Abstract
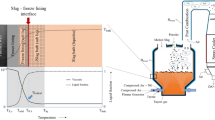
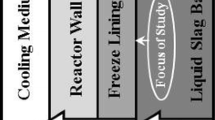
A major industrial problem in high-temperature liquid reaction systems is the attack of furnace components by chemically aggressive molten reactants. Freeze-lining technologies involving the deliberate formation of controlled frozen deposits are increasingly being applied to extend the range of liquid bath compositions and process temperatures that can be used; this has resulted in significant increases in process performance and productivity. It has been widely assumed that the interface between the stationary frozen layer and the agitated molten bath at steady state consists of the primary phase, which stays in contact with the bulk liquid at the liquidus temperature, T liquidus. It has been shown in the current laboratory-based studies through the use of a cold finger technique that, at steady state and in selected ranges of process conditions and bath compositions, the phase assemblage present at the deposit/liquid interface is not that of the primary phase alone. The microstructural observations clearly demonstrate that the temperature of the deposit/liquid bath interface, T f, can be lower than the liquidus temperature of the bulk liquid, T liquidus. These observations point to a significant change in the mechanism and behavior of the systems. To explain this phenomenon, it is proposed that the steady-state thickness of freeze linings is not the result of equilibrium freezing but rather represents a state of dynamic equilibrium that is critically dependent on the relative rates of crystallization, mass, and heat transfer processes, occurring close to and at the deposit interface. The mechanisms taking place in the boundary liquid layer involve both partial crystallization/remelting and continuous removal of solids. This finding has important implications for the design of the high-temperature industrial reactors and selection of ranges of melt chemistries and conditions that can be used. This finding means that temperatures below the liquidus can be selected for some processes, resulting potentially in significant savings of energy and increases in throughput of pyrometallurgical reactors. The findings are generic and are not limited to the specific chemical systems reported in the article.









Similar content being viewed by others
References
F. Guevara and G. Irons: Metall. Mater. Trans. B, 2011, vol. 42B, pp. 652-63.
F. Guevara and G. Irons: Metall. Mater. Trans. B, 2011, vol. 42B, pp. 664-76.
P.C. Pistorius: SAIMM, 2003, vol. 103, no. 8, pp. 509-14.
P.C. Pistorius: SAIMM, 2004, vol. 104, no. 7, pp. 417-22.
J.H. Zietsman and C. Pistorius: Miner. Eng., 2005, vol. 19, no. 3, pp. 262- 79.
D.G.C. Robertson and S. Kang: Fluid Flow Phenomena in Minerals Processing, TMS, Warrendale, PA, 1999, pp. 157-68.
K. Verscheure, M.V. Camp, B. Blanpain, P. Wollants, P. Hayes, and E. Jak: Metall. Mater. Trans. B, 2007, vol. 38B, pp. 13-20.
K. Verscheure, F. Verhaeghe, E. Boydens, M.V. Camp, B. Blanpain, and P. Wollants: Metall. Mater. Trans. B, 2006, vol. 37B, pp. 929-40.
K. Verscheure, A.K. Kyllo, A. Filzwieser, B. Blanpain, and P. Wollants: Proc. Conf. on Non-Ferrous Materials Extraction and Processing, TMS, Warrendale, PA, 2006, pp. 139-54.
A. Fallah-Mehrjardi, P.C. Hayes, and E. Jak: Metall. Mater. Trans. B, 2013. doi:10.1007/s11663-013-9806-z.
M. Campforts, B. Blanpain, and P. Wollants: Metall. Mater. Trans. B, 2009, vol. 40B, pp. 643-55.
M. Campforts, E. Jak, B. Blanpain, and P. Wollants: Metall. Mater. Trans. B, 2009, vol. 40B, pp. 619-31.
M. Campforts, E. Jak, B. Blanpain, and P. Wollants: Metall. Mater. Trans. B, 2009, vol. 40B, pp. 632-42.
M. Campforts, K. Verscheure, E. Boydens, T.V. Rompaey, B. Blanpain, and P. Wollants: Metall. Mater. Trans. B, 2008, vol. 39B, pp. 408-17.
M. Campforts, K. Verscheure, T.V. Rompaey, E. Boydens, B. Blanpain, and P. Wollants: Metall. Mater. Trans. B, 2007, vol. 38B, pp. 841-51.
MiekeCampforts, K. Verscheure, F. Verhaeghe, T.V. Rompaey, E. Boydens, B. Blanpain, and P. Wollants: Non-Ferrous Materials Extraction and Processing, TMS, Warrendale, PA, 2006, pp. 309–21.
FactSage ver. 6.2., CRCT-Thermfact Inc & GTT-Technologies, Montreal, Canada, 2010.
T. Hidayat and E. Jak: CALPHAD XLI (private communication), Berkeley, CA, June 3–8, 2012.
P.V. Danckwerts: Ind. Eng. Chem., 1951, vol. 43, p. 1460.
M.C. Flemings: Solidification Processing, McGraw-Hill, New York, NY, 1974.
W. Kurz and D.J. Fisher: Fundamentals of Solidification, 3rd ed., Trans Tech Publications, Aedermannsdorf, Switzerland, 1989.
D. Ruvalcaba, R.H. Mathiesen, D.G. Eskin, L. Arnberg, and L. Katgerman: Acta Mater., 2007, vol. 55, no. 13, pp. 4287-92.
D.N. Thomas and G.S. Dieckmann: Sea Ice: An Introduction to its Physics, Chemistry, Biology, and Geology, Blackwell Science, New York, NY, 2003.
Acknowledgments
The authors would like to thank the Australian Research Council Linkage program, Rio Tinto Kennecott Utah Copper, Corp., Xstrata Technology, Xstrata Copper, BHP Billiton Olympic Dam Operation, and Outotec Finland Oy for their financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted July 27, 2012.
Rights and permissions
About this article
Cite this article
Fallah-Mehrjardi, A., Hayes, P.C. & Jak, E. Investigation of Freeze Linings in Copper-Containing Slag Systems: Part II. Mechanism of the Deposit Stabilization. Metall Mater Trans B 44, 549–560 (2013). https://doi.org/10.1007/s11663-013-9807-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-013-9807-y