Abstract
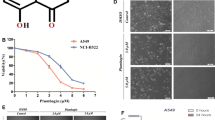
Daidzein belongs to the group of isoflavones, found in a wide variety of plant-derived foods, especially in soybeans and soy-based foods. In this study, the effect of daidzein on human gastric carcinoma cells (BGC-823) and its mechanism were investigated. MTT assay was applied in the detection of the inhibitory effects of daidzein on cell proliferation. Hoechst–propidium iodide staining and flow cytometry were used to examine the apoptosis as well as the mitochondrial transmembrane potential. Western blotting was performed to detect the expression of apoptosis-associated proteins: cleaved PARP, cleaved caspase-9, cleaved caspase-3, Bcl-2, and Bax. Daidzein significantly inhibited the growth and proliferation of human gastric carcinoma cells (BGC-823) in a concentration- and time-dependent manner. Furthermore, it was found that an insult of daidzein to BGC-823 cells caused them to die by disruption of mitochondrial transmembrane potential, demonstrated not only by staining dead cells for phosphatidylserine but also by the up-regulation (cleaved PARP, cleaved caspase-9, cleaved caspase-3, Bax) and down-regulation (Bcl-2) of proteins associated with apoptosis and survival; whereas, the pan-caspase inhibitor z-VAD-fmk could partially rescue cells against damage of daidzein. Taken together, the results of this study demonstrate that daidzein significantly induces apoptosis via a mitochondrial pathway. Specifically, daidzein induced a change in the Bax/Bcl-2 ratios and activation of caspases-3 and -9 and the cleavage of PARP. Therefore, daidzein has the potential for use as a therapeutic agent for the treatment of gastric carcinoma.



Similar content being viewed by others
References
Ferlay, J., Parkin, D. M., Raymond, L., Whelan, S. L., & Young, J. (1997). Cancer incidence in five continents. IARC Scientific Publications, 7(143), i–xxxiv, 1–1240.
Larsson, S. C., Bergkvist, L., & Wolk, A. (2006). Processed meat consumption, dietary nitrosamines and stomach cancer risk in a cohort of Swedish women. International Journal of Cancer, 119, 915–919.
Friedman, G. D., & Parsonnet, J. (1992). Salt intake and stomach cancer: some contrary evidence. Cancer Epidemiology, Biomarkers and Prevention, 1, 607–608.
Honjo, S., Kono, S., & Yamaguchi, M. (1994). Salt and geographic variation in stomach cancer mortality in Japan. Cancer Causes and Control, 5, 285–286.
Kim, H. J., Chang, W. K., Kim, M. K., Lee, S. S., & Choi, B. Y. (2002). Dietary factors and gastric cancer in Korea: a case-control study. International Journal of Cancer, 97, 531–535.
Wu, A. H., Yang, D., & Pike, M. C. (2000). A meta-analysis of soyfoods and risk of stomach cancer: the problem of potential confounders. Cancer Epidemiology, Biomarkers and Prevention, 9, 1051–1058.
Hoshiyama, Y., & Sasaba, T. (1992). A case-control study of stomach cancer and its relation to diet, cigarettes, and alcohol consumption in Saitama prefecture, Japan. Cancer Causes and Control, 3, 441–448.
Chyou, P. H., Nomura, A. M., Hankin, J. H., & Stemmermann, G. N. (1990). A case-cohort study of diet and stomach cancer. Cancer Research, 50, 7501–7504.
Kurahashi, N., Iwasaki, M., Sasazuki, S., Otani, T., Inoue, M., & Tsugane, S. (2007). Soy product and isoflavone consumption in relation to prostate cancer in Japanese men. Cancer Epidemiology, Biomarkers and Prevention, 16, 538–545.
Iwasaki, M., Inoue, M., Otani, T., Sasazuki, S., Kurahashi, N., Miura, T., et al. (2008). Japan Public Health Center-based prospective study group. Plasma isoflavone level and subsequent risk of breast cancer among Japanese women: a nested case-control study from the Japan Public Health Center-based prospective study group. Journal of Clinical Oncology, 26, 1677–1683.
Sarkar, F. H., & Li, Y. (2002). Mechanisms of cancer chemoprevention by soy isoflavone genistein. Cancer and Metastasis Reviews, 21, 265–280.
Tatsuta, M., Iishi, H., Baba, M., Yano, H., Uehara, H., & Nakaizumi, A. (1999). Attenuation by genistein of sodium-chloride-enhanced gastric carcinogenesis induced by N-methyl-N′-nitro-N-nitrosoguanidine in Wistar rats. International Journal of Cancer, 80, 396–399.
Verdrengh, M., Jonsson, I. M., Holmdahl, R., & Tarkowski, A. (2003). Genistein as an anti-inflammatory agent. Inflammation Research, 52, 341–346.
Nagata, C., Takatsuka, N., Kawakami, N., & Shimizu, H. (2002). A prospective cohort study of soy product intake and stomach cancer death. British Journal of Cancer, 87, 31–36.
Liggins, J., Bluck, L. J., Runswick, S., Atkinson, C., Coward, W. A., & Bingham, S. A. (2000). Daidzein and genistein content of fruits and nuts. Journal of Nutritional Biochemistry, 11, 326–331.
Liggins, J., Mulligan, A., Runswick, S., & Bingham, S. A. (2002). Daidzein and genistein content of cereals. European Journal of Clinical Nutrition, 56, 961–966.
Guo, J. M., Xiao, B. X., Dai, D. J., Liu, Q., & Ma, H. H. (2004). Effects of daidzein on estrogen-receptor-positive and negative pancreatic cancer cells in vitro. World Journal of Gastroenterology, 10, 860–863.
Guo, J. M., Xiao, B. X., Liu, D. H., Grant, M., Zhang, S., Lai, Y. F., et al. (2004). Biphasic effect of daidzein on cell growth of human colon cancer cells. Food and Chemical Toxicology, 42, 1641–1646.
Gercel-Taylor, C., Feitelson, A. K., & Taylor, D. D. (2004). Inhibitory effect of genistein and daidzein on ovarian cancer cell growth. Anticancer Research, 24, 795–800.
Choi, E. J., & Kim, G. H. (2008). Daidzein causes cell cycle arrest at the G1 and G2/M phases in human breast cancer MCF-7 and MDA-MB-453 cells. Phytomedicine, 15, 683–690.
Constantinou, A. I., Lantvit, D., Hawthorne, M., Xu, X., van Breemen, R. B., & Pezzuto, J. M. (2001). Chemopreventive effects of soy protein and purified soy isoflavones on DMBA-induced mammary tumors in female Sprague–Dawley rats. Nutrition and Cancer, 41, 75–81.
Jin, Z., & MacDonald, R. S. (2002). Soy isoflavones increase latency of spontaneous mammary tumors in mice. Journal of Nutrition, 132, 3186–3190.
Ko, K. P., Park, S. K., Park, B., Yang, J. J., Cho, L. Y., Kang, C., et al. (2010). Isoflavones from phytoestrogens and gastric cancer risk: A nested case-control study within the Korean multicenter cancer cohort. Cancer Epidemiology, Biomarkers and Prevention, 19, 1292–1300.
Hirsch, T., Marzo, I., & Kroemer, G. (1997). Role of the mitochondrial permeability transition pore in apoptosis. Bioscience Reports, 17, 67–76.
Iannolo, G., Conticello, C., Memeo, L., & De Maria, R. (2008). Apoptosis in normal and cancer stem cells. Critical Reviews in Oncology Hematology, 66, 42–51.
Mohamad, N., Gutierrez, A., Nunez, M., Cocca, C., Martín, G., Cricco, G., et al. (2005). Mitochondrial apoptotic pathways. Biocell, 29, 149–161.
Orrenius, S., Gogvadze, V., & Zhivotovsky, B. (2007). Mitochondrial oxidative stress: implications for cell death. Annual Review of Pharmacology and Toxicology, 47, 143–183.
Yin, X. M. (2000). Signal transduction mediated by Bid, a pro-death Bcl-2 family proteins, connects the death receptor and mitochondria apoptosis pathways. Cell Research, 10, 161–167.
Takahashi, A., Masuda, A., Sun, M., Centonze, V. E., & Herman, B. (2004). Oxidative stress-induced apoptosis is associated with alterations in mitochondrial caspase activity and Bcl-2-dependent alterations in mitochondrial pH (pHm). Brain Research Bulletin, 62, 497–504.
Li, M., Kondo, T., Zhao, Q. L., Li, F. J., Tanabe, K., Arai, Y., et al. (2000). Apoptosis induced by cadmium in human lymphoma U937 cells through Ca2+-calpain and caspase-mitochondria dependent pathways. Journal of Biological Chemistry, 275, 39702–39709.
Acknowledgments
The authors are grateful to Dr. Xue (Harbin veterinary research institute, CAAS) for his technical help. This work was supported by the Heilongjiang Provincial Health Department research projects (2010-024).
Author information
Authors and Affiliations
Corresponding author
Additional information
Shuyao Tang, Jing Hu, and Qingfeng Meng contributed equally to this work.
Rights and permissions
About this article
Cite this article
Tang, S., Hu, J., Meng, Q. et al. Daidzein Induced Apoptosis via Down-Regulation of Bcl-2/Bax and Triggering of the Mitochondrial Pathway in BGC-823 Cells. Cell Biochem Biophys 65, 197–202 (2013). https://doi.org/10.1007/s12013-012-9418-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-012-9418-2