Abstract
Purpose
Osteoporosis is a condition characterized by decreased bone density and bone strength, commonly observed among older individuals. Caveolin-3 (CAV3) is a principal structural protein of the caveolae membrane domains, which has been reported to participate in cell signaling as well as the maintenance of cell structure. The aim of the current study was to investigate the effects involved with the silencing of CAV3 on bone formation among osteoporotic rat models via the Wnt signaling pathway.
Methods
Osteoporosis was initially induced by means of ovariotomy among rat models in order to determine the expression of CAV3. Then, to confirm the specific function and mechanism of CAV3 from an osteoporosis perspective, the CAV3 expression vector was constructed and transfected into the osteoblasts of the osteoporotic rats. Afterward, the mRNA and protein expressions of CAV3, β-catenin, low-density lipoprotein receptor-related protein 5 (LRP5), T-cell factor (TCF), and Wnt3a in addition to cell proliferation and apoptosis were detected accordingly.
Results
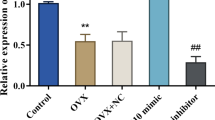
Positive expression of CAV3 exhibited diminished levels in the bone tissues of osteoporotic rats. The osteoblasts of the osteoporotic rats treated with overexpressed CAV3 displayed elevated mRNA and protein expression levels of β-catenin, LRP5, TCF, and Wnt3a. Increased cell proliferation and decreased cell apoptosis were also observed, while the osteoblasts of the osteoporotic rats treated with si-CAV3 exhibited an opposite result.
Conclusion
Overexpressed CAV3 promotes bone formation and suppresses the osteoporosis progression via the activation of the Wnt signaling in rat models, suggesting CAV3 as a potential target biomarker in the treatment of osteoporosis.










Similar content being viewed by others
Change history
18 September 2020
A Correction to this paper has been published: https://doi.org/10.1007/s12020-020-02440-z
11 March 2022
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1007/s12020-022-03032-9
References
E.M. Curtis, R.J. Moon, N.C. Harvey, C. Cooper, The impact of fragility fracture and approaches to osteoporosis risk assessment worldwide. Bone 104, 29–38 (2017)
R. Baron, M. Kneissel, WNT signaling in bone homeostasis and disease: from human mutations to treatments. Nat. Med. 19(2), 179–192 (2013)
S.W. Wade, C. Strader, L.A. Fitzpatrick, M.S. Anthony, C.D. O’Malley, Estimating prevalence of osteoporosis: examples from industrialized countries. Arch. Osteoporos. 9, 182 (2014)
N.D. Zhang, T. Han, B.K. Huang, K. Rahman, Y.P. Jiang, H.T. Xu, L.P. Qin, H.L. Xin, Q.Y. Zhang, Y.M. Li, Traditional Chinese medicine formulas for the treatment of osteoporosis: Implication for antiosteoporotic drug discovery. J. Ethnopharmacol. 189, 61–80 (2016)
K. Lippuner, The future of osteoporosis treatment - a research update. Swiss Med. Wkly 142, w13624 (2012)
R. Yuan, S. Ma, X. Zhu, J. Li, Y. Liang, T. Liu, Y. Zhu, B. Zhang, S. Tan, H. Guo, S. Guan, P. Ao, G. Zhou, Core level regulatory network of osteoblast as molecular mechanism for osteoporosis and treatment. Oncotarget 7(4), 3692–3701 (2016)
J. Cheng, C.R. Valdivia, R. Vaidyanathan, R.C. Balijepalli, M.J. Ackerman, J.C. Makielski, Caveolin-3 suppresses late sodium current by inhibiting nNOS-dependent S-nitrosylation of SCN5A. J. Mol. Cell. Cardiol. 61, 102–110 (2013)
P. Nassoy, C. Lamaze, Stressing caveolae new role in cell mechanics. Trends Cell Biol. 22(7), 381–389 (2012)
M. Maceckova, H. Martiskova, A. Koudelka, L. Kubala, A. Lojek, M. Pekarova, Bone marrow-derived macrophages exclusively expressed caveolin-2: the role of inflammatory activators and hypoxia. Immunobiology 220(11), 1266–1274 (2015)
C. Niehrs, The complex world of WNT receptor signalling. Nat. Rev. Mol. Cell Biol. 13(12), 767–779 (2012)
J.N. Anastas, R.T. Moon, WNT signalling pathways as therapeutic targets in cancer. Nat. Rev. Cancer 13(1), 11–26 (2013)
D.G. Monroe, M.E. McGee-Lawrence, M.J. Oursler, J.J. Westendorf, Update on Wnt signaling in bone cell biology and bone disease. Gene 492(1), 1–18 (2012)
G. Yuan, I. Regel, F. Lian, T. Friedrich, I. Hitkova, R.D. Hofheinz, P. Strobel, R. Langer, G. Keller, C. Rocken, W. Zimmermann, R.M. Schmid, M.P. Ebert, E. Burgermeister, WNT6 is a novel target gene of caveolin-1 promoting chemoresistance to epirubicin in human gastric cancer cells. Oncogene 32(3), 375–387 (2013)
H. Namkung-Matthai, R. Appleyard, J. Jansen, J. Hao Lin, S. Maastricht, M. Swain, R.S. Mason, G.A. Murrell, A.D. Diwan, T. Diamond, Osteoporosis influences the early period of fracture healing in a rat osteoporotic model. Bone 28(1), 80–86 (2001)
K. Jensen, R. Krusenstjerna-Hafstrom, J. Lohse, K.H. Petersen, H. Derand, A novel quantitative immunohistochemistry method for precise protein measurements directly in formalin-fixed, paraffin-embedded specimens: analytical performance measuring HER2. Mod. Pathol. 30(2), 180–193 (2017)
T.D. Rachner, S. Khosla, L.C. Hofbauer, Osteoporosis: now and the future. Lancet 377(9773), 1276–1287 (2011)
P.J. Marie, M. Kassem, Osteoblasts in osteoporosis: past, emerging, and future anabolic targets. Eur. J. Endocrinol. 165(1), 1–10 (2011)
Y. Miyauchi, Y. Sato, T. Kobayashi, S. Yoshida, T. Mori, H. Kanagawa, E. Katsuyama, A. Fujie, W. Hao, K. Miyamoto, T. Tando, H. Morioka, M. Matsumoto, P. Chambon, R.S. Johnson, S. Kato, Y. Toyama, T. Miyamoto, HIF1alpha is required for osteoclast activation by estrogen deficiency in postmenopausal osteoporosis. Proc. Natl. Acad. Sci. USA 110(41), 16568–16573 (2013)
L.M. Havill, L.G. Hale, D.E. Newman, S.M. Witte, M.C. Mahaney, Bone, ALP and OC reference standards in adult baboons (Papio hamadryas) by sex and age. J. Med. Primatol. 35(2), 97–105 (2006)
C. Li, Z. Jiang, X. Liu, Biochemical mechanism of gallium on prevention of fatal cage-layer osteoporosis. Biol. Trace Elem. Res. 134(2), 195–202 (2010)
S. Zenger, K. Hollberg, J. Ljusberg, M. Norgard, B. Ek-Rylander, R. Kiviranta, G. Andersson, Proteolytic processing and polarized secretion of tartrate-resistant acid phosphatase is altered in a subpopulation of metaphyseal osteoclasts in cathepsin K-deficient mice. Bone 41(5), 820–832 (2007)
Z. Tang, P.E. Scherer, T. Okamoto, K. Song, C. Chu, D.S. Kohtz, I. Nishimoto, H.F. Lodish, M.P. Lisanti, Molecular cloning of caveolin-3, a novel member of the caveolin gene family expressed predominantly in muscle. J. Biol. Chem. 271(4), 2255–2261 (1996)
Z. Wang, N. Wang, W. Li, P. Liu, Q. Chen, H. Situ, S. Zhong, L. Guo, Y. Lin, J. Shen, J. Chen, Caveolin-1 mediates chemoresistance in breast cancer stem cells via beta-catenin/ABCG2 signaling pathway. Carcinogenesis 35(10), 2346–2356 (2014)
J. Korvala, H. Juppner, O. Makitie, E. Sochett, D. Schnabel, S. Mora, C.F. Bartels, M.L. Warman, D. Deraska, W.G. Cole, H. Hartikka, L. Ala-Kokko, M. Mannikko, Mutations in LRP5 cause primary osteoporosis without features of OI by reducing Wnt signaling activity. Bmc. Med. Genet. 13, 26 (2012)
M. Rossini, D. Gatti, S. Adami, Involvement of WNT/beta-catenin signaling in the treatment of osteoporosis. Calcif. Tissue Int. 93(2), 121–132 (2013)
R.K. Bikkavilli, C.C. Malbon, Arginine methylation of G3BP1 in response to Wnt3a regulates beta-catenin mRNA. J. Cell. Sci. 124(Pt 13), 2310–2320 (2011)
J. Chen, F. Long, beta-catenin promotes bone formation and suppresses bone resorption in postnatal growing mice. J. Bone Miner. Res. 28(5), 1160–1169 (2013)
J. Su, A. Zhang, Z. Shi, F. Ma, P. Pu, T. Wang, J. Zhang, C. Kang, Q. Zhang, MicroRNA-200a suppresses the Wnt/beta-catenin signaling pathway by interacting with beta-catenin. Int. J. Oncol. 40(4), 1162–1170 (2012)
Z. von Marschall, L.W. Fisher, Secreted Frizzled-related protein-2 (sFRP2) augments canonical Wnt3a-induced signaling. Biochem. Biophys. Res. Commun. 400(3), 299–304 (2010)
S. Iyer, E. Ambrogini, S.M. Bartell, L. Han, P.K. Roberson, R. de Cabo, R.L. Jilka, R.S. Weinstein, C.A. O’Brien, S.C. Manolagas, M. Almeida, FOXOs attenuate bone formation by suppressing Wnt signaling. J. Clin. Invest. 123(8), 3409–3419 (2013)
J. Wang, J.S. Park, Y. Wei, M. Rajurkar, J.L. Cotton, Q. Fan, B.C. Lewis, H. Ji, J. Mao, TRIB2 acts downstream of Wnt/TCF in liver cancer cells to regulate YAP and C/EBPalpha function. Mol. Cell 51(2), 211–225 (2013)
S. Bousserouel, M. Raymondjean, A. Brouillet, G. Bereziat, M. Andreani, Modulation of cyclin D1 and early growth response factor-1 gene expression in interleukin-1beta-treated rat smooth muscle cells by n-6 and n-3 polyunsaturated fatty acids. Eur. J. Biochem. 271(22), 4462–4473 (2004)
F. Fang, W.Y. Zhao, R.K. Li, X.M. Yang, J. Li, J.P. Ao, S.H. Jiang, F.Z. Kong, L. Tu, C. Zhuang, W.X. Qin, P. He, W.M. Zhang, H. Cao, Z.G. Zhang, Silencing of WISP3 suppresses gastric cancer cell proliferation and metastasis and inhibits Wnt/beta-catenin signaling. Int. J. Clin. Exp. Pathol. 7(10), 6447–6461 (2014)
N. Pecina-Slaus, Wnt signal transduction pathway and apoptosis: a review. Cancer Cell. Int. 10, 22 (2010)
H. Yamamoto, H. Komekado, A. Kikuchi, Caveolin is necessary for Wnt-3a-dependent internalization of LRP6 and accumulation of beta-catenin. Dev. Cell. 11(2), 213–223 (2006)
Acknowledgements
We would like to acknowledge the helpful comments on this paper received from our reviewers.
Funding
This study was supported by Fuzhou Science and Technology Bureau (No. 2013-S-123-4) and Natural Science Foundation of Fujian Province (No. 2016J01598).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All animal operations in this study were performed was in line with the local principles of management and use of experimental animals.
Additional information
This article has been retracted. Please see the retraction notice for more detail:https://doi.org/10.1007/s12020-022-03032-9
About this article
Cite this article
Yang, RB., Lin, FF., Yang, J. et al. RETRACTED ARTICLE: Overexpression of CAV3 facilitates bone formation via the Wnt signaling pathway in osteoporotic rats. Endocrine 63, 639–650 (2019). https://doi.org/10.1007/s12020-018-1803-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-018-1803-1