Abstract
In this paper, we develop a sharp interface tumor growth model to study the effect of the tumor microenvironment using a complex far-field geometry that mimics a heterogeneous distribution of vasculature. Together with different nutrient uptake rates inside and outside the tumor, this introduces variability in spatial diffusion gradients. Linear stability analysis suggests that the uptake rate in the tumor microenvironment, together with chemotaxis, may induce unstable growth, especially when the nutrient gradients are large. We investigate the fully nonlinear dynamics using a spectrally accurate boundary integral method. Our nonlinear simulations reveal that vascular heterogeneity plays an important role in the development of morphological instabilities that range from fingering and chain-like morphologies to compact, plate-like shapes in two dimensions.












Similar content being viewed by others
References
Alfonso JCL, Talkenberger K, Seifert M, Klink B, Hawkins-Daarud A, Swanson KR, Hatzikirou H, Deutsch A (2017) The biology and mathematical modelling of glioma invasion: a review. J R Soc Interface 14(136):20170490
Araujo RP, McElwain DLS (2004) A history of the study of solid tumour growth: the contribution of mathematical modelling. Bull Math Biol 66(5):1039–1091
Baker GR, Shelley MJ (1990) On the connection between thin vortex layers and vortex sheets. J Fluid Mech 215:161–194
Bellomo N, de Angelis E (2008) Selected topics in cancer modeling: genesis, evolution, immune competition, and therapy. Springer, New York
Byrne HM (2010) Dissecting cancer through mathematics: from the cell to the animal model. Nat Rev Cancer 10(3):221
Byrne HM (2012) Mathematical biomedicine and modeling avascular tumor growth
Cristini V, Li X, Lowengrub JS, Wise SM (2009) Nonlinear simulations of solid tumor growth using a mixture model: Invasion and branching. J Math Biol 4–5(06):723–763
Cristini V, Lowengrub J (2010) Multiscale modeling of cancer: an integrated experimental and mathematical modeling approach. Cambridge University Press, Cambridge
Cristini V, Koay E, Wang Z (2017) An introduction to physical oncology. How mechanistic mathematical modeling can improve cancer therapy outcomes. CRC Press, Boca Raton
Cristini V, Lowengrub J, Nie Q (2003) Nonlinear simulation of tumor growth. J Math Biol 46(3):191–224
Cristini V, Frieboes HB, Gatenby R, Caserta S, Ferrari M, Sinek J (2005) Morphologic instability and cancer invasion. Clin Cancer Res 11(19):6772–6779
Escher J, Matioc A-V (2013) Analysis of a two-phase model describing the growth of solid tumors. Eur J Appl Math 24(1):25–48
Fasano A, Bertuzzi A, Gandolfi A (2006) Mathematical modelling of tumour growth and treatment. In: Complex systems in biomedicine, pp 71–108. Springer
Friedman A, Hu B (2007) Bifurcation for a free boundary problem modeling tumor growth by stokes equation. SIAM J Math Anal 39(1):174–194. https://doi.org/10.1137/060656292
Friedman A, Bei H (2006) Bifurcation from stability to instability for a free boundary problem arising in a tumor model. Arch Ration Mech Anal 180(2):293–330
Friedman A, Bei H (2007) Bifurcation from stability to instability for a free boundary problem modeling tumor growth by stokes equation. J Math Anal Appl 327(1):643–664
Friedman A, Bei H (2008) Stability and instability of liapunov-schmidt and hopf bifurcation for a free boundary problem arising in a tumor model. Trans Am Math Soc 360(10):5291–5342
Friedman A, Reitich F (2001a) On the existence of spatially patterned dormant malignancies in a model for the growth of non-necrotic vascular tumors. Math Models Methods Appl Sci 11(04):601–625
Friedman A, Reitich F (2001b) Symmetry-breaking bifurcation of analytic solutions to free boundary problems: an application to a model of tumor growth. Trans Am Math Soc 353(4):1587–1634
Fritz M, Lima EABF, Nikolić V, Tinsley Oden J, Wohlmuth B (2019) Local and nonlocal phase-field models of tumor growth and invasion due to ecm degradation. arXiv preprint arXiv:1906.07788
Garcke H, Lam KF, Sitka E, Styles V (2016) A cahn-hilliard-darcy model for tumour growth with chemotaxis and active transport. Math Models Methods Appl Sci 26(06):1095–1148
Garcke H, Lam KF, Nürnberg R, Sitka E (2018) A multiphase cahn-hilliard-darcy model for tumour growth with necrosis. Math Models Methods Appl Sci 28(03):525–577
Greenspan HP (1976) On the growth and stability of cell cultures and solid tumors. J Theor Biol 56(1):229–242
Hao W, Bei H, Li S, Song L (2018) Convergence of boundary integral method for a free boundary system. J Comput Appl Math 334:128–157. https://doi.org/10.1016/j.cam.2017.11.016
Hou TY, Lowengrub JS, Shelley MJ (1994) Removing the stiffness from interfacial flows with surface tension. J Comput Phys 114(2):312–338
Jarrett AM, Lima EABF 2nd, Hormuth DA, McKenna MT, Feng X, Ekrut DA, Resende ACM, Brock A, Yankeelov TE (2018) Mathematical models of tumor cell proliferation: a review of the literature. Expert Rev Anticancer Ther 18:1271–1286
Jou HJ, Leo PH, Lowengrub JS (1997) Microstructural evolution in inhomogeneous elastic media. J Comput Phys 131(1):109–148
Kim Y, Othmer HG (2015) Hybrid models of cell and tissue dynamics in tumor growth. Math Biosci Eng 12:1141–1156
Krasny R (1986) A study of singularity formation in a vortex sheet by the point-vortex approximation. J Fluid Mech 167:65–93
Kress R (1995) On the numerical solution of a hypersingular integral equation in scattering theory. J Comput Appl Math 61(3):345–360
Kress R (2013) Linear integral equations, vol 82. Springer, New York
Li S, Li X (2011) A boundary integral method for computing the dynamics of an epitaxial island. SIAM J Sci Comput 33(6):3282–3302
Li X, Lowengrub J, Rätz A, Voigt A (2009) Solving pdes in complex geometries: a diffuse domain approach. Commun Math Sci 7(1):81
Lowengrub JS, Frieboes HB, Jin F, Chuang Y-L, Li X, Macklin P, Wise SM, Cristini V (2009) Nonlinear modelling of cancer: bridging the gap between cells and tumors. Nonlinearity 23(1):1
Lu M-J, Liu C, Li S (2019) Nonlinear simulation of an elastic tumor-host interface. Comput Math Biophys 7(1):25–47
Macklin P, Lowengrub J (2007) Nonlinear simulation of the effect of microenvironment on tumor growth. J Theor Biol 245(4):677–704
Mullins WW, Sekerka RF (1963) Morphological stability of a particle growing by diffusion or heat flow. J Appl Phys 34(2):323–329
Pennacchietti S, Michieli P, Galluzzo M, Mazzone M, GIordano S, Cornoglio PM (2003) Hypoxia promotes invasive growth by transcriptional activation of the met protooncogene. Cancer Cell 3:347–361
Pham Kara, Turian Emma, Liu Kai, Li Shuwang, Lowengrub John (2018) Nonlinear studies of tumor morphological stability using a two-fluid flow model. Journal of mathematical biology
Roose T, Jonathan Chapman S, Maini PK (2007) Mathematical models of avascular tumor growth. SIAM Rev 49(2):179–208
Saad Y, Schultz MH (1986) Gmres: a generalized minimal residual algorithm for solving nonsymmetric linear systems. SIAM J Sci Stat Comput 7(3):856–869
Acknowledgements
We would like to acknowledge the referee’s insightful suggestions and contributions. S. L. acknowledges the support from the National Science Foundation, Division of Mathematical Sciences Grant DMS-1720420. S. L. was also partially supported by Grant ECCS-1307625. M. L. acknowledges the F. R. Buck McMorris Summer Research support from the College of Science, IIT. C. L. is partially supported by the National Science Foundation, Division of Mathematical Sciences Grant DMS-1759536. J.L. acknowledges partial support from the NSF through Grants DMS-1714973, DMS-1719960, and DMS-1763272 and the Simons Foundation (594598QN) for a NSF-Simons Center for Multiscale Cell Fate Research. J. L. also thanks the National Institutes of Health for partial support through Grants 1U54CA217378-01A1 for a National Center in Cancer Systems Biology at UC Irvine and P30CA062203 for the Chao Family Comprehensive Cancer Center at UC Irvine.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix A: Linear Stability Analysis
The governing equations are
and
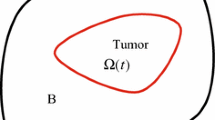
Consider a perturbed tumor interface \(\varGamma \):
In cylindrical coordinates, the modified Helmholtz equation is
Assuming axial symmetry, e.g., \(\sigma =\sigma (r, \theta )\) is independent of z, then
1.1 Radial Solutions
We first consider the radial solution, i.e., \(\sigma =\sigma (r)\), then (57) reduces to the modified Bessel differential equation
Recall the general form of modified Bessel differential equation is
The general solutions are
where \(J_{n}(x)\) is the Bessel function of the first kind, \(Y_{n}(x)\) is the Bessel function of the second kind, \(I_{n}(x)\) is a modified Bessel function of the first kind and \(K_{n}(x)\) is a modified Bessel function of the second kind.
The following recurrence relations are useful in the linear analysis
The modified Bessel functions of the second kind all have the property that
Hence, the constant \(c_2\) in Eq. (60) must be zero. We therefore obtain
Applying the boundary conditions (49), (50), (51) on the circles \(r=R,R_\infty \), we have
Solving for \(A_{1}, A_{2}, A_{3}\), we obtain
1.2 Perturbation of Radial Solutions
Now we seek a solution of the modified Helmholtz equation on the perturbed circle given by (55). Since \(\delta \) is the perturbation size, following Mullins and Sekerka (1963) we consider the Fourier expansion of the solution up to the first order in \(\delta \):
Note here that \(r, \theta \) and \(\delta \) are all functions of time t, i.e., \(r=r(t), \theta =\theta (t), \delta =\delta (t)\). Multiplying Eq. (57) by \(r^2\), we obtain
Therefore, it is sufficient to consider the expression
Apply (49), (50), (51) on the interface \(r=R+\delta e^{i l \theta }\) with \(\delta \ll 1\). Orders higher than \(O(\delta )\) are all discarded in the following calculations.
At O(1), the equations are the same as the radial solution.
The equations at \(O(\delta )\) determine the coefficients \(B_{1}, B_{2}, B_{3}:\)
Solving for \(B_{1}, B_{2}, B_{3}\), we have
The nutrient \(\sigma \) on \(\varGamma \) is given by
The normal derivative of \(\sigma \) on \(\varGamma \) is given by
where we use the identity in Eq. (6162),
Similarly, we seek a solution of Laplace equation, which reduces to Eq. (53) on the perturbed circle given by Eq. (55). It is sufficient to consider the expression
For the perturbed circle defined by Eq. (55), \(\kappa \) is given by
On the interface from Eqs. (53),(55), and (77) we obtain
It is straightforward to derive that
Thus,
The normal derivative of p is given by
Note that
Combining (78),(85),(86) and (54), we obtain
where \(C=\mu _{1} A_{1} I_{1}\left( \mu _{1} R\right) \).
Equating coefficients of like harmonics, we obtain
and
The equation of shape perturbation is given by
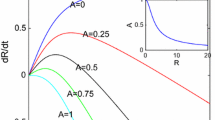
The critical apoptosis parameter \(\mathscr {A}_{c}\) is the function of R such that \(\frac{\mathrm{d}}{\mathrm{d}t}{\left( \frac{{\delta }}{R}\right) }=0\) and is given by
Appendix B: The Evaluation of the Boundary Integrals
With the integral formulation above, we assume interface curves \(\varGamma \) and \(\varGamma _{\infty }\) are analytic and given by \(\big \{\mathbf {x}(\alpha ,t)=(x(\alpha ,t),y(\alpha ,t): 0\le \alpha \le 2 \pi \big \}\), where \(\mathbf {x}\) is \(2 \pi \)-periodic in the parametrization \(\alpha \). The unit tangent and normal (outward) vectors can be calculated as \(\mathbf {s}=(x_\alpha ,y_\alpha )/s_\alpha \), \(\mathbf {n}=(y_\alpha ,-x_\alpha )/s_\alpha \), where the local variation of the arclength \(s_\alpha =\sqrt{x_\alpha ^2+y_\alpha ^2}\). Subscripts refer to partial differentiation. We track the interfaces \(\varGamma \) and \(\varGamma _{\infty }\) by introducing N marker points to discretize the planar curves, parametrized by \(\alpha _j=jh\), \(h=\frac{2\pi }{N}\), N is a power of 2. Here, we focus on the numerical evaluation of integrals following Jou et al. (1997), Li and Li (2011), Lu et al. (2019). A rigorous convergence and error analysis of the boundary integral method for a simplified tumor problem can be found in Hao et al. (2018).
Computation of the single-layer potential-type integral
In Eqs. (35), (36), (37) and (42), the single-layer potential-type integrals contain the Green functions with a logarithmic singularity at \(r=0\). They can be rewritten in the following form under the parametrization \(\alpha \)
where \(\Phi \) are the Green functions G or \(G_i\) , \(\varGamma \) may be either \(\varGamma \) or \(\varGamma _{\infty }\) and \(\phi \) may be \(\eta \) ,\(\frac{\partial \sigma _1}{\partial \mathbf {n}}\) or \(\frac{\partial \sigma _2}{\partial \mathbf {n}_\infty }\). We may decompose the Green functions as below
where \(I_{0}\) is a modified Bessel function of the first kind, \(r=|\mathbf {x}(\alpha )-\mathbf {x}'(\alpha ')|\). The square brackets on the right-hand side of Eqs. (94), (95) have removable singularity at \(\alpha =\alpha '\), since \(r= s_{\alpha }\left| \alpha -\alpha '\right| \sqrt{1+\mathcal {O}(\alpha -\alpha ')} =s_{\alpha }\left| \alpha -\alpha '\right| (1+\mathcal {O}(\alpha -\alpha '))\) for \(\alpha \approx \alpha '\), where \(\mathcal {O}{(\alpha -\alpha ')}\) denotes a smooth function that vanishes as \(\alpha \rightarrow \alpha '\), and since \(K_0\) has the expansion
Thus, for an analytic and \(2\pi \)-periodic function \(f(\alpha ,\alpha ')\), a standard trapezoidal rule or alternating point rule can be used to evaluate the integral
The remaining terms on the right-hand side of Eqs. (94), (95) have logarithmic singularity and can be evaluated through the following spectrally accurate quadrature Kress (1995)
where \(m=\frac{N}{2}\), \(\alpha _i=\frac{\pi i}{m}\) for \(i=0,1,\ldots ,2m-1\), and weight coefficients
The derivative \(\frac{d}{ds}\) in Eq. (42) is approximated using fast Fourier transform spectral derivatives, thus maintaining spectral accuracy.
Computation of the double-layer potential-type integral
In Eqs. (35), (36), (37) and (41), the double-layer potential-type integrals contain the Green functions with singularity at \(r=0\) (logarithmic for Eqs. (35), (36), (37)). They can be rewritten as in the following form under the parametrization \(\alpha \)
where \(\Phi \) are the Green functions G or \(G_i\), \(\varGamma \) may be either \(\varGamma \) or \(\varGamma _{\infty }\) and \(\phi \) may be \(\eta \), \(\sigma _1\) or \(\sigma _2=1\). Further,
where the auxiliary function \(h(\alpha ,\alpha ')=\frac{(\mathbf {x}(\alpha )-\mathbf {x}(\alpha '))\cdot \mathbf {n}(\alpha ')s_\alpha (\alpha ')}{2\pi r}\) with \(r=\left| \mathbf {x}(\alpha )-\mathbf {x}(\alpha ')\right| \). Note that \(h(\alpha ,\alpha ')\sim \mathcal {O}(\alpha -\alpha ')\). Since \(\frac{\partial G}{\partial \mathbf {n}}\) has no logarithmic singularity, we may simply use the alternating point rule to evaluate it. For \(\frac{\partial G_i}{\partial \mathbf {n}}\), we decompose it as below
where \(g_1(\alpha ,\alpha ')\) and \(g_2(\alpha ,\alpha ')\) are analytic and \(2\pi \)-periodic functions with
where we have used the fact
Since \(K_1\) has the expansion
the square bracket on the right-hand side of Eq. (104) also has removable singularity at \(\alpha =\alpha '\), and thus, the integral involving \(g_2(\alpha ,\alpha ')\) can be evaluated by a standard trapezoidal rule or alternating point rule. Note that
The first term on the right-hand side of Eq. (102) is still singular and evaluated through the quadrature given in Eqs. (98), and (99).
To summarize, using Nyström discretization with the Kress quadrature rule described above, we reduce the boundary integral Eqs. (35), (36), (37) and (41) to two dense linear systems with the unknowns as the discretization of \(\eta \) , \(\sigma _1\), \(\frac{\partial \sigma _1}{\partial \mathbf {n'}}\) on \(\varGamma \) and \(\frac{\partial \sigma _2}{\partial \mathbf {n'_\infty }}\) on \(\varGamma _\infty \), which can be solved using an iterative solver, e.g., GMRES Saad and Schultz (1986).
Appendix C: The Evolution of the Interface
As indicated by Hou et al. (1994), the curvature-driven motion introduces high-order derivatives, both non-local and nonlinear, into the dynamics through the Laplace–Young condition at the interface. Explicit time integration methods thus suffer from severe stability constraints, and implicit methods are difficult to apply since the stiffness enters nonlinearly. Hou et al. resolved these difficulties by adopting the \(\theta -L\) formulation and the small-scale decomposition (SSD), which we apply here.
\(\theta -L\) formulation
This formulation helps to circumvent the problem of point clustering. Consider a point \(\mathbf {x}(\alpha ,t)=(x(\alpha ,t),y(\alpha ,t))\in \varGamma (t)\). Denote the unit tangent and normal (outward) vectors as \(\hat{\mathbf {s}}=(x_\alpha ,y_\alpha )/s_\alpha \) and \(\hat{\mathbf {n}}=(y_\alpha ,-x_\alpha )/s_\alpha \), the normal velocity and tangent velocity by \(V(\alpha ,t)=u\cdot \hat{\mathbf {n}}\) and \(T(\alpha ,t)=u\cdot \hat{\mathbf {s}}\), respectively, where \(u=\mathbf {x}_t=V \hat{\mathbf {n}}+T \hat{\mathbf {s}}\) gives the motion of \(\varGamma (t)\). The tangent angle that the planar curve \(\varGamma (t)\) forms with the horizontal axis at \(\mathbf {x}\), called \(\theta \), satisfies \(\theta =\tan ^{-1}{\frac{y_\alpha }{x_\alpha }}\). The length of one period of the curve is \(L(t)=\int _0^{2\pi }s_\alpha \mathrm{d}\alpha \), where \(s_\alpha \), the derivative of the arclength, satisfies \(s_\alpha ^2=x_\alpha ^2+y_\alpha ^2\). Differentiating these two equations in time, we obtain the following evolution equations:
Instead of using the (x, y) coordinates, \((L,\theta )\) becomes the dynamical variables. The unit tangent and normal vectors become \(\hat{\mathbf {s}}=(\cos {\theta },\sin {\theta })\), \(\hat{\mathbf {n}}=(\sin {\theta },-\cos {\theta })\).
The normal velocity V is calculated using Eq. (30). The tangent velocity T is chosen (independent of the morphology of the interface) such that the marker points are equally spaced in arclength to prevent point clustering:
It follows that \(s_\alpha \) is independent of \(\alpha \) and thus is everywhere equal to its mean:
The procedure for obtaining the initial equal arclength parametrization is presented in “Appendix B” of Baker and Shelley (1990). The idea is to solve the nonlinear equation
for \(\alpha _j\) using Newton’s method and evaluate the equal arclength marker points \(\mathbf {x}(\alpha _j)\) by interpolation in Fourier space. We may recover the interface by simply integrating:
Small-scale decomposition (SSD)
The idea of the small scale decomposition (SSD) is to extract the dominant part of the equations at small spatial scales Hou et al. (1994). To remove the stiffness, we use SSD in our problem and develop an explicit, non-stiff time integration algorithm. In Eqs. (35), (36), (37), (41) and (42), based on the analysis of the single-layer- and double-layer-type terms, the only singularity in the integrands comes from the logarithmic kernel. Following Hou et al. (1994) and noticing the curvature term in Eq. (41), one can show that at small spatial scales,
where \(\mathscr {H}(\xi )=\frac{1}{2\pi }\int _0^{2\pi }\xi '\cot {\frac{\alpha -\alpha '}{2}}\mathrm{d}\alpha '\) is the Hilbert transform for a \(2\pi \)-periodic function \(\xi \).
We rewrite Eq. (108),
where the Hilbert transform term is the dominating high-order term at small spatial scales, and \( \displaystyle N= (\kappa T-V_s)-\frac{1}{s_\alpha ^3} \mathscr {H}[\theta _{\alpha \alpha \alpha }]\) contains other lower-order terms in the evolution. This demonstrates that an explicit time-stepping method has the high-order constraint \(\displaystyle \varDelta t \le \left( \frac{h}{s_\alpha } \right) ^3\) where \(\varDelta t\) and h are the time-step and spatial grid size, respectively. This has been demonstrated numerically in the seminal work Hou et al. (1994) for a Hele-Shaw problem. For the tumor growth problem, the semi-implicit time-stepping scheme (see Eq. (115)) requires \(\varDelta t = O(h)\) instead of explicit schemes which would require \(\varDelta t = O(h^3)\). In Sect. 5.1, we show a numerical example using \(N=1024\) to simulate a twofold tumor. In this simulation, we could use \(\varDelta t\) as large as \(\varDelta t=1.0 \times 10^{-2}\) for stability instead of \(\varDelta t<10^{-6}\) for an explicit scheme with the equal arclength parametrization. For the purpose of numerical accuracy, we used a smaller time step in our simulation.
Appendix D: Semi-implicit Time-Stepping Scheme
Taking the Fourier transform of Eq. (115), we get
We solve Eq. (116) using the second-order accurate linear propagator method in the Adams–Bashforth form Hou et al. (1994) in Fourier space and apply the inverse Fourier transform to recover \(\theta \). Specifically, we discretize Eq. (116) as
where the superscript n denotes the numerical solutions at \(t=t_n\) and the integrating factor
Note that by setting the integrating factors in Eq. (117) to 1, we recover the Adams–Bashforth explicit time-stepping method. The integrating factors in Eq. (117) can be evaluated simply using the trapezoidal rule,
To compute the arclength \(s_\alpha \), equation (109) is discretized using the explicit second-order Adams–Bashforth method Hou et al. (1994),
where M is calculated using
Note that the second-order linear propagator and Adams–Bashforth methods are multi-step method and require two previous time steps. The first time step is realized using an explicit Euler method for \(s_\alpha ^1\) and a first-order linear propagator of a similar form for \(\hat{\theta }^1\).
Tumor morphologies with different symmetric far-field geometries in the nutrient-poor regime (\(D=1\)). The far-field boundaries are \(R_\infty =13+2\cos (k\theta ),k=3,4,5,6\) (Rows 1,3); \(R_\infty =13+2\cos (k\theta -\pi /2),k=3,5,~R_\infty =13+2\cos (k\theta -\pi ),k=4,6\) (Rows 2,4). The initial tumor boundaries are \(r=2.0+ 0.1\cos (2 \theta )\) (Rows 1,2); and \(\frac{x^2}{2.1^2}+\frac{y^2}{1.9^2}=1\) (Rows 3,4). The remaining parameters are \(\lambda =0.01\), \(\chi _{\sigma }=10\), \(\mathscr {P}=0.5\), \(\mathscr {A}=0\), \(\mathscr {G}^{-1}=0.001\). Here, \(N=512\), and \(\varDelta t=0.005\) (Color figure online)
To reconstruct the tumor–host interface \((x(\alpha ,t_{n+1}),y(\alpha ,t_{n+1}))\) from the updated \(\theta ^{n+1}(\alpha )\) and \(s_\alpha ^{n+1}\), we first update a reference point \((x(0,t_{n+1}),y(0,t_{n+1})\) using a second-order explicit Adams–Bashforth method to discretize the equation of motion \(\mathbf {x}_t=V\hat{\mathbf {n}}\) with the tangential part dropped since it does not change the morphology:
Once we update the reference point, we obtain the configuration of the interface from the \(\theta ^{n+1}(\alpha )\) and \(s_\alpha ^{n+1}\) by integrating Eq. (113) following Hou et al. (1994):
where the indefinite integration is performed using the discrete Fourier transform.
We use a 25th-order Fourier filter to damp the highest nonphysical mode and suppress the aliasing error Hou et al. (1994). We also use Krasny filtering Krasny (1986) to prevent the accumulation of round-off errors during the computation.
We solve first the nutrient field \(\sigma \) and then the pressure field p. Next we compute the normal velocity V and update the interface \(\varGamma (t)\) and repeat this procedure.
Appendix E
In Fig. 13, we present tumor morphologies at similar sizes under the same growth conditions but using different symmetric far-field boundaries: \(R_\infty =13+2\cos (k\theta ),k=3,4,5,6\) (Row 1,3) and \(R_\infty =13+2\cos (k\theta -\pi /2),k=3,5,R_\infty =13+2\cos (k\theta -\pi ),k=4,6\) (Row 2,4). The parameters are the same as in Fig. 7 in the main text, where \(\chi _\sigma =5\), except that here in Fig. 13 we use \(\chi _\sigma =10\). As in Fig. 7, initial tumor boundary in rows 1 and 2 is the perturbed circle \(R_\infty =2.0+ 0.1\cos (2 \theta )\) while in rows 3 and 4 the initial tumor boundary is the ellipse \(\frac{x^2}{2.1^2}+\frac{y^2}{1.9^2}=1\). Hence, by comparing the evolution within each row, and between rows 1 and 2 and rows 3 and 4, we can see the effect of the far-field boundary shapes. By comparing rows 1 and 3 and rows 2 and 4, we can see the effect of the different initial shapes.
The results are similar to those obtained in Fig. 7 although here, because the chemotaxis coefficient \(\chi _\sigma \) is increased, the tubular structures develop faster and are narrower than those observed in Fig. 7. In particular, when the initial condition is the perturbed circle (rows 1 and 2), the far-field geometry has limited influence on tumor morphologies consistent with that observed in Figs. 5 and 6. However, when the initial condition is an ellipse, which contains many modes, the morphologies are much more sensitive to the far-geometry because of the instability.
Rights and permissions
About this article
Cite this article
Lu, MJ., Liu, C., Lowengrub, J. et al. Complex Far-Field Geometries Determine the Stability of Solid Tumor Growth with Chemotaxis. Bull Math Biol 82, 39 (2020). https://doi.org/10.1007/s11538-020-00716-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-020-00716-z