding MZ et al (2013) Metabolic analysis reveals the amino acid responses of Streptomyces lydicus to pitching ratios during improving streptolydigin production. Appl Microbiol Biotechnol 97(13):5943–5954
Lee H, Song M, Hwang S (2003) Optimizing bioconversion of deproteinated cheese whey to mycelia of Ganoderma lucidum. Process Biochem 38:1685–1693
Bernal C, Diaz I, Coello N (2006) Response surface methodology for the optimization of keratinase production in culture medium containing feathers produced by Kocuria rosea. Can J Microbiol 52:445–450
Zhang J, Gao N (2007) Application of response surface methodology in medium optimization for pyruvic acid production of Torulopsis glabrata TP19 in batch fermentation. J Zhejiang Univ Sci 8:98–104
Tellez SJ, Moldes AB, Alonso JL et al (2003) Optimization of lactic acid production by Lactobacillus delbrueckii through response surface methodology. J Food Sci 68:1454–1458
Li Y, Jiang H, Xu Y et al (2008) Optimization of nutrient components for enhanced phenazine-1-carboxylic acid production by gacA-inactivated Pseudomonas sp. M18G using response surface method. Appl Microbiol Biotechnol 77(6):1207–1224
Velayudam S, Murugan K (2015) Sequential optimization approach for enhanced production of antimicrobial compound from Streptomyces rochei BKM-4. South Indian J Biol Sci 1(2):72–79
Gao H, Liu M, Liu JT et al (2009) Medium optimization for the production of avermectin B1a by Streptomyces avermitilis 14-12A using response surface methodology. Bioresour Technol 100(17):4012–4016
Shih IL, Shen MH (2006) Application of response surface methodology to optimize production of poly-ε-lysine by Streptomyces albulus IFO 14147. Enzyme Microb Technol 39(1):15–21
Souagui Y, Tritsch D, Grosdemange-Billiard C et al (2015) Optimization of antifungal production by analkaliphilic and halotolerant actinomycete, Streptomyces sp. SY-BS5, using response surface methodology. Journal de Mycologie Médicale 25(2):108–115
Wang ZW, Liu XL (2008) Medium optimization for antifungal active substances production from a newly isolated Paenibacillus sp. using response surface methodology. Bioresour Technol 99(17):8245–8251
Chen XC, Bai JX, Cao JM et al (2009) Medium optimization for the production of cyclic adenosine 3′,5′-monophosphate by Microbacterium sp. no. 205 using response surface methodology. Bioresour Technol 100(2):919–924
Gams W, Hoekstra ES, Aptroot A (1998) CBS course of mycology, 4th edn. Centraalbureau voor Schimmelcultures, Baarn
Gamliel A, Katan J, Cohen E (1989) Toxicity of chloronitrobenzenes to Fusarium oxysporum and Rhizoctoniasolani as related to their structure. Phytoparasitica 17(2):101–106
Wiwattanapatapee R, Chumthong A, Pengnoo A et al (2007) Effervescent fast-disintegrating bacterial formulation for biological control of rice sheath blight. J Control Release 119(2):229–235
Guo WH (2007) A preliminary study for optimization and separation of agricultural antifungal antibiotic fermentation process. Tianjin University, Tianjin (in Chinese)
Wei YP (2009) Study on optimization of fermentation technology for antifungal antibiotics. Tianjin University, Tianjin (in Chinese)
Zhu CH, Lu FP, He YN et al (2007) Regulation of avilamycin biosynthesis in Streptomyces viridochromogenes: effects of glucose, ammonium ion, and inorganic phosphate. Appl Microbiol Biotechnol 73(5):1031–1038
Zhang HC, Fan WM, Zhang YX et al (2002) Effect of glucose and phosphate on growth metabolism and blue-pigment production in Streptomyces coelicolor fermentation. Chem React Eng Technol 18(3):244–248
Castro JM, Liras P, Cortes J et al (1985) Regulation of α-aminoadipyl-cysteinyl-valine, isopenicillin N synthetase, isopenicillin N isomerase and deacetoxycephalosporin C synthetase by nitrogen sources in Streptomyces lactamdurans. Appl Microbiol Biotechnol 22(1):32–40
Shen YQ, Heim J, Solomon NA et al (1984) Repression of β-lactam production in Cephalosporium acremonium by nitrogen sources. J Antibiot 37(5):503–511
Zhang TT, Shen HM (2011) Optimization of culture medium for laccase production from Pycnoporus sanguineus (Fr.) Murr by Plackett–Burman design and response surface methodology. Sci Technol Food Ind 32(9):223–226
Tang LR (2010) Preparation, properties and application of nanocellulose crystal. Fujian Agriculture and Forestry University, Fuzhou (in Chinese)
Acknowledgements
Supported by the National Basic Research Program of China (“973” Program: 2014CB745100) and the National Natural Science Foundation of China (No. 21576201 and No. 21176183).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhao, Y., Liang, Y., Liu, L. et al. Medium Optimization for Antifungal Active Substance Production from Streptomyces Lydicus Using Response Surface Methodology. Trans. Tianjin Univ. 23, 78–86 (2017). https://doi.org/10.1007/s12209-016-0023-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12209-016-0023-0