Abstract
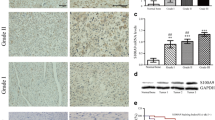
The aim of the study was to explore the expression of three genes, HSPD1, SCUBE3, and CXCL14, in osteosarcoma cells and tissue, as well as their association with the prognosis of patients with osteosarcoma. The expression of HSPD1, SCUBE3, and CXCL14 in osteosarcoma cells was detected by using Western blotting method. siRNA was used to knockdown the expression of the three genes. CCK8 cell proliferation assay was used to observe the effect of siRNA interference on U2OS cell proliferation. The expression of the three genes in osteosarcoma tissue was detected employing immunohistochemical method. Kaplan–Meier survival analysis was used to compare the relations between the expression of the three genes and prognosis. The Western blotting results showed that the expression of Hsp70, SCUBE3 protein, and CXCL14 chemotactic factor in osteosarcoma cells was significantly higher than that in normal osteocytes (p < 0.05). After the three genes were interfered by siRNA, the mRNA and protein expression levels of these genes in osteosarcoma cells were significantly decreased (p < 0.05). The growth rate of U2OS cell after the siRNA interference was significantly lower than that before interference and that in the control group transfected with negative control siRNA (p < 0.05). The result of immunohistochemistry demonstrated that the expression of Hsp70, SCUBE3 protein, and CXCL14 chemotactic factor in osteosarcoma tissues was significantly higher than that in adjacent muscle tissue (p < 0.05). Kaplan–Meier survival analysis indicated that the survival rate of the patients with high expression of those three kinds of genes was obviously lower than that of other patients (p < 0.05). There was no significant difference between the survival rates of patients with high or low expression of two genes (p > 0.05). The expression of HSPD1, SCUBE3, and CXCL14 was all high in osteosarcoma tissues and cells; moreover, the three kinds of genes had close correlations with the prognosis of the patients. Targeted inhibition of these three genes could inhibit the proliferation of the tumor, which may become a new therapeutic target.


Similar content being viewed by others
References
Lang, K., Hao, Y., Huang, H., Lin, I., Rogerio, J. W., & Menzin, J. (2014). Treatment patterns among elderly patients with stage IV breast cancer treated with HER-2-targeted therapy. Journal of Comparative Effectiveness Research, 3(5), 481–490.
Tian, Z., Guo, B., Yu, M., Wang, C., Zhang, H., Liang, Q., et al. (2014). Upregulation of micro-ribonucleic acid-128 cooperating with downregulation of PTEN confers metastatic potential and unfavorable prognosis in patients with primary osteosarcoma. OncoTargets and Therapy, 7, 1601–1608.
Rappa, F., Farina, F., Zummo, G., David, S., Campanella, C., Carini, F., et al. (2012). HSP-molecular chaperones in cancer biogenesis and tumor therapy: An overview. Anticancer Research, 32(12), 5139–5150.
Lai, C. H., Park, K. S., Lee, D. H., Alberobello, A. T., Raffeld, M., Pierobon, M., et al. (2014). HSP-90 inhibitor ganetespib is synergistic with doxorubicin in small cell lung cancer. Oncogene, 33(40), 4867–4876.
Tanegashima, K., Tsuji, K., Suzuki, K., Shigenaga, A., Otaka, A., & Hara, T. (2013). Dimeric peptides of the C-terminal region of CXCL14 function as CXCL12 inhibitors. FEBS Letters, 587(23), 3770–3775.
Grzegrzolka, J., Kurnol, K., Piotrow, P., Pula, B., Kobierzycki, C., Piotrowska, A., et al. (2012). Hsp-27 expression in invasive ductal breast carcinoma. Folia histochemica et cytobiologica/Polish Academy of Sciences, Polish Histochemical and Cytochemical Society, 50(4), 527–533.
Nishizawa, S., Hirohashi, Y., Torigoe, T., Takahashi, A., Tamura, Y., Mori, T., et al. (2012). HSP DNAJB8 controls tumor-initiating ability in renal cancer stem-like cells. Cancer Research, 72(11), 2844–2854.
Kubo, T., Shimose, S., Fujimori, J., Furuta, T., Arihiro, K., & Ochi, M. (2015). Does expression of glucose transporter protein-1 relate to prognosis and angiogenesis in osteosarcoma? Clinical Orthopaedics and Related Research, 473, 305–310.
Zhang, C., Yao, C., Li, H., Wang, G., & He, X. (2014). Serum levels of microRNA-133b and microRNA-206 expression predict prognosis in patients with osteosarcoma. International Journal of Clinical and Experimental Pathology, 7(7), 4194–4203.
Lu, J., Wang, J., Yong, B., Song, G., Zhao, Z., Tang, Q., et al. (2013). Expression of chemokine CXCL14 in primary osteosarcoma and its association with prognosis. Nan fang yi ke da xue xue bao = Journal of Southern Medical University, 33(6), 798–803.
Gao, F., Hu, X., Xie, X., et al. (2015). Heat shock protein 90 stimulates rat mesenchymal stem cell migration via PI3K/Akt and ERK1/2 pathways. Cell Biochemistry and Biophysics, 71, 481–489.
Healy, E. F., Little, C., & King, P. J. (2014). A model for small heat shock protein inhibition of polyglutamine aggregation. Cell Biochemistry and Biophysics, 69(2), 275–281.
Ohashi, K., Burkart, V., Flohé, S., et al. (2000). Cutting edge: heat shock protein 60 is a putative endogenous ligand of the toll-like receptor-4 complex. The Journal of Immunology, 164(2), 558–561.
Lu, H. Q., Wang, Y. Z., Sun, P. H., Liang, S. P., Li, J., Wang, X. L., et al. (2012). Effects of heat shock protein 70-2 gene polymorphisms on the transcription of HSP 70-2 mRNA and the translation of HSP 70 protein in lung cancer. Zhonghua yu fang yi xue za zhi (Chinese Journal of Preventive Medicine), 46(5), 452–455.
Xavier, G. M., Panousopoulos, L., & Cobourne, M. T. (2013). Scube3 is expressed in multiple tissues during development but is dispensable for embryonic survival in the mouse. PLoS ONE, 8(1), e55274.
Chou, C. H., Cheng, Y. F., Siow, T. Y., Kumar, A., Peck, K., & Chang, C. (2013). SCUBE3 regulation of early lung cancer angiogenesis and metastatic progression. Clinical & Experimental Metastasis, 30(6), 741–752.
Wu, Y. Y., Peck, K., Chang, Y. L., Pan, S. H., Cheng, Y. F., Lin, J. C., et al. (2011). SCUBE3 is an endogenous TGF-beta receptor ligand and regulates the epithelial-mesenchymal transition in lung cancer. Oncogene, 30(34), 3682–3693.
Gu, X. L., Ou, Z. L., Lin, F. J., Yang, X. L., Luo, J. M., Shen, Z. Z., et al. (2012). Expression of CXCL14 and its anticancer role in breast cancer. Breast Cancer Research and Treatment, 135(3), 725–735.
Zeng, J., Yang, X., Cheng, L., Liu, R., Lei, Y., Dong, D., et al. (2013). Chemokine CXCL14 is associated with prognosis in patients with colorectal carcinoma after curative resection. Journal of Translational Medicine, 11, 6.
Park, C. R., You, D. J., Kim, D. K., Moon, M. J., Lee, C., Oh, S. H., et al. (2013). CXCL14 enhances proliferation and migration of NCI-H460 human lung cancer cells overexpressing the glycoproteins containing heparan sulfate or sialic acid. Journal of Cellular Biochemistry, 114(5), 1084–1096.
Acknowledgments
This work was supported by the Science Foundation of Zhejiang Province (LY13H060005), Public Technology Applied Research Projects of Zhejiang Province (2014C33254), General Foundation of Zhejiang Province (2013 KYA201), General Research Plan B of Zhejiang province (2012KYB213), and Shaoxing Science Project (Nos. 2013B70081, 2014B70075).
Author information
Authors and Affiliations
Corresponding author
Additional information
Wenqing Liang and Chengwei Yang have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Liang, W., Yang, C., Peng, J. et al. The Expression of HSPD1, SCUBE3, CXCL14 and Its Relations with the Prognosis in Osteosarcoma. Cell Biochem Biophys 73, 763–768 (2015). https://doi.org/10.1007/s12013-015-0579-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-015-0579-7