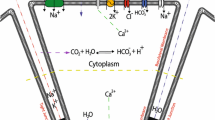
Abstract
Saliva is secreted from the acinar cells of the salivary glands, using mechanisms that are similar to other types of water-transporting epithelial cells. Using a combination of theoretical and experimental techniques, over the past 20 years we have continually developed and modified a quantitative model of saliva secretion, and how it is controlled by the dynamics of intracellular calcium. However, over approximately the past 5 years there have been significant developments both in our understanding of the underlying mechanisms and in the way these mechanisms should best be modelled. Here, we review the traditional understanding of how saliva is secreted, and describe how our work has suggested important modifications to this traditional view. We end with a brief description of the most recent data from living animals and discuss how this is now contributing to yet another iteration of model construction and experimental investigation.












Similar content being viewed by others
References
Almássy J, Won JH, Begenisich TB, Yule DI (2012) Apical Ca\(^{2+}\)-activated potassium channels in mouse parotid acinar cells. J Gen Physiol 139(2):121–33
Almássy J, Siguenza E, Skaliczki M, Matesz K, Sneyd J, Yule DI, Nánási PP (2018) New saliva secretion model based on the expression of Na\(^+\)-K\(^+\) pump and K\(^+\) channels in the apical membrane of parotid acinar cells. Pflugers Arch 470(4):613–621. https://doi.org/10.1007/s00424-018-2109-0
Ambudkar IS (2012) Polarization of calcium signaling and fluid secretion in salivary gland cells. Curr Med Chem 19(34):5774–81
Ambudkar IS (2014) Ca\(^{2+}\) signaling and regulation of fluid secretion in salivary gland acinar cells. Cell Calcium 55(6):297–305. https://doi.org/10.1016/j.ceca.2014.02.009
Baum BJ, Zheng C, Cotrim AP, Goldsmith CM, Atkinson JC, Brahim JS, Chiorini JA, Voutetakis A, Leakan RA, Van Waes C, Mitchell JB, Delporte C, Wang S, Kaminsky SM, Illei GG (2006) Transfer of the AQP1 cDNA for the correction of radiation-induced salivary hypofunction. Biochim Biophys Acta 1758(8):1071–7. https://doi.org/10.1016/j.bbamem.2005.11.006
Baum BJ, Adriaansen J, Cotrim AP, Goldsmith CM, Perez P, Qi S, Rowzee AM, Zheng C (2010) Gene therapy of salivary diseases. Methods Mol Biol 666:3–20. https://doi.org/10.1007/978-1-60761-820-1
Baum BJ, Alevizos I, Chiorini JA, Cotrim AP, Zheng C (2015) Advances in salivary gland gene therapy—oral and systemic implications. Expert Opin Biol Ther 15(10):1443–54. https://doi.org/10.1517/14712598.2015.1064894
Baum BJ, Afione S, Chiorini JA, Cotrim AP, Goldsmith CM, Zheng C (2017) Gene therapy of salivary diseases. Methods Mol Biol 1537:107–123. https://doi.org/10.1007/978-1-4939-6685-1
Berridge MJ (1990) Calcium oscillations. J Biol Chem 265:9583–86
Berridge MJ (2009) Inositol trisphosphate and calcium signalling mechanisms. Biochim Biophys Acta 1793(6):933–40
Berridge MJ, Galione A (1988) Cytosolic calcium oscillators. FASEB J 2:3074–3082
Berridge MJ, Lipp P, Bootman MD (2000) The versatility and universality of calcium signalling. Nat Rev Mol Cell Biol 1(1):11–21
Cao P, Donovan G, Falcke M, Sneyd J (2013) A stochastic model of calcium puffs based on single-channel data. Biophys J 105(5):1133–42. https://doi.org/10.1016/j.bpj.2013.07.034
Chara O, Brusch L (2015) Mathematical modelling of fluid transport and its regulation at multiple scales. Biosystems 130:1–10. https://doi.org/10.1016/j.biosystems.2015.02.004
Cook D, Van Lennep E, Ml R, Ja Y (1994) Secretion by the major salivary glands. In: Johnson L (ed) Physiology of the gastrointestinal tract, 3rd edn. Raven Press, New York, pp 1061–2017
Cuthbertson KSR (1989) Intracellular calcium oscillators. In: Goldbeter A (ed) Cell to cell signalling: from experiments to theoretical models. Academic Press, London, pp 435–447
Daniels TE, Wu AJ (2000) Xerostomia—clinical evaluation and treatment in general practice. J Calif Dent Assoc 28(12):933–41
Dawson D (1992) Water transport principles and perspectives. In: Seldin D, Giebisch G (eds) The kidney physiology and pathophysiology. Raven Press, New York, pp 301–316
Diamond JM, Bossert WH (1967) Standing-gradient osmotic flow. A mechanism for coupling of water and solute transport in epithelia. J Gen Physiol 50(8):2061–83
Dolmetsch RE, Xu K, Lewis RS (1998) Calcium oscillations increase the efficiency and specificity of gene expression. Nature 392(6679):933–6. https://doi.org/10.1038/31960
Dupont G, Combettes L, Bird GS, Putney JW (2011) Calcium oscillations. Cold Spring Harb Perspect Biol 3(3):a004226. https://doi.org/10.1101/cshperspect.a004226
Dupont G, Falcke M, Kirk V, Sneyd J (2016) Models of calcium signalling, interdisciplinary applied mathematics, vol 43. Springer, Berlin. https://doi.org/10.1007/978-3-319-29647-0
Ermentrout G, Kopell N (1984) Frequency plateaus in a chain of weakly coupled oscillators. SIAM J Math Anal 15:215–37
Evans RL, Park K, Turner RJ, Watson GE, Nguyen HV, Dennett MR, Hand AR, Flagella M, Shull GE, Melvin JE (2000) Severe impairment of salivation in Na\(^+\)/K\(^+\)/2Cl\(^-\) cotransporter (NKCC1)-deficient mice. J Biol Chem 275(35):26720–26726. https://doi.org/10.1074/jbc.M003753200
Fong S, Chiorini JA, Sneyd J, Suresh V (2017) Computational modeling of epithelial fluid and ion transport in the parotid duct after transfection of human aquaporin-1. Am J Physiol Gastrointest Liver Physiol 312(2):G153–G163. https://doi.org/10.1152/ajpgi.00374.2016
Foskett JK, Melvin JE (1989) Activation of salivary secretion: coupling of cell volume and [Ca\(^{2+}\)]\(_i\) in single cells. Science 244(4912):1582–5
Fox RI, Maruyama T (1997) Pathogenesis and treatment of Sjögren’s syndrome. Curr Opin Rheumatol 9(5):393–9
Gao R, Yan X, Zheng C, Goldsmith CM, Afione S, Hai B, Xu J, Zhou J, Zhang C, Chiorini JA, Baum BJ, Wang S (2010) AAV2-mediated transfer of the human aquaporin-1 cDNA restores fluid secretion from irradiated miniature pig parotid glands. Gene Ther 18(1):38–42
Garcia GJM, Boucher RC, Elston TC (2013) Biophysical model of ion transport across human respiratory epithelia allows quantification of ion permeabilities. Biophys J 104(3):716–26. https://doi.org/10.1016/j.bpj.2012.12.040
Gin E, Crampin EJ, Brown DA, Shuttleworth TJ, Yule DI, Sneyd J (2007) A mathematical model of fluid secretion from a parotid acinar cell. J Theor Biol 248(1):64–80
Gu X, Spitzer NC (1995) Distinct aspects of neuronal differentiation encoded by frequency of spontaneous Ca\(^{2+}\) transients. Nature 375(6534):784–7. https://doi.org/10.1038/375784a0
Hill AE (2008) Fluid transport: a guide for the perplexed. J Membr Biol 223(1):1–11. https://doi.org/10.1007/s00232-007-9085-1
Hong JH, Park S, Shcheynikov N, Muallem S (2014) Mechanism and synergism in epithelial fluid and electrolyte secretion. Pflugers Arch 466(8):1487–99. https://doi.org/10.1007/s00424-013-1390-1
Kasai H, Li YX, Miyashita Y (1993) Subcellular distribution of \({\rm Ca}^{2+}\) release channels underlying \({\rm Ca}^{2+}\) waves and oscillations in exocrine pancreas. Cell 74:669–77
Keener J, Sneyd J (2008) Mathematical physiology, 2nd edn. Springer, New York
Kopell N, Ermentrout G (1986) Symmetry and phaselocking in chains of weakly coupled oscillators. Commun Pure Appl Math 39(5):623–60
Kopell N, Ermentrout G (1990) Phase transitions and other phenomena in chains of coupled oscillators. SIAM J Appl Math 50(4):1014–52
Kuramoto Y (2003) Chemical oscillations, waves, and turbulence. Courier Corporation, New York
Larsen EH (2002) Analysis of the sodium recirculation theory of solute-coupled water transport in small intestine. J Physiol 542(1):33–50
Larsen EH, Sørensen JB, Sørensen JN (2000) A mathematical model of solute coupled water transport in toad intestine incorporating recirculation of the actively transported solute. J Gen Physiol 116(2):101–24. https://doi.org/10.1085/jgp.116.2.101
Layton AT (2011a) A mathematical model of the urine concentrating mechanism in the rat renal medulla. I. Formulation and base-case results. Am J Physiol Renal Physiol 300(2):F356–F371. https://doi.org/10.1152/ajprenal.00203.2010
Layton AT (2011b) A mathematical model of the urine concentrating mechanism in the rat renal medulla. II. Functional implications of three-dimensional architecture. Am J Physiol Renal Physiol 300(2):F372–F384. https://doi.org/10.1152/ajprenal.00204.2010
Layton AT, Layton HE (2005) A region-based mathematical model of the urine concentrating mechanism in the rat outer medulla. I. Formulation and base-case results. Am J Physiol Renal Physiol 289(6):F1346–F1366. https://doi.org/10.1152/ajprenal.00346.2003
Layton AT, Layton HE (2005) A region-based mathematical model of the urine concentrating mechanism in the rat outer medulla. II. Parameter sensitivity and tubular inhomogeneity. Am J Physiol Renal Physiol 289(6):F1367–F1381. https://doi.org/10.1152/ajprenal.00347.2003
Lee MG, Xu X, Zeng W, Diaz J, Wojcikiewicz RJ, Kuo TH, Wuytack F, Racymaekers L, Muallem S (1997) Polarized expression of Ca\(^{2+}\) channels in pancreatic and salivary gland cells Correlation with initiation and propagation of [Ca\(^{2+}]_i\) waves. J Biol Chem 272(25):15765–15770
Lee MG, Ohana E, Park HW, Yang D, Muallem S (2012) Molecular mechanism of pancreatic and salivary gland fluid and HCO\(_3\) secretion. Physiol Rev 92(1):39–74. https://doi.org/10.1152/physrev.00011.2011
Li W, Llopis J, Whitney M, Zlokarnik G, Tsien RY (1998) Cell-permeant caged InsP\(_3\) ester shows that Ca\(^{2+}\) spike frequency can optimize gene expression. Nature 392(6679):936–41. https://doi.org/10.1038/31965
Lundberg A (1956) Secretory potentials and secretion in the sublingual gland of the cat. Nature 177(4519):1080–1. https://doi.org/10.1038/1771080a0
Lundberg A (1957a) The mechanism of establishment of secretory potentials in sublingual gland cells. Acta Physiol Scand 40(1):35–58. https://doi.org/10.1111/j.1748-1716.1957.tb01476.x
Lundberg A (1957b) Secretory potentials in the sublingual gland of the cat. Acta Physiol Scand 40(1):21–34. https://doi.org/10.1111/j.1748-1716.1957.tb01475.x
Maclaren OJ, Sneyd J, Crampin EJ (2012) Efficiency of primary saliva secretion: an analysis of parameter dependence in dynamic single-cell and acinus models, with application to aquaporin knockout studies. J Membr Biol 245(1):29–50. https://doi.org/10.1007/s00232-011-9413-3
Mangos JA, McSherry NR, Irwin K, Hong R (1973a) Handling of water and electrolytes by rabbit parotid and submaxillary glands. Am J Physiol 225(2):450–5. https://doi.org/10.1152/ajplegacy.1973.225.2.450
Mangos JA, McSherry NR, Nousia-Arvanitakis S, Irwin K (1973b) Secretion and transductal fluxes of ions in exocrine glands of the mouse. Am J Physiol 225(1):18–24. https://doi.org/10.1152/ajplegacy.1973.225.1.18
Martinez JR (1990) Cellular mechanisms underlying the production of primary secretory fluid in salivary glands. Crit Rev Oral Biol Med 1(1):67–78
Mathias RT, Wang H (2005) Local osmosis and isotonic transport. J Membr Biol 208(1):39–53
Melvin JE (1991) Saliva and dental diseases. Curr Opin Dent 1(6):795–801
Melvin JE (1999) Chloride channels and salivary gland function. Crit Rev Oral Biol Med 10(2):199–209
Melvin JE, Yule D, Shuttleworth T, Begenisich T (2005) Regulation of fluid and electrolyte secretion in salivary gland acinar cells. Annu Rev Physiol 67(1):445–469
Mirollo RE, Strogatz SH (1990) Synchronization of pulse-coupled biological oscillators. SIAM J Appl Math 50(6):1645–1662
Nash MS, Schell MJ, Atkinson PJ, Johnston NR, Nahorski SR, Challiss RAJ (2002) Determinants of metabotropic glutamate receptor-5-mediated Ca\(^{2+}\) and inositol 1,4,5-trisphosphate oscillation frequency. Receptor density versus agonist concentration. J Biol Chem 277(39):35947–35960. https://doi.org/10.1074/jbc.M205622200
Nathanson MH, Fallon MB, Padfield PJ, Maranto AR (1994) Localization of the type 3 inositol 1,4,5-trisphosphate receptor in the Ca\(^{2+}\) wave trigger zone of pancreatic acinar cells. J Biol Chem 269(7):4693–6
Nauntofte B (1992) Regulation of electrolyte and fluid secretion in salivary acinar cells. Am J Physiol 263(6 Pt 1):G823–37
Neu J (1979) Chemical waves and the diffusive coupling of limit cycle oscillators. SIAM J Appl Math 36:509–515
Novotny JA, Jakobsson E (1996a) Computational studies of ion-water flux coupling in the airway epithelium. I. Construction of model. Am J Physiol 270(6 Pt 1):C1751–C1763
Novotny JA, Jakobsson E (1996b) Computational studies of ion-water flux coupling in the airway epithelium. II. Role of specific transport mechanisms. Am J Physiol 270(6 Pt 1):C1764–C1772
Pages N, Vera-Sigüenza E, Rugis J, Kirk V, Yule DI, Sneyd J (2019) A model of Ca\(^{2+}\) dynamics in an accurate reconstruction of parotid acinar cells. Bull Math Biol 81(5):1394–1426. https://doi.org/10.1007/s11538-018-00563-z
Palk L, Sneyd J, Shuttleworth TJ, Yule DI, Crampin EJ (2010) A dynamic model of saliva secretion. J Theor Biol 266(4):625–40
Palk L, Sneyd J, Patterson K, Shuttleworth TJ, Yule DI, Maclaren O, Crampin EJ (2012) Modelling the effects of calcium waves and oscillations on saliva secretion. J Theor Biol 305:45–53
Patterson K, Catalán MA, Melvin JE, Yule DI, Crampin EJ, Sneyd J (2012) A quantitative analysis of electrolyte exchange in the salivary duct. Am J Physiol Gastrointest Liver Physiol 303(10):G1153–63. https://doi.org/10.1152/ajpgi.00364.2011
Peña-Münzenmayer G, Catalán MA, Kondo Y, Jaramillo Y, Liu F, Shull GE, Melvin JE (2015) Ae4 (Slc4a9) anion exchanger drives Cl-uptake-dependent fluid secretion by mouse submandibular gland acinar cells. J Biol Chem 290(17):10677–10688. https://doi.org/10.1074/jbc.M114.612895
Reuss L (2002) Water transport controversies—an overview. J Physiol 542(1):1–2
Sandefur CI, Boucher RC, Elston TC (2017) Mathematical model reveals role of nucleotide signaling in airway surface liquid homeostasis and its dysregulation in cystic fibrosis. Proc Natl Acad Sci USA 114(35):E7272–E7281. https://doi.org/10.1073/pnas.1617383114
Ship JA, Fox PC, Baum BJ (1991) How much saliva is enough? “Normal” function defined. J Am Dent Assoc 122(3):63–9
Siekmann I, Wagner LE, Yule D, Crampin EJ, Sneyd J (2012) A kinetic model for type I and II IP\(_3\)R accounting for mode changes. Biophys J 103(4):658–68
Silva P, Stoff J, Field M, Fine L, Forrest JN, Epstein FH (1977) Mechanism of active chloride secretion by shark rectal gland: role of Na-K-ATPase in chloride transport. Am J Physiol 233(4):F298–306. https://doi.org/10.1152/ajprenal.1977.233.4.F298
Sneyd J, Tsaneva-Atanasova K, Bruce JIE, Straub SV, Giovannucci DR, Yule DI (2003) A model of calcium waves in pancreatic and parotid acinar cells. Biophys J 85(3):1392–405. https://doi.org/10.1016/S0006-3495(03)74572-X
Sneyd J, Tsaneva-Atanasova K, Reznikov V, Bai Y, Sanderson MJ, Yule DI (2006) A method for determining the dependence of calcium oscillations on inositol trisphosphate oscillations. Proc Natl Acad Sci USA 103(6):1675–80. https://doi.org/10.1073/pnas.0506135103
Sneyd J, Crampin E, Yule D (2014) Multiscale modelling of saliva secretion. Math Biosci 257:69–79. https://doi.org/10.1016/j.mbs.2014.06.017
Sneyd J, Han JM, Wang L, Chen J, Yang X, Tanimura A, Sanderson MJ, Kirk V, Yule DI (2017) On the dynamical structure of calcium oscillations. Proc Natl Acad Sci USA 114(7):1456–1461. https://doi.org/10.1073/pnas.1614613114
Swanson CH (1977) Isotonic water transport in secretory epithelia. Yale J Biol Med 50(2):153–63
Thorn P (1996) Spatial domains of Ca\(^{2+}\) signaling in secretory epithelial cells. Cell Calcium 20:203–214
Thorn P, Lawrie AM, Smith PM, Gallacher DV, Petersen OH (1993) Local and global cytosolic Ca\(^{2+}\) oscillations in exocrine cells evoked by agonists and inositol trisphosphate. Cell 74(4):661–8
Thul R, Bellamy TC, Roderick HL, Bootman MD, Coombes S (2008) Calcium oscillations. Adv Exp Med Biol 641:1–27
Torre V (1975) Synchronization of non-linear biochemical oscillators coupled by diffusion. Biol Cybern 17:137–144
Tosteson D, Hoffman J (1960) Regulation of cell volume by active cation transport in high and low potassium sheep red cells. J Gen Physiol 44:169–94
Turner RJ, Sugiya H (2002) Understanding salivary fluid and protein secretion. Oral Dis 8(1):3–11
Vera-Sigüenza E, Catalán MA, Peña-Münzenmayer G, Melvin JE, Sneyd J (2018) A mathematical model supports a key role for Ae4 (Slc4a9) in salivary gland secretion. Bull Math Biol 80(2):255–282. https://doi.org/10.1007/s11538-017-0370-6
Vera-Sigüenza E, Pages N, Rugis J, Yule DI, Sneyd J (2019) A mathematical model of fluid transport in an accurate reconstruction of parotid acinar cells. Bull Math Biol 81(3):699–721. https://doi.org/10.1007/s11538-018-0534-z
Vera-Sigüenza E, Pages N, Rugis J, Yule DI, Sneyd J (2020) A multicellular model of primary saliva secretion in the parotid gland. Bull Math Biol 82(3):38. https://doi.org/10.1007/s11538-020-00712-3
Warren NJ, Tawhai MH, Crampin EJ (2009) A mathematical model of calcium-induced fluid secretion in airway epithelium. J Theor Biol 259(4):837–849
Watt SD, Gu X, Smith RD, Spitzer NC (2000) Specific frequencies of spontaneous Ca\(^{2+}\) transients upregulate GAD 67 transcripts in embryonic spinal neurons. Mol Cell Neurosci 16(4):376–87. https://doi.org/10.1006/mcne.2000.0871
Weinstein A (1994) Mathematical models of tubular transport. Ann Rev Physiol 56:691–709
Weinstein AM (1999) Modeling epithelial cell homeostasis: steady-state analysis. Bull Math Biol 61(6):1065–1091
Weinstein AM (2020) A mathematical model of the rat kidney. Antidiuresis. Am J Physiol Renal Physiol II. https://doi.org/10.1152/ajprenal.00046.2020
Weinstein AM, Stephenson JL (1979) Electrolyte transport across a simple epithelium. Steady-state and transient analysis. Biophys J 27(2):165–86. https://doi.org/10.1016/S0006-3495(79)85209-1
Weinstein AM, Stephenson JL (1981a) Coupled water transport in standing gradient models of the lateral intercellular space. Biophys J 35(1):167–191
Weinstein AM, Stephenson JL (1981b) Models of coupled salt and water transport across leaky epithelia. J Membr Biol 60(1):1–20
Wu D, Boucher RC, Button B, Elston T, Lin CL (2018) An integrated mathematical epithelial cell model for airway surface liquid regulation by mechanical forces. J Theor Biol 438:34–45. https://doi.org/10.1016/j.jtbi.2017.11.010
Acknowledgements
Since this review appears in a special volume to mark the 90th birthday of Jim Murray, it would be remiss of me (J.S.) not to acknowledge the enormous debt I owe to Jim. Jim has been, for my entire career, an inspiration to me. Not just to me, of course; Jim has been a giant of mathematical biology for decades now, and I am only one of the many who has read, and reread, his book “Mathematical Biology” for both duty and pleasure. It is absolutely fitting that this review, dedicated as it is to a series of results in which it is not always easy to decide whether the modelling stimulated the experiment, or vice versa, appears in a volume dedicated to Jim. I suspect (and hope) he will like this approach, similar as it is to his own. He is, after all, the shoulders I stand on. And, even then, I’m not entirely certain I see any further than he does. This work was supported by NIH Grant 2R01DE019245, and by the Marsden Fund of the Royal Society of New Zealand.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sneyd, J., Vera-Sigüenza, E., Rugis, J. et al. Calcium Dynamics and Water Transport in Salivary Acinar Cells. Bull Math Biol 83, 31 (2021). https://doi.org/10.1007/s11538-020-00841-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-020-00841-9