Abstract
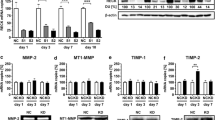
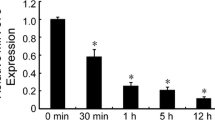
The objective of this study was to determine the role of Hsp90α in regulating the migration of mesenchymal stem cells (MSCs) and to investigate the underlying mechanisms of this effect. MSCs migration was assessed by wound healing assay and transwell migration assay. Hsp90α expression was silenced in MSC by siRNA (sirHsp90α). The activity of secreted metalloproteases MMP-2 and MMP-9, and their expression levels in MSC were evaluated using gelatin zymography, Western blot analysis and real-time PCR. Gene expression of VCAM-1 and CXCR4 cytokines was evaluated by real-time PCR. Akt and ERK activity were analyzed by Western blotting using antibodies against phosphorylated forms of these proteins. Treatment with Hsp90α significantly enhanced MSC migration, and this effect was blocked by transfecting MSC with sirHsp90α. Treating the cells with recombinant human Hsp90α (rhHsp90α) enhanced gene expression and protein levels of MMP-2 and MMP-9, as well as their secretion and activity. MSC incubated with rhHsp90α exhibited increased gene expression of CXCR4 and VCAM-1. Finally, the levels of phosphorylated Akt and Erk were markedly increased by rhHsp90α treatment. These findings indicate that Hsp90α promotes MSCs migration via PI3K/Akt and ERK signaling pathways, and that this effect is possibly mediated by MMPs, SDF-1/CXCR4 pathway, and VCAM-1.







Similar content being viewed by others
Change history
15 June 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
15 June 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Majumdar, M. K., Thiede, M. A., Haynesworth, S. E., Bruder, S. P., & Gerson, S. L. (2000). Human marrow-derived mesenchymal stem cells (MSCs) express hematopoietic cytokines and support long-term hematopoiesis when differentiated toward stromal and osteogenic lineages. Journal of Hematotherapy & Stem Cell Research, 9(6), 841–848.
Wright, V., Peng, H., Usas, A., et al. (2002). BMP4-expressing muscle-derived stem cells differentiate into osteogenic lineage and improve bone healing in immunocompetent mice. Molecular Therapy, 6(2), 169–178.
Mizuno, K., Muneta, T., Morito, T., et al. (2008). Exogenous synovial stem cells adhere to defect of meniscus and differentiate into cartilage cells. Journal of Medical and Dental Sciences, 55(1), 101–111.
Li, Y., Yu, J., Li, M., Qu, Z., & Ruan, Q. (2011). Mouse mesenchymal stem cells from bone marrow differentiate into smooth muscle cells by induction of plaque-derived smooth muscle cells. Life Sciences, 88(3–4), 130–140.
Wang, Q. W., Chen, Z. L., & Piao, Y. J. (2005). Mesenchymal stem cells differentiate into tenocytes by bone morphogenetic protein (BMP) 12 gene transfer. Journal of Bioscience and Bioengineering, 100(4), 418–422.
Elabd, C., Chiellini, C., Carmona, M., et al. (2009). Human multipotent adipose-derived stem cells differentiate into functional brown adipocytes. Stem Cells, 27(11), 2753–2760.
Psaltis, P. J., Zannettino, A. C., Worthley, S. G., & Gronthos, S. (2008). Concise review: Mesenchymal stromal cells: potential for cardiovascular repair. Stem Cells, 26(9), 2201–2210.
Marenzi, G., & Bartorelli, A. L. (2007). Improved clinical outcome after intracoronary administration of bone marrow-derived progenitor cells in acute myocardial infarction: final 1-year results of the REPAIR-AMI trial. European Heart Journal, 28(17), 2172–2173. author reply 2173–2174.
Fuh, E., & Brinton, T. J. (2009). Bone marrow stem cells for the treatment of ischemic heart disease: A clinical trial review. Journal of Cardiovascular Translational Research, 2(2), 202–218.
Barbash, I. M., Chouraqui, P., Baron, J., et al. (2003). Systemic delivery of bone marrow-derived mesenchymal stem cells to the infarcted myocardium: feasibility, cell migration, and body distribution. Circulation, 108(7), 863–868.
Langer, T., Rosmus, S., & Fasold, H. (2003). Intracellular localization of the 90 kDA heat shock protein (HSP90alpha) determined by expression of a EGFP-HSP90alpha-fusion protein in unstressed and heat stressed 3T3 cells. Cell Biology International, 27(1), 47–52.
Yano, A., Tsutsumi, S., Soga, S., et al. (2008). Inhibition of Hsp90 activates osteoclast c-Src signaling and promotes growth of prostate carcinoma cells in bone. Proceedings of the National Academy of Sciences of the United States of America, 105(40), 15541–15546.
Wu, L. X., Xu, J. H., Huang, X. W., Zhang, K. Z., Wen, C. X., & Chen, Y. Z. (2006). Down-regulation of p210(bcr/abl) by curcumin involves disrupting molecular chaperone functions of Hsp90. Acta Pharmacologica Sinica, 27(6), 694–699.
Zsebik, B., Citri, A., Isola, J., Yarden, Y., Szollosi, J., & Vereb, G. (2006). Hsp90 inhibitor 17-AAG reduces ErbB2 levels and inhibits proliferation of the trastuzumab resistant breast tumor cell line JIMT-1. Immunology Letters, 104(1–2), 146–155.
Giaccone, G., & Rajan, A. (2009). Met amplification and HSP90 inhibitors. Cell Cycle, 8(17), 2682.
Grammatikakis, N., Lin, J. H., Grammatikakis, A., Tsichlis, P. N., & Cochran, B. H. (1999). p50(cdc37) acting in concert with Hsp90 is required for Raf-1 function. Molecular and Cellular Biology, 19(3), 1661–1672.
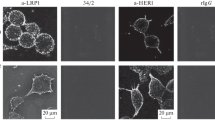
Sidera, K., Gaitanou, M., Stellas, D., Matsas, R., & Patsavoudi, E. (2008). A critical role for HSP90 in cancer cell invasion involves interaction with the extracellular domain of HER-2. Journal of Biological Chemistry, 283(4), 2031–2041.
Tsutsumi, S., Scroggins, B., Koga, F., et al. (2008). A small molecule cell-impermeant Hsp90 antagonist inhibits tumor cell motility and invasion. Oncogene, 27(17), 2478–2487.
Gao, F., Hu, X. Y., Xie, X. J., et al. (2010). Heat shock protein 90 protects rat mesenchymal stem cells against hypoxia and serum deprivation-induced apoptosis via the PI3K/Akt and ERK1/2 pathways. Journal of Zhejiang University-Science B, 11(8), 608–617.
Xie, X. J., Wang, J. A., Cao, J., & Zhang, X. (2006). Differentiation of bone marrow mesenchymal stem cells induced by myocardial medium under hypoxic conditions. Acta Pharmacologica Sinica, 27(9), 1153–1158.
Heussen, C., & Dowdle, E. B. (1980). Electrophoretic analysis of plasminogen activators in polyacrylamide gels containing sodium dodecyl sulfate and copolymerized substrates. Analytical Biochemistry, 102(1), 196–202.
Baer, P. C., & Geiger, H. (2010). Mesenchymal stem cell interactions with growth factors on kidney repair. Current Opinion in Nephrology and Hypertension, 19(1), 1–6.
Walsh, D., Li, Z., Wu, Y., & Nagata, K. (1997). Heat shock and the role of the HSPs during neural plate induction in early mammalian CNS and brain development. Cellular and Molecular Life Sciences, 53(2), 198–211.
Lesko, E., Gozdzik, J., Kijowski, J., Jenner, B., Wiecha, O., & Majka, M. (2007). HSP90 antagonist, geldanamycin, inhibits proliferation, induces apoptosis and blocks migration of rhabdomyosarcoma cells in vitro and seeding into bone marrow in vivo. Anti-Cancer Drugs, 18(10), 1173–1181.
Minami, Y., Kawasaki, H., Miyata, Y., Suzuki, K., & Yahara, I. (1991). Analysis of native forms and isoform compositions of the mouse 90-kDa heat shock protein, HSP90. Journal of Biological Chemistry, 266(16), 10099–10103.
Nemoto, T., & Sato, N. (1998). Oligomeric forms of the 90-kDa heat shock protein. Biochemical Journal, 330(Pt 2), 989–995.
Grenert, J. P., Sullivan, W. P., Fadden, P., et al. (1997). The amino-terminal domain of heat shock protein 90 (hsp90) that binds geldanamycin is an ATP/ADP switch domain that regulates hsp90 conformation. Journal of Biological Chemistry, 272(38), 23843–23850.
Basso, A. D., Solit, D. B., Chiosis, G., Giri, B., Tsichlis, P., & Rosen, N. (2002). Akt forms an intracellular complex with heat shock protein 90 (Hsp90) and Cdc37 and is destabilized by inhibitors of Hsp90 function. Journal of Biological Chemistry, 277(42), 39858–39866.
Roe, S. M., Ali, M. M., Meyer, P., et al. (2004). The mechanism of Hsp90 regulation by the protein kinase-specific cochaperone p50(cdc37). Cell, 116(1), 87–98.
Sato, S., Fujita, N., & Tsuruo, T. (2000). Modulation of Akt kinase activity by binding to Hsp90. Proceedings of the National Academy of Sciences of the United States of America, 97(20), 10832–10837.
Abi-Younes, S., Sauty, A., et al. (2000). The stromal cell-derived factor-1 chemokine is a potent platelet agonist highly expressed in atherosclerotic plaques. Circulation Research, 86(2), 131–138.
Yamaguchi, J., Kusano, K. F., et al. (2003). Stromal cell-derived factor-1 effects on ex vivo expanded endothelial progenitor cell recruitment for ischemic neovascularization. Circulation, 107(9), 1322–1328.
Oberlin, E., Amara, A., et al. (1996). The CXC chemokine SDF-1 is the ligand for LESTR/fusin and prevents infection by T-cell-line-adapted HIV-1. Nature, 382(6594), 833–835.
Jo, D. Y., Rafii, S., et al. (2000). Chemotaxis of primitive hematopoietic cells in response to stromal cell-derived factor-1. Journal of Clinical Investigation, 105(1), 101–111.
Lima e Silva, R., Shen, J., et al. (2007). The SDF-1/CXCR4 ligand/receptor pair is an important contributor to several types of ocular neovascularization. FASEB Journal, 21(12), 3219–3230.
Abbott, J. D., Huang, Y., et al. (2004). Stromal cell-derived factor-1alpha plays a critical role in stem cell recruitment to the heart after myocardial infarction but is not sufficient to induce homing in the absence of injury. Circulation, 110(21), 3300–3305.
Guo, Y., Hangoc, G., et al. (2005). SDF-1/CXCL12 enhances survival and chemotaxis of murine embryonic stem cells and production of primitive and definitive hematopoietic progenitor cells. Stem Cells, 23(9), 1324–1332.
Shao, H., Tan, Y., et al. (2008). Statin and stromal cell-derived factor-1 additively promote angiogenesis by enhancement of progenitor cells incorporation into new vessels. Stem Cells, 26(5), 1376–1384.
Hyun, Y. M., Chung, H. L., et al. (2009). Activated integrin VLA-4 localizes to the lamellipodia and mediates T cell migration on VCAM-1. The Journal of Immunology, 183(1), 359–369.
Bowden, R. A., Ding, Z. M., et al. (2002). Role of alpha4 integrin and VCAM-1 in CD18-independent neutrophil migration across mouse cardiac endothelium. Circulation Research, 90(5), 562–569.
Matsuura, K., Honda, A., et al. (2009). Transplantation of cardiac progenitor cells ameliorates cardiac dysfunction after myocardial infarction in mice. Journal of Clinical Investigation, 119(8), 2204–2217.
Lu, L., Zhang, J. Q., et al. (2004). Molecular and cellular events at the site of myocardial infarction: from the perspective of rebuilding myocardial tissue. Biochemical and Biophysical Research Communications, 320(3), 907–913.
Acknowledgments
This project was supported by the National Natural Science Foundation of China (Nos. 30670868, 30770887, and 30770887/H0220), the Traditional Chinese Medicine of Zhejiang Province (No. 2014ZA005), and the Zhejiang Provincial Natural Science foundation of China (Y2100362) and Science Technology Department of Zhejiang Province (2010R10G2010160). China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gao, F., Hu, X., Xie, X. et al. Heat Shock Protein 90 Stimulates Rat Mesenchymal Stem Cell Migration via PI3K/Akt and ERK1/2 Pathways. Cell Biochem Biophys 71, 481–489 (2015). https://doi.org/10.1007/s12013-014-0228-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0228-6