Abstract
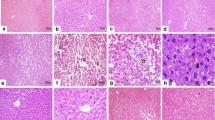
The aim of the study was to assess the protective effect of Picrorhiza kurroa hydroalcoholic extract (PCK), a glycoside-rich extract, against potassium dichromate (PDC)-induced liver oxidative stress in Wistar albino rats. Thirty-six male Wistar rats were divided into six groups: the control group (which received distilled water), the SIL group (which received 60 mg/kg silymarin), the PDC group (which received 30 mg/kg K2Cr2O7), and the treatment groups (which received 25, 50, 100 mg/kg PCK). Administration of PDC resulted in increased levels of liver enzymes such as alanine transaminase (ALT), aspartate transaminase (AST), and alkaline phosphatase (ALP); up-regulated peroxidation biomarkers, i.e., thiobarbutric acid-reactive species (TBARS) and protein carbonyls in serum; and decreased activities of antioxidant enzymes like superoxide dismutase (SOD) and catalase (CAT) significantly in the liver tissue. Gene expression studies of tumor necrosis factor (TNF), mitogen-activated protein kinase (MAPK), growth arrest, and DNA damage-inducible protein (GADD45) revealed that there was a liver damage at the molecular level, and histopathological studies further confirmed the morphological changes by PDC administration. However, PCKs at 50 and 100 mg/kg promoted significant restoration of liver enzyme levels and the activities of antioxidant enzymes were kept close to the values of the control and SIL groups. Our current study confirms that the active compounds present in the PCK might have conferred a strong protection against potassium dichromate-induced oxidative stress.








Similar content being viewed by others
References
Mishra S, Bharagava RN (2016) Toxic and genotoxic effects of hexavalent chromium in environment and its bioremediation strategies. J Environ Sci Heal C 34:1–32. https://doi.org/10.1080/10590501.2015.1096883
U.S. EPA. IRIS toxicological review of hexavalent chromium (2010). U.S. Environmental Protection Agency, Washington, DC, EPA/635/R-10/004A
Laborda R, Diaz-Mayans J, Nunez A (1986) Nephrotoxic and hepatotoxic effects of chromium compounds in rats. Bull Environ Contam Toxicol 36:332–336
Soudani N, Ben Amara I, Sefi M, Boudawara T, Zeghal N (2011) Effects of selenium on chromium (VI)-induced hepatotoxicity in adult rats. Exp Toxicol Pathol 63:541–548. https://doi.org/10.1016/j.etp.2010.04.005
Sugiyama M (1992) Role of physiological antioxidants in chromium (VI)-induced cellular injury. Free Radical Biol. Med 12:397–407. https://doi.org/10.1016/0891-5849 (92)90089-Y
Patlolla C, Barnes C, Yedjou V, Velma P (2009) Oxidative stress, DNA damage, and antioxidant enzyme activity induced by hexavalent chromium in Sprague-Dawley rats. Environ Toxicol 24:66–73. https://doi.org/10.1002/tox.20395
Das AP, Mishra S (2008) Hexavalent chromium (VI): environment pollutant and health hazard. J Environ Research and Development 2:386–392
Saber TM, Farag MR, Cooper RG (2015) Ameliorative effect of extra virgin olive oil on hexavalent chromium-induced nephrotoxicity and genotoxicity in rat. Rev. Med. Vet. 166:11-19
Sahu B, Meghana K, Shriharsh R (2014) Chromium-induced nephrotoxicity and ameliorative effect of carvedilol in rats: involvement of oxidative stress, apoptosis and inflammation. Chem. Biol. Interact 223:69–79. https://doi.org/10.1016/j.cbi.2014.09.009
Li S, Tan HY, Wang N, Zhang ZJ, Lao L, Wong CW, Feng Y (2015) The role of oxidative stress and antioxidants in liver diseases. Int J Mol Sci 16:26087–26124. https://doi.org/10.3390/ijms161125942
Ravishankar B, Shukla VJ (2007) Indian systems of medicine: a brief profile. Afr J Tradit Complement Altern Med 4:319–337
Jadeja R, Devkar RV, Nammi S (2014) Herbal medicines for the treatment of non-alcoholic steatohepatitis: current scenario and future prospects. Evid Based Complement Alternat Med 2014:648308. https://doi.org/10.1155/2014/648308
Stuppner H, Wagner H (1989) New cucurbitacin glycosides from Picrorhiza kurrooa. Planta Med 55:559–563. https://doi.org/10.1055/s-2006-962095
Abou Zid S (2012) Silymarin, Natural Flavonolignans from Milk Thistle. In: Venketeshwer R. Phytochemicals-A Global Perspective of Their Role in Nutrition and Health. Rijeka: Croatia. InTech 2012: 255-272
Gutteridge J, Halliwell B (2000) Free radicals and antioxidants in the year 2000: a historical look to the future. Ann N Y Acad Sci 899:136–147
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Rad Bio Med 26:231–237. https://doi.org/10.1016/S0891-5849(98)00315-3
Ruch RJ, Cheng SJ, Klaunig JE (1989) Prevention of cytotoxicity and inhibition of intracellular communication by antioxidant catechins isolated from Chinese green tea. Carcinogenesis 10:1003–1008
Benzie IFF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal Biochem 239:70–76. https://doi.org/10.1006/abio.1996.0292
Adjroud O (2010) Protective effects of selenium against potassium dichromate-induced hematotoxicity in female and male Wistar albino rats. Ann Toxicol Anal 22:165–172. https://doi.org/10.1051/ata/2010025
Jeyakumar R, Rajesh R, Meena B, Rajaprabhu D, Ganesan B, Buddhan S, Anandan R (2008) Antihepatotoxic effect of Picrorhiza kurroa on mitochondrial defense system in antitubercular drugs (isoniazid and rifampicin)-induced hepatitis in rats. J Med Plants Res 2:017–019
Garcia N, Tapia WR, Zazueta E, Zatarain C, Barron ZL (2013) Curcumin pretreatment prevents potassium dichromate induced hepatotoxicity, oxidative stress, decreased respiratory complex I activity, and membrane permeability transition pore opening. Evid Based Complement Alternat Med 2013:1–19. https://doi.org/10.1155/2013/424692
Dalle-Donne I, Rossi R, Giustarini D, Milzani A, Colombo R (2003) Protein carbonyl groups as biomarkers of oxidative stress. Clin Chim Acta 329:23–38. https://doi.org/10.1016/S0009-8981(03)00003-2
Chandrasekhar Y, Ramya EM, Navya K, Kumar GP, Anilakumar KR (2017) Antidepressant like effects of hydrolysable tannins of Terminalia catappa leaf extract via modulation of hippocampal plasticity and regulation of monoamine neurotransmitters subjected to chronic mild stress (CMS). Biomed Pharmacother 86:414–425
Duruibe JO, Ogwuegbu MOC, Egwurugwu JN (2007) Heavy metal pollution and human biotoxic effects. Int J Phys Sci 2:112–118
García-Niño WR, Tapia E, Zazueta C, Zatarain-Barrón ZL, Hernández-Pando R, Vega-García CC, Pedraza-Chaverrí J (2013) Curcumin pre-treatment prevents potassium dichromate-induced hepatotoxicity, oxidative stress, decreased respiratory complex I activity, and membrane permeability transition pore opening. Evid Based Complement Alternat Med 2013:424692. https://doi.org/10.1155/2013/424692
Sterekhova NP, Zeleneva NI, Solomina SN, Tiushniakova NV, Miasnikova AG (1978) Stomach pathology in the workers of chromium salts industries. Gig Trud Prof Zabol 3:19–23
Susa N, Ueno S, Furukawa Y, Ueda J, Sugiyama M (1997) Potent protective effect of melatonin on chromium(VI)-induced DNA single-strand breaks, cytotoxicity, and lipid peroxidation in primary cultures of rat hepatocytes. Toxicol Appl Pharmacol 144:377–384. https://doi.org/10.1006/taap.1997.8151
Jayakumar T, Ramesh E, Geraldine P (2006) Antioxidant activity of the oyster mushroom, Pleurotus ostreatus, on CCl4-induced liver injury in rats. Food Chem Toxicol 44:1989–1996. https://doi.org/10.1016/j.fct.2006.06.025
Kumar GP, Anilakumar KR, Naveen S (2015) Phytochemicals having neuroprotective properties from dietary sources and medicinal herbs. Pharmacogn J 7:1–17. https://doi.org/10.5530/pj.2015.7.1
Del Carmen G-RM, Nicolás-Méndez T, Montaño-Rodríguez AR, Altamirano-Lozano MA (2014) Antigenotoxic effects of (−)-epigallocatechin-3-gallate (EGCG), quercetin, and rutin on chromium trioxide-induced micronuclei in the polychromatic erythrocytes of mouse peripheral blood. J Tox Env Health, Part A 77(6):324–336. https://doi.org/10.1080/15287394.2013.865006
Valko MMHCM, Morris H, Cronin MTD (2005) Metals, toxicity and oxidative stress. Curr Med Chem 12:1161–1208. https://doi.org/10.2174/0929867053764635
Abreu PL, Ferreira LMR, Alpoim MC, Urbano AM (2014) Impact of hexavalent chromium on mammalian cell bioenergetics: phenotypic changes, molecular basis and potential relevance to chromate-induced lung cancer. Biometals 27:409–443
Navya K, Phani Kumar G, Anilakumar KR (2017). Ameliorating effect of Curculigo orchoides on chromium (VI) induced oxidative stress via, modulation of cytokines, transcription factors and apoptotic genes. J Appl Biomed. doi.org/10.1016/j.jab.2017.03.003. (in press)
Tracey MD, Cerami A (1994) Tumor necrosis factor: a pleiotropic cytokine and therapuetic target. Annu Rev Med 45:491–503. https://doi.org/10.1146/annurev.med.45.1.491
Ip YT, Davis RJ (1998) Signal transduction by the c-Jun N-terminal kinase (JNK)–from inflammation to development. Curr Opin Cell Biol 10:205–219. https://doi.org/10.1016/S0955-0674(98)80143-9
Kyriakis JM, Avruch J (1996) Protein kinase cascades activated by stress and inflammatory cytokines. BioEssays 18:567–577. https://doi.org/10.1002/bies.950180708
Liu KJ, Shi X (2001) In vivo reduction of chromium (VI) and its related free radical generation. Mol Cell Biochem 222:41–47
Pourahmad J, Mihajlovic A, Brien PJO (2001) Hepatocyte lysis induced by environmental metal toxins may involve apoptotic death signals initiated by mitochondrial injury. Adv Exp Med Biol 500:249–252
Sonnenbichler J, Goldbero M, Hane L, Madubunyi I, Vogl S, Zetl I (1986) Stimulatory effect of silibinin on the DNA synthesis in partially hepatectomized rat livers: non-response in hepatoma and other malign cell lines. Biochem Pharmacol 35:538–541. https://doi.org/10.1016/0006-2952(86)90233-9
Tsapakos MJ, Hampton TH, Wetterhahn KE (1983) Chromium (VI)-induced DNA lesions and chromium distribution in rat kidney, liver, and lung. Cancer Res 43:5662–5667
Acknowledgements
This research was supported by the Defense Food Research Laboratory, Mysore.
Author information
Authors and Affiliations
Contributions
KN and CSY conducted the experiments and collected and analyzed the data. PKG and KRA helped in designing and discussing the study and writing the manuscript. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Animal studies were conducted according to the Institute Animal Ethical Committee (IAEC) regulations approved by the Committee for the Purpose of the Control and Supervision of Experiments on Animals (CPCSEA) with the IAEC approval no. IAEC-2016/AN/12 dated 25 October 2016.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
K, N., G, P., Y, C. et al. Evaluation of Potassium Dichromate (K2Cr2O7)-Induced Liver Oxidative Stress and Ameliorative Effect of Picrorhiza kurroa Extract in Wistar Albino Rats. Biol Trace Elem Res 184, 154–164 (2018). https://doi.org/10.1007/s12011-017-1172-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-017-1172-2