Abstract
Background
This study investigated if cerebral blood flow (CBF) regulation by changes of the arterial partial pressure of carbon dioxide (PaCO2) can be used therapeutically to increase CBF and improve neurological outcome after subarachnoid hemorrhage (SAH).
Methods
In 12 mechanically ventilated poor-grade SAH-patients, a daily trial intervention was performed between day 4 and 14. During this intervention, PaCO2 was decreased to 30 mmHg and then gradually increased to 40, 50, and 60 mmHg in 15-min intervals by modifications of the respiratory minute volume. CBF and brain tissue oxygen saturation (StiO2) were the primary and secondary endpoints. Intracranial pressure was controlled by an external ventricular drainage.
Results
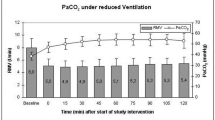
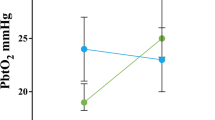
CBF reproducibly decreased during hyperventilation and increased to a maximum of 141 ± 53 % of baseline during hypercapnia (PaCO2 60 mmHg) on all days between day 4 and 14 after SAH. Similarly, StiO2 increased during hypercapnia. CBF remained elevated within the first hour after resetting ventilation to baseline parameters and no rebound effect was observed within this time-span. PaCO2-reactivities of CBF and StiO2 were highest between 30 and 50 mmHg and slightly decreased at higher levels.
Conclusion
CBF and StiO2 reproducibly increased by controlled hypercapnia of up to 60 mmHg even during the period of the maximum expected vasospasm. The absence of a rebound effect within the first hour after hypercapnia indicates that an improvement of the protocol is possible. The intervention may yield a therapeutic potential to prevent ischemic deficits after aneurysmal SAH.





Similar content being viewed by others
References
Diringer MN, Kirsch JR, Hanley DF, Traystman RJ. Altered cerebrovascular CO2 reactivity following subarachnoid hemorrhage in cats. J Neurosurg. 1993;78(6):915–21.
Schmieder K, Jarus-Dziedzic K, Wronski J, Harders A. CO2 reactivity in patients after subarachnoid haemorrhage. Acta Neurochir (Wien). 1997;139(11):1038–41.
Hassler W, Chioffi F. CO2 reactivity of cerebral vasospasm after aneurysmal subarachnoid haemorrhage. Acta Neurochir (Wien). 1989;98(3–4):167–75.
Carrera E, Schmidt JM, Fernandez L, et al. Spontaneous hyperventilation and brain tissue hypoxia in patients with severe brain injury. J Neurol Neurosurg Psychiatry. 2010;81(7):793–7.
Yamamoto M, Meyer J, Naritomi H, Sakai F, Yamaguchi F, Shaw T. Noninvasive measurement of cerebral vasospasm in patients with subarachnoid hemorrhage. J Neurol Sci. 1979;43(2):301–15.
Curley G, Kavanagh BP, Laffey JG. Hypocapnia and the injured brain: more harm than benefit. Crit Care Med. 2010;38(5):1348–59.
Carrera E, Kurtz P, Badjatia N, et al. Cerebrovascular carbon dioxide reactivity and delayed cerebral ischemia after subarachnoid hemorrhage. Arch Neurol. 2010;67(4):434–9.
Westermaier T, Stetter C, Kunze E, et al. Controlled transient hypercapnia: a novel approach for the treatment of delayed cerebral ischemia after subarachnoid hemorrhage? J Neurosurg. 2014;121(5):1056–62.
von Kummer R, Holle R, Gizyska U, et al. Interobserver agreement in assessing early CT signs of middle cerebral artery infarction. AJNR Am J Neuroradiol. 1996;17(9):1743–8.
Cross DT, Tirschwell DL, Clark MA, et al. Mortality rates after subarachnoid hemorrhage: variations according to hospital case volume in 18 states. J Neurosurg. 2003;99(5):810–7.
Rivero-Arias O, Gray A, Wolstenholme J. Burden of disease and costs of aneurysmal subarachnoid haemorrhage (aSAH) in the United Kingdom. Cost Eff Resour Alloc. 2010;8(6):6.
Frontera JA, Fernandez A, Schmidt JM, et al. Defining vasospasm after subarachnoid hemorrhage: what is the most clinically relevant definition? Stroke. 2009;40(6):1963–8.
Petridis AK, Doukas A, Kienke S, et al. The effect of lung-protective permissive hypercapnia in intracerebral pressure in patients with subarachnoid haemorrhage and ARDS: a retrospective study. Acta Neurochir (Wien). 2010;152(12):2143–5. doi:10.1007/s00701-010-0761-z.
Claassen J, Carhuapoma JR, Kreiter KT, Du EY, Connolly ES, Mayer SA. Global cerebral edema after subarachnoid hemorrhage: frequency, predictors, and impact on outcome. Stroke. 2002;33(5):1225–32.
Stengl M, Ledvinova L, Chvojka J, et al. Effects of clinically relevant acute hypercapnic and metabolic acidosis on the cardiovascular system: an experimental porcine study. Crit Care. 2013;17(6):R303.
Chuang IC, Dong HP, Yang RC, et al. Effect of carbon dioxide on pulmonary vascular tone at various pulmonary arterial pressure levels induced by endothelin-1. Lung. 2010;188(3):199–207.
Karlsson T, Stjernstrom EL, Stjernstrom H, Norlen K, Wiklund L. Central and regional blood flow during hyperventilation: an experimental study in the pig. Acta Anaesthesiol Scand. 1994;38(2):180–6.
Macdonald RL. Delayed neurological deterioration after subarachnoid haemorrhage. Nat Rev Neurol. 2014;10(1):44–58.
Diringer MN, Kirsch JR, Traystman RJ. Reduced cerebral blood flow but intact reactivity to hypercarbia and hypoxia following subarachnoid hemorrhage in rabbits. J Cereb Blood Flow Metab. 1994;14(1):59–63.
Ma XD, Hauerberg J, Pedersen DB, Juhler M. Effects of morphine on cerebral blood flow autoregulation CO2-reactivity in experimental subarachnoid hemorrhage. J Neurosurg Anesthesiol. 1999;11(4):264–72.
Dernbach PD, Little JR, Jones SC, Ebrahim ZY. Altered cerebral autoregulation and CO2 reactivity after aneurysmal subarachnoid hemorrhage. Neurosurgery. 1988;22(5):822–6.
Poulin MJ, Liang PJ, Robbins PA. Fast and slow components of cerebral blood flow response to step decreases in end-tidal PCO2 in humans. J Appl Physiol. 1998;85(2):388–97.
Komori M, Takada K, Tomizawa Y, Nishiyama K, Kawamata M, Ozaki M. Permissive range of hypercapnia for improved peripheral microcirculation and cardiac output in rabbits. Crit Care Med. 2007;35(9):2171–5.
Hickling KG, Joyce C. Permissive hypercapnia in ARDS and its effect on tissue oxygenation. Acta Anaesthesiol Scand Suppl. 1995;107:201–8.
Rabinstein AA, Friedman JA, Weigand SD, et al. Predictors of cerebral infarction in aneurysmal subarachnoid hemorrhage. Stroke. 2004;35(8):1862–6.
Westermaier T, Pham M, Stetter C, et al. Value of transcranial Doppler, perfusion-CT and neurological evaluation to forecast secondary ischemia after aneurysmal SAH. Neurocrit Care. 2014;20(3):406–12.
Agnoli A. Adaptation of CBF during induced chronic normooxic respiratory acidosis. Scand J Clin Lab Investig Suppl. 1968;102:VIII.
Fencl V, Vale JR, Broch JA. Respiration and cerebral blood flow in metabolic acidosis and alkalosis in humans. J Appl Physiol. 1969;27(1):67–76.
Skinhoj E. CBF adaption in man to chronic hypo- and hypercapnia and its relation to CSF pH. Scand J Clin Lab Investig Suppl. 1968;102:8.
Jones TH, Morawetz RB, Crowell RM, et al. Thresholds of focal cerebral ischemia in awake monkeys. J Neurosurg. 1981;54(6):773–82.
Raichle ME, Posner JB, Plum F. Cerebral blood flow during and after hyperventilation. Arch Neurol. 1970;23(5):394–403.
Acknowledgments
The study was supported by the Else-Kröner-Fresenius Stiftung (2013_A171).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None of the authors has a conflict of interest to declare.
Rights and permissions
About this article
Cite this article
Westermaier, T., Stetter, C., Kunze, E. et al. Controlled Hypercapnia Enhances Cerebral Blood Flow and Brain Tissue Oxygenation After Aneurysmal Subarachnoid Hemorrhage: Results of a Phase 1 Study. Neurocrit Care 25, 205–214 (2016). https://doi.org/10.1007/s12028-016-0246-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-016-0246-x