Abstract
Many mathematical models for the disease transmission dynamics of Ebola have been developed and studied, particularly during and after the 2014 outbreak in West Africa. Most of these models are systems of ordinary differential equations (ODEs). One of the common assumptions made in these ODE models is that the duration of disease stages, such as latent and infectious periods, follows an exponential distribution. Gamma distributions have also been used in some of these models. It has been demonstrated that, when the models are used to evaluate disease control strategies such as quarantine or isolation, the models with exponential and Gamma distribution assumptions may generate contradictory results (Feng et al. in Bull Math Biol 69(5):1511–1536, 2007). Several Ebola models are considered in this paper with various stage distributions, including exponential, Gamma and arbitrary distributions. These models are used to evaluate control strategies such as isolation (or hospitalization) and timely burial and to identify potential discrepancies between the results from models with exponential and Gamma distributions.







Similar content being viewed by others
References
Althaus CL (2014) Estimating the reproduction number of Ebola virus (EBOV) during the 2014 outbreak in West Africa. PLoS Curr 6(1):353–357
Browne CJ, Gulbudak H, Webb G (2015) Modeling contact tracing in outbreaks with application to Ebola. J Theor Biol 384:33–49
Butler D (2014) Models overestimate Ebola cases. Nature 515(7525):18. doi:10.1038/515018a
Chowell G, Nishiura H (2014) Transmission dynamics and control of Ebola virus disease (EVD): a review. BMC Med 12(1):196
Chowell D, Castillo-Chavez C, Krishna S, Qiu X, Anderson K (2015) Modelling the effect of early detection of Ebola. Lancet Infect Dis 15(2):148–149
Feng Z, Thieme HR (2000) Endemic models with arbitrarily distributed periods of infection I: fundamental properties of the model. SIAM J Appl Math 61(3):803–833
Feng Z, Xu D, Zhao H (2007) Epidemiological models with non-exponentially distributed disease stages and applications to disease control. Bull Math Biol 69(5):1511–1536
Feng Z, Zheng Y, Heranndez-Ceron N, Zhao H, Glasser JW, Hill AN (2016) Mathematical models of Ebola-consequences of underlying assumptions. Math Biosci 277:89–107
Hernandez-Ceron N, Feng Z, Castillo-Chavez C (2013a) Discrete epidemic models with arbitrary stage distributions and applications to disease control. Bull Math Biol 75(10):1716–1746
Hernandez-Ceron N, Feng Z, van den Driessche P (2013b) Reproduction numbers for discrete-time epidemic models with arbitrary stage distributions. J Differ Equ Appl 19:1671–1693
Hethcote H, Tudor D (1980) Integral equation models for endemic infectious diseases. J Math Biol 9:37–47
Legrand J, Grais RF, Boelle PY, A Valleron AJ, Flahault A (2007) Understanding the dynamics of Ebola epidemics. Epidemiol Infect 135:610–621
Lewnard JA, Ndeffo Mbah ML, Alfaro-Murillo JA, Altice FL, Bawo L, Nyenswah TG, Galvani AP (2014) Dynamics and control of Ebola virus transmission in Montserrado, Liberia: a mathematical modelling analysis. Lancet Infect Dis 14(12):1189–1195
Lloyd A (2001) Realistic distributions of infectious periods in epidemic models. Theor. Pop. Biol. 60:59–71
MacDonald N (1978) Time Lags in Biological Models. Springer, New York
Rivers CM et al (2014) Modeling the impact of interventions on an epidemic of Ebola in Sierra Leone and Liberia. PLoS Curr 6(6):e62
Shen M, Xiao Y, Rong L (2015) Modeling the effect of comprehensive interventions on Ebola virus transmission. Sci Rep 5:15818
Towers S, Patterson-Lomba O, Castillo-Chavez C (2014) Temporal variations in the effective reproduction number of the 2014 West Africa Ebola outbreak. PLoS Curr. doi:10.1371/currents.outbreaks
Velásquez GE, Aibana O, Ling E, Diakite I, Mooring EQ, Murray MB (2015) Time from infection to disease and infectiousness for Ebola virus disease, a systematic review. Clin Infect Dis 61:1135–1140
Wearing H, Rohani P, Keeling M (2005) Appropriate models for the management of infectious diseases. PLoS Med 2(7):e174
WHO (2014) WHO Ebola Response Team. Ebola virus disease in West Africa—the first 9 months of the epidemic and forward projections. N. Engl. J. Med. 371(16):1481–1495
Acknowledgements
We thank the Reviewers for their comments and suggestions which helped to improve the presentation of the paper. The research is partially supported by the Natural Science Foundation of China (11371048) and the BUCEA Post Graduate Innovation Project (PG2017031).
Author information
Authors and Affiliations
Corresponding authors
Appendices
Appendix 1: Derivation of \(p_c\) for Model C
Recall that in Model C (see (30)), \(T_{P_c}\) and \(T_{L_c}\) follow exponential distributions with parameters \(\gamma \) and \(\chi \), respectively (for ease of notation the subscripts have been dropped), and \(T_{M_c}\) follows Gamma distribution with parameters \((\mu , q \mu )\). That is, \(P_{c}(t)=G^{1}_{\gamma }\), \(L_{c}(t)=G^{1}_{\chi }\), and \(M_{c}(t)=G^{q}_{q\mu }\) (G for Gamma with the superscript being the shape parameter). Let \(g_X(s)=-\dot{X}(s)\) for \(X=P_c, L_c\) and \(M_c\). Note that the proportion of infected individuals that are hospitalized is given by
Thus,
Appendix 2
In this appendix, we provide the detailed derivation for the reduction in the system (7) to an ODE system under the specific survival functions given in (10) and (11).
Using these functions and from the I equation in (7), we obtain
Let
for \(j=1,2,\ldots ,n\). Then from (39) and (40),
Differentiating \(I_j(t)\) in (40), we arrive at
and
It follows that
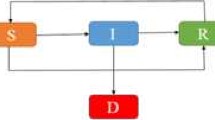
The above derivation indicates that the assumption of \(T_M\) being Gamma leads to the fact that the I class can be divided into n subclasses, denoted by \(I_j\) for \(1 \le j \le n\), while each substage j can be described by an exponential distribution with parameter \(n\mu \) (see the transition diagram Fig. 2).
Let
Using the H equation in (7) and the M function in (11), we have
It follows from (44) that
Differentiating \(H_j\) in (44) we have
and
It follows that
The derivation of the equations for \(H_j\) indicates that the H class can also be divided into n subclasses, denoted by \(H_j\) for \(1 \le j \le n\), and each substage j can be described by an exponential distribution with parameter \(n\mu \) (see the transition diagram Fig. 2).
For the D equation, we can use the density function \(g_M(s)\) in (11) to get
Differentiation of the above equation leads to
Similarly, for the R equation, note that
Then,
It follows from (40), (41), (44) and (45) that
From the differential Eqs. (42), (43), (46), (47), (48) and (49), we arrive at the ODE model (22).
When M(s) is also an exponential function, i.e., when \(n=1\), the system (22) is further reduced to the following system (which is also the special case of model (9) when \(n=1\)):
Rights and permissions
About this article
Cite this article
Wang, X., Shi, Y., Feng, Z. et al. Evaluations of Interventions Using Mathematical Models with Exponential and Non-exponential Distributions for Disease Stages: The Case of Ebola. Bull Math Biol 79, 2149–2173 (2017). https://doi.org/10.1007/s11538-017-0324-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-017-0324-z