Abstract
Purpose
Minimally invasive treatment of solid cancers, especially in the breast and liver, remains clinically challenging, despite a variety of treatment modalities, including radiofrequency ablation (RFA), microwave ablation or high-intensity focused ultrasound. Each treatment modality has advantages and disadvantages, but all are limited by placement of a probe or US beam in the target tissue for tumor ablation and monitoring. The placement is difficult when the tumor is surrounded by large blood vessels or organs. Patient-specific image-based 3D modeling for thermal ablation simulation was developed to optimize treatment protocols that improve treatment efficacy.
Methods
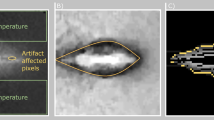
A tissue-mimicking breast gel phantom was used to develop an image-based 3D computer-aided design (CAD) model for the evaluation of a planned RF ablation. First, the tissue-mimicking gel was cast in a breast mold to create a 3D breast phantom, which contained a simulated solid tumor. Second, the phantom was imaged in a medical MRI scanner using a standard breast imaging MR sequence. Third, the MR images were converted into a 3D CAD model using commercial software (ScanIP, Simpleware), which was input into another commercial package (COMSOL Multiphysics) for RFA simulation and treatment planning using a finite element method (FEM). For validation of the model, the breast phantom was experimentally ablated using a commercial (RITA) RFA electrode and a bipolar needle with an electrosurgical generator (DRE ASG-300). The RFA results obtained by pre-treatment simulation were compared with actual experimental ablation.
Results
A 3D CAD model, created from MR images of the complex breast phantom, was successfully integrated with an RFA electrode to perform FEM ablation simulation. The ablation volumes achieved both in the FEM simulation and the experimental test were equivalent, indicating that patient-specific models can be implemented for pre-treatment planning of solid tumor ablation.
Conclusion
A tissue-mimicking breast gel phantom and its MR images were used to perform FEM 3D modeling and validation by experimental thermal ablation of the tumor. Similar patient-specific models can be created from preoperative images and used to perform finite element analysis to plan radiofrequency ablation. Clinically, the method can be implemented for pre-treatment planning to predict the effect of an individual’s tissue environment on the ablation process, and this may improve the therapeutic efficacy.
Article PDF
Similar content being viewed by others
References
Wood BJ, Ramkaransingh JR, Fojo T, Walther MM, Libutti SK (2002) Percutaneous tumor ablation with radiofrequency. Cancer 94: 43–451
Lau WY, Leung TW, Yu SC, Ho SK (2003) Percutaneous local ablative therapy for hepatocellular carcinoma: a review and look into the future. Ann Surg 237: 171–179
Morrow M (2009) Minimally invasive surgery for breast cancer. Br Med J 338
Fornage BD, Sneige N, Ross MI, Mirza AN, Kuerer HM, Edeiken BS, Ames FC, Newman LA, Babiera GV, Singletary SE (2004) Small ( < or = 2-cm) breast cancer treated with US-guided radiofrequency ablation: feasibility study. Radiology 231: 215–224
Okhunov Z, Roy O, Duty B, Waingankar N, Herati A, Morgenstern N, Sheikh-Fayyaz S, Kavoussi LR (2012) Clinical evaluation of a novel bipolar radiofrequency ablation system for renal masses. BJU Int
Payne S, Flanagan R, Pollari M, Alhonnoro T, Bost C, O’Neill D, Peng T, Stiegler P (2011) Image-based multi-scale modelling and validation of radio-frequency ablation in liver tumours. Philos Trans R Soc A Math Phys Eng Sci 369: 4233–4254
dos Santos I, Haemmerich D, Pinheiro C, da Rocha A (2008) Effect of variable heat transfer coefficient on tissue temperature next to a large vessel during radiofrequency tumor ablation. BioMed Eng OnLine 7: 21
Rieder C, Kroeger T, Schumann C, Hahn HK (2011) GPU-based real-time approximation of the ablation zone for radiofrequency ablation. IEEE Trans Vis Comput Graph 17: 1812–1821
Tungjitkusolmun S, Staelin ST, Haemmerich D, Jang-Zern T, Hong C, Webster JG, Lee FT Jr, Mahvi DM, Vorperian VR (2002) Three-dimensional finite-element analyses for radio- frequency hepatic tumor ablation. IEEE Trans Biomed Eng 49: 3–9
Barauskas R, Gulbinas A, Vanagas T, Barauskas G (2008) Finite element modeling of cooled-tip probe radiofrequency ablation processes in liver tissue. Comput Biol Med 38: 694–708
Datta A, Bansal V, Diaz J, Patel J, Reato D, Bikson M (2009) Gyri-precise head model of transcranial direct current stimulation: improved spatial focality using a ring electrode versus conventional rectangular pad. Brain Stimul 2:201–207, e201
Fuentes D, Cardan R, Stafford RJ, Yung J, Dodd GD 3rd, Feng Y (2010) High-fidelity computer models for prospective treatment planning of radiofrequency ablation with in vitro experimental correlation. J Vasc Interv Radiol 21: 1725–1732
Bu-Lin Z, Bing H, Sheng-Li K, Huang Y, Rong W, Jia L (2008) A polyacrylamide gel phantom for radiofrequency ablation. Int J Hyperthermia 24: 568–576
Rossi MA, Stebbins G, Murphy C, Greene D, Brinker S, Sarcu D, Tenharmsel A, Stoub T, Stein MA, Hoeppner TJ, Byrne RW, Moseley ME, Bammer RA, Bild S, Dennis J, Arnett N, Balabanov A, Bergen D, Kanner AM, Smith MC (2010) Predicting white matter targets for direct neurostimulation therapy. Epilepsy Res 91: 176–186
Haemmerich D, Webster JG (2005) Automatic control of finite element models for temperature-controlled radiofrequency ablation. Biomed Eng Online 4: 42
Ritz J-P, Lehmann KS, Isbert C, Reissfelder C, Albrecht T, Stein T, Buhr HJ (2006) In-vivo evaluation of a novel bipolar radiofrequency device for interstitial thermotherapy of liver tumors during normal and interrupted hepatic perfusion. J Surg Res 133: 176–184
Duck FA (1990) Physical properties of tissue: a comprehensive reference book. Academic Press, London
Butz T, Warfield S, Tuncali K, Silverman S, van Sonnenberg E, Jolesz F, Kikinis R (2000) Pre- and intra-operative planning and simulation of percutaneous tumor ablation. In: Delp S, DiGoia A, Jaramaz B (eds) Medical image computing and computer-assisted intervention—MICCAI 2000. Springer, Berlin, pp 395–416
Häcker A, Vallo S, Weiss C, Grobholz R, Stein T, Knoll T, Michel MS (2006) Bipolar and multipolar radio frequency ablation with resistance controlled power output: standardized ex vivo kidney tissue evaluation. J Urol 175: 1122–1126
Stoffner R, Kremser C, Schullian P, Haidu M, Widmann G, Bale RJ (2012) Multipolar radiofrequency ablation using 4–6 applicators simultaneously: a study in the ex vivo bovine liver. Eur J Radiol
Patterson EJ, Scudamore CH, Owen DA, Nagy AG, Buczkowski AK (1998) Radiofrequency ablation of porcine liver in vivo: effects of blood flow and treatment time on lesion size. Ann Surg 227: 559–565
Zlotta AR, Wildschutz T, Raviv G, Peny MO, van Gansbeke D, Noel JC, Schulman CC (1997) Radiofrequency interstitial tumor ablation (RITA) is a possible new modality for treatment of renal cancer: ex vivo and in vivo experience. J Endourol 11: 251–258
Cha J, Choi D, Lee MW, Rhim H, Kim YS, Lim HK, Yoon JH, Park CK (2009) Radiofrequency ablation zones in ex vivo bovine and in vivo porcine livers: comparison of the use of internally cooled electrodes and internally cooled wet electrodes. Cardiovasc Intervent Radiol 32: 1235–1240
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Wang, Z., Aarya, I., Gueorguieva, M. et al. Image-based 3D modeling and validation of radiofrequency interstitial tumor ablation using a tissue-mimicking breast phantom. Int J CARS 7, 941–948 (2012). https://doi.org/10.1007/s11548-012-0769-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-012-0769-3