Abstract
Background and Aims
Studies suggest that stroke patients with thrombus in a major cerebral vessel respond less favorably to intravenous (IV) thrombolysis. The purpose of this study was to test the feasibility of a protocol comparing IV versus intra-arterial (IA) recombinant tissue plasminogen activator (TPA) in an acute ischemic stroke with major vessel occlusion.
Methods
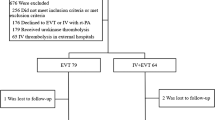
Consecutive ischemic stroke patients presenting <3 h from symptom onset with major vessel occlusion on CT angiogram (CTA) were randomly assigned to IV TPA (per NINDS protocol) or IA TPA (22 mg over 2 h). Demographics, times to presentation and thrombolysis, presenting NIH stroke scale (NIHSS) and 90-day NIHSS, Barthel Index, and modified Rankin Scale were recorded. CT-scans at 24-h were reviewed for presence of hemorrhage. Recanalization was determined by post-procedure MR angiograms, which are obtained the day after thrombolytic therapy.
Results
Seven patients (median NIHSS = 16) were randomized to IV (N = 4) or IA (N = 3) TPA. There were no significant differences in the presentation NIHSS, time to presentation, or time to treatment between the two groups. Hemorrhage was noted in one patient in the IA group (asymptomatic) and one patient in the IV group (symptomatic). Recanalization was seen in all patients treated with IA TPA and none treated with IV TPA (P = 0.03, Fisher’s Exact test).
Conclusions
We found that it is feasible to conduct a trial comparing IV vs. IA TPA in ischemic stroke patients with major vessel occlusion presenting <3 h from onset. Patients treated with IA TPA showed a trend toward higher rate of recanalization. A larger trial may be designed to test safety and effectiveness of IA TPA in this specific group of stroke patients.
Similar content being viewed by others
References
NINDS Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. N Engl J Med. 1995;333(24):1581–7. doi:10.1056/NEJM199512143332401.
Hacke W, Kaste M, Bluhmki E, Brozman M, Dávalos A, Guidetti D, et al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med. 2008;359(13):1317–29. doi:10.1056/NEJMoa0804656.
Furlan A, Higashida R, Wechsler L, Gent M, Rowley H, Kase C, et al. Intra-arterial prourokinase for acute ischemic stroke. The PROACT II study a randomized controlled trial. Prolyse in acute cerebral thromboembolism. JAMA. 1999;282(21):2003–11. doi:10.1001/jama.282.21.2003.
Adams HP Jr, del Zoppo G, Alberts MJ, Bhatt DL, Brass L, Furlan A, et al. Guidelines for the early management of adults with ischemic stroke. Stroke. 2007;38(5):1655–711. doi:10.1161/STROKEAHA.107.181486.
Lisboa RC, Jovanovic BD, Alberts MJ. Analysis of the safety and efficacy of intra-arterial thrombolytic therapy in ischemic stroke. Stroke. 2002;33(12):2866–71. doi:10.1161/01.STR.0000038987.62325.14.
Lev MH, Farkas J, Rodriguez VR, Schwamm LH, Hunter GJ, Putman CM, et al. CT angiography in the rapid triage of patients with hyperacute stroke to intraarterial thrombolysis: accuracy in the detection of large vessel thrombus. J Comput Assist Tomogr. 2001;25(4):520–8. doi:10.1097/00004728-200107000-00003.
IMS Study Investigators. Combined intravenous and intra-arterial recanalization for acute ischemic stroke: the Interventional Management of Stroke Study. Stroke. 2004;35(4):904–11. doi:10.1161/01.STR.0000121641.77121.98.
Ciccone A, Valvassori L, Gasparotti R, Scomazzoni F, Ballabio E, Sterzi R. Debunking 7 myths that hamper the realization of randomized controlled trials on intra-arterial thrombolysis for acute ischemic stroke. Stroke. 2007;38(7):2191–5. doi:10.1161/STROKEAHA.106.465567.
Bookstein JJ, Bookstein FL. Augmented experimental pulse-spray thrombolysis with tissue plasminogen activator, enabling dose reduction by one or more orders of magnitude. J Vasc Interv Radiol. 2000;11:299–303. doi:10.1016/S1051-0443(07)61421-3.
TIMI Study Group The Thrombolysis in Myocardial Infarction (TIMI) Trial. Phase I findings. N Engl J Med. 1985;312:932–6.
Neumann-Haefelin T, de Rochemont R, Fiebach JB, Gass A, Nolte C, Kucinski T, et al. Effect of incomplete (spontaneous and postthrombolytic) recanalization after middle cerebral artery occlusion: a magnetic resonance imaging study. Stroke. 2004;35:109–14. doi:10.1161/01.STR.0000106482.31425.D1.
Bastianello S, Pierallini A, Colonnese C, Brughitta G, Angeloni U, Antonelli M, et al. Fieschi, Bozzao L. Hyperdense middle cerebral artery CT sign. Neuroradiology. 1991;33:207–11. doi:10.1007/BF00588219.
Moulin T, Cattin F, Crépin-Leblond T, Tatu L, Chavot D, Piotin M, et al. Early CT signs in acute middle cerebral artery infarction: predictive value for subsequent infarct locations and outcome. Neurology. 1996;47:366–75.
Zorzon M, Mase G, Pozzi-Mucelli F, Biasutti E, Antonutti L, Iona L, et al. Increased density in the middle cerebral artery by nonenhanced computed tomography: prognostic value in acute cerebral infarction. Eur Neurol. 1993;33:256–9. doi:10.1159/000116949.
Launes J, Ketonen L. Dense middle cerebral artery sign: an indicator of poor outcome in middle cerebral artery area infarction. J Neurol Neurosurg Psychiatry. 1987;50:1550–2. doi:10.1136/jnnp.50.11.1550.
Manelfe C, Larrue V, von Kummer R, Bozzao L, Ringleb P, Bastianello S, et al. Association of hyperdense middle cerebral artery sign with clinical outcome in patients with tissue plasminogen activator. Stroke. 1999;30:769–72.
Tomsick T, Brott T, Barsan W, Broderick J, Clarke Haley E, Spilker J, et al. Prognostic value of the hyperdense middle cerebral artery sign and stroke scale score before ultraearly thrombolytic therapy. AJNR Am J Neuroradiol. 1996;17:79–85.
The NINDS rtPA Stroke Study Group. Generalized efficacy of rtPA for acute stroke: subgroup analysis of the NINDS rtPA stroke trial. Stroke. 1997;28:2119–25.
Qureshi AI. Endovascular treatment of cerebrovascular diseases and intracranial neoplasms. Lancet. 2004;363:804–13. doi:10.1016/S0140-6736(04)15697-3.
Agarwal P, Kumar S, Hariharan S, Eshkar N, Verro P, Cohen B, et al. Hyperdense middle cerebral artery sign: can it be used to select intraarterial versus intravenous thrombolysis in acute ischemic stroke? Cerebrovasc Dis. 2004;17:182–90. doi:10.1159/000075789.
Garg N, Eshkar N, Tanenbaum L, Cohen B, Sen S. Computed tomography angiographic correlates of early computed tomography signs in acute ischemic stroke. J Neuroimaging. 2004;14(3):242–5. doi:10.1177/1051228404264938.
http://clinicaltrials.gov/ct2/show/NCT00640367?term=synthesis&rank=10. Accessed December 27, 2008.
http://www.neurorad-bern.ch/combatstroke/. Accessed December 27, 2008.
Macleod MR, Davis SM, Mitchell PJ, Gerraty RP, Fitt G, Hankey GJ, et al. Results of a multicentre, randomised controlled trial of intra-arterial urokinase in the treatment of acute posterior circulation ischaemic stroke. Cerebrovasc Dis. 2005;20(1):12–7. doi:10.1159/000086121.
Broderick JP. Practical considerations in the early treatment of ischemic stroke. Am Fam Physician. 1998;57:73–9.
Mattle HP, Arnold M, Georgiadis D, Baumann C, Nedeltchev K, Benninger D, et al. Comparison of intraarterial and intravenous thrombolysis for ischemic stroke with hyperdense middle cerebral artery sign. Stroke. 2008;39(2):379–83. doi:10.1161/STROKEAHA.107.492348.
Rha JH, Saver JL. The impact of recanalization on ischemic stroke ourcome, a meta-analysis. Stroke. 2007;38:967–73. doi:10.1161/01.STR.0000258112.14918.24.
Author information
Authors and Affiliations
Corresponding author
Additional information
Investigator initiated study (ClinicalTrials.gov Identifier: NCT00624000). TPA (Alteplase) for IV and IA treatment was supplied by Genentech Inc., San Francisco, USA.
Rights and permissions
About this article
Cite this article
Sen, S., Huang, D.Y., Akhavan, O. et al. IV vs. IA TPA in Acute Ischemic Stroke with CT Angiographic Evidence of Major Vessel Occlusion: A Feasibility Study. Neurocrit Care 11, 76–81 (2009). https://doi.org/10.1007/s12028-009-9204-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-009-9204-1