Abstract
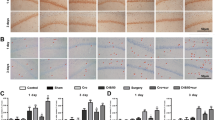
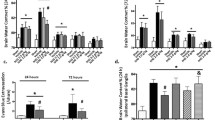
Brain inflammation has a critical role in the pathophysiology of brain diseases. Microglia, the resident immune cells in the brain, play an important role in brain inflammation, while brain mast cells are the “first responder” in the injury rather than microglia. Functional aspects of mast cell-microglia interactions remain poorly understood. Our results demonstrated that site-directed injection of the “mast cell degranulator” compound 48/80 (C48/80) in the hypothalamus induced mast cell degranulation, microglial activation, and inflammatory factor production, which initiated the acute brain inflammatory response. “Mast cell stabilizer” disodium cromoglycate (cromolyn) inhibited this effect, including decrease of inflammatory cytokines, reduced microglial activation, inhibition of MAPK and AKT pathways, and repression of protein expression of histamine receptor 1 (H1R), histamine receptor 4 (H4R), protease-activated receptor 2 (PAR2), and toll-like receptor 4 (TLR4) in microglia. We also demonstrated that C48/80 had no effect on microglial activation in mast cell-deficient KitW-sh/W-sh mice. These results implicate that activated brain mast cells trigger microglial activation and stabilization of mast cell inhibits microglial activation-induced central nervous system (CNS) inflammation. Interactions between mast cells and microglia could constitute a new and unique therapeutic target for CNS immune inflammation-related diseases.






Similar content being viewed by others
References
Nilupul Perera M, Ma HK, Arakawa S, Howells DW, Markus R, Rowe CC, Donnan GA (2006) Inflammation following stroke. J Clin Neurosci 13:1–8
De Chiara G, Marcocci ME, Sgarbanti R, Civitelli L, Ripoli C, Piacentini R, Garaci E, Grassi C et al (2012) Infectious agents and neurodegeneration. Mol Neurobiol 46:614–638
Block ML, Hong JS (2005) Microglial and inflammation-mediated neurodegeneration: multiple triggers with a common mechanism. Prog Neurobiol 76:77–98
Herrera AJ, Tomas-Camardiel M, Venero JL, Cano J, Machado A (2005) Inflammatory process as a determinant factor for the degeneration of substantia nigra dopaminergic neurons. J Neural Transm 112:111–119
Gao HM, Liu B, Zhang W, Hong JS (2003) Novel anti-inflammatory therapy for Parkinson’s disease. Trends Pharmacol Sci 24:395–401
Skaper SD, Facci L, Giusti P (2014) Mast cells, glia and neuroinflammation: partners in crime? Immunology 141:314–327
Dropp JJ (1976) Mast cells in mammalian brain. Acta Anat 94:1–21
Theoharides TC, Stewart JM, Panagiotidou S, Melamed I. (2015). Mast cells, brain inflammation and autism. Eur J Pharmacol. doi: 10.1016/j.ejphar.2015.03.086
Silverman AJ, Sutherland AK, Wilhelm M, Silver R (2000) Mast cells migrate from blood to brain. J Neurosci 20:401–408
Theoharides TC, Angelidou A, Alysandratos KD, Zhang B, Asadi S, Francis K, Toniato E, Kalogeromitros D (2012) Mast cell activation and autism. Biochim Biophys Acta 1822:34–41
Khalil MH, Silverman AJ, Silver R (2003) Mast cells in the rat brain synthesize gonadotropin-releasing hormone. Neurobiol 56:113–124
Silver R, Silverman AJ, Vitkovic L, Lederhendler I (1996) Mast cells in the brain: evidence and functional significance. Trends Neurosci 19:25–31
Ikarashi Y, Yuzurihara M (2002) Experimental anxiety induced by histaminergics in mast cell-deficient and congenitally normal mice. Pharmacol Biochem Behav 72:437–441
Theoharides TC, Cochrane DE (2004) Critical role of mast cells in inflammatory diseases and the effect of acute stress. J Neuroimmunol 146:1–12
Jutel M, Watanabe T, Klunker S, Akdis M, Thomet OA, Malolepszy J, Zak-Nejmark T, Koga R et al (2001) Histamine regulates T-cell and antibody responses by differential expression of H1 and H2 receptors. Nature 413:420–425
Skaper SD, Facci L (2012) Mast cell–glia axis in neuroinflammation and therapeutic potential of the anandamide congener palmitoylethanolamide. Philos Trans R Soc LondB Biol Sci 367:3312–3325
Yuan H, Zhu X, Zhou S, Chen Q, Zhu X, Ma X, He X, Tian M et al (2010) Role of mast cell activation in inducing microglial cells to release neurotrophin. J Neurosci Res 88:1348–1354
Zhang S, Zeng X, Yang H, Hu G, He S (2012) Mast cell tryptase induces microglia activation via protease-activated receptor 2 signaling. Cell Physiol Biochem 29:931–940
Dong H, Zhang W, Zeng X, Hu G, Zhang H, He S, Zhang S (2014) Histamine induces upregulated expression of histamine receptors and increases release of inflammatorymediators from microglia. Mol Neurobiol 49:1487–1500
Paxinos G, Watson C (1986) The rat brain in stereotaxic coordinates. Academic Press, Inc, Orlando, Florida, p 32887
Paxinos G, Franklin K (2001) The mouse brain in stereotatic coordinates. Acadmic Press. pp. Figs, London, pp 1–100
Akundi RS, Candelario-Jalil E, Hess S, Hull M, Lieb K, Gebicke-Haerter PJ, Fiebich BL (2005) Signal transduction pathways regulating cyclooxygenase-2 in lipopolysaccharide-activated primary rat microglia. Glia 51:199–208
Ciallella JR, Saporito M, Lund S, Leist M, Hasseldam H, McGann N, Smith CS, Bozyczko-Coyne D et al (2005) CEP-11004, an inhibitor of the SAPK/JNK pathway, reduces TNF-alpha release from lipopolysaccharide-treated cells and mice. Eur J Pharmacol 515:179–187
Desai P, Thurmond RL (2011) Histamine H4 receptor activation enhances LPS-induced IL-6 production in mast cells via ERK and PI3K activation. Eur J Immunol 41:1764–1773
Skaper SD, Giusti P, Facci L (2012) Microglia and mast cells: two tracks on the road to neuroinflammation. Faseb J 26:3103–3017
Biran V, Cochois V, Karroubi A, Arrang JM, Charriaut-Marlangue C, Heron A (2008) Stroke induces histamine accumulation and mast cell degranulation in the neonatal rat brain. Brain Pathol 18:1–9
Lozada A, Maegele M, Stark H, Neugebauer EM, Panula P (2005) Traumatic brain injury results in mast cell increase and changes in regulation of central histamine receptors. Neuropathol Appl Neurobiol 31:150–162
Jin Y, Silverman AJ, Vannucci SJ (2009) Mast cells are early responders after hypoxia-ischemia in immature rat brain. Stroke 40:3107–3112
Gregersen R, Lambertsen K, Finsen B (2000) Microglia and macrophages are the major source of tumor necrosis factor in permanent middle cerebral artery occlusion in mice. J Cereb Blood Flow Metab 20:53–65
Lambertsen KL, Meldgaard M, Ladeby R, Finsen B (2005) A quantitative study of microglial-macrophage synthesis of tumor necrosis factor during acute and late focal cerebral ischemia in mice. J Cereb Blood Flow Metab 25:119–135
Hallenbeck JM (2002) The many faces of tumor necrosis factor in stroke. Nat Med 8:1363–1368
David S, Kroner A (2011) Repertoire of microglial and macrophage responses after spinal cord injury. Nat Rev Neurosci 12:388–399
Minghetti L (2005) Role of inflammation in neurodegenerative diseases. Curr Opin Neurol 18:315–321
Maurer M, Theoharides T, Granstein RD, Bischoff SC, Bienenstock J, Henz B, Kovanen P, Piliponsky AM et al (2003) What is the physiological function of mast cells? Exp Dermatol 12:886–910
Galli SJ, Nakae S, Tsai M (2005) Mast cells in the development of adaptive immune responses. Nat Immunol 6:135–142
Galli SJ, Kalesnikoff J, Grimbaldeston MA, Piliponsky AM, Williams CM, Tsai M (2005) Mast cells as “Tunable” effector and immunoregulatory cells: recent advances. Annu Rev Immunol 23:749–786
Feuser K, Thon KP, Bischoff SC, Lorentz A (2012) Human intestinal mast cells are a potent source of multiple chemokines. Cytokine 58:178–185
Bulanova E, Bulfone-Paus S (2010) P2 receptor-mediated signaling in mast cell biology. Purinergic Signal 6:3–17
Khalil MH, Silverman AJ, Silver R (2004) Mast cell mediators alter electrical activity of rat thalamic neurons. In: Keystone symposium on mast cells in physiology, host defense and disease. Beyond IgE, Taos, New Mexico, p 57
Esposito P, Gheorghe D, Kandere K, Pang X, Connolly R, Jacobson S, Theoharides TC (2001) Acute stress increases permeability of the blood–brain barrier through activation of brain mast cells. Brain Res 888:117–127
Skuljec J, Sun H, Pul R, Bénardais K, Ragancokova D, Moharregh-Khiabani D, Kotsiari A, Trebst C et al (2011) CCL5 induces a pro-inflammatory profile in microglia in vitro. Cell Immunol 270:164–171
Chakraborty S, Kaushik DK, Gupta M, Basu A (2010) Inflammasome signaling at the heart of central nervous system pathology. J Neurosci Res 88:1615–1631
Acknowledgments
This project was sponsored by the National Natural Science Foundation of China (nos. 81102422, 81373398, and 81471410), a Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that there is no conflict of interests regarding the publication of this paper.
Additional information
Hongquan Dong and Xiang Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Dong, H., Zhang, X., Wang, Y. et al. Suppression of Brain Mast Cells Degranulation Inhibits Microglial Activation and Central Nervous System Inflammation. Mol Neurobiol 54, 997–1007 (2017). https://doi.org/10.1007/s12035-016-9720-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-016-9720-x