Abstract
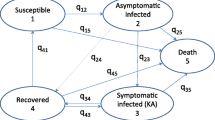
In this manuscript, we propose and analyze a compartmental model of visceral leishmaniasis (VL). We model the human population with six compartments including asymptomatic, symptomatic and PKDL-infected, animal population as second host and sandfly population as the vector. Furthermore, the non-adult stage of the sandfly population is introduced in the system, which was not considered before in the literature. We show that the increase in the number of host of sandfly population generates a backward bifurcation. Thus, multiple hosts will cause disease persistence even if the basic reproduction number (\(R_{0}\)) is below unity. We perform a sensitivity analysis of important model parameters with respect to some epidemiologically significant responses. We validate our model by calibrating it to weekly VL incidence data from South Sudan for the year 2013. We perform cost-effectiveness analysis on different interventions: treatment, non-adult control, adult control and their different layered combinations based on their implementation cost (in USD) and case reduction. We also use a global sensitivity analysis technique to understand the effect of important parameters of our model on the implementation cost of different controls. This cost-effectiveness study and cost–sensitivity analysis are relatively new in existing literature of this disease.









Similar content being viewed by others
References
Abate A, Tiwari A, Sastry S (2009) Box invariance in biologically-inspired dynamical systems. Automatica 45:1601–1610
Alexander B, Maroli M (2003) Control of phlebotomine sandflies. Med Vet Entomol 17(1):1–18
Arias JR, Monteiro PS, Zicker F (1996) The reemergence of visceral leishmaniasis in Brazil. Emerg Infect Dis 2(2):145
Ashford DA, David JR, Freire M, David R, Sherlock I, Eulálio MDC, Sampaio DP, Badaro R (1998) Studies on control of visceral leishmaniasis: impact of dog control on canine and human visceral leishmaniasis in Jacobina, Bahia, Brazil. Am J Trop Med Hyg 59(1):53–57
Biodiversity and infectious diseases (2016). http://www.chgeharvard.org/topic/ [Online Accessed 28/4/2016]
Boggiatto PM, Gibson-Corley KN, Metz K, Gallup JM, Hostetter JM, Mullin K, Petersen CA (2011) Transplacental transmission of Leishmania infantum as a means for continued disease incidence in North America. PLoS Negl Trop Dis 5(4):e1019
Burattini MN, Coutinho FAB, Lopez LF, Massad E (1998) Modelling the dynamics of leishmaniasis considering human, animal host and vector populations. J Biol Syst 6(4):337–356
Bureau of Labor Statistics (2016) United States Department of Labor. http://www.bls.gov/, 2016. [Online Accessed 22/2/2016]
Casanova C, Andrighetti MTM, Sampaio SMP, Marcoris MLG, Colla-Jacques FE, Prado AP (2013) Larval breeding sites of Lutzomyia longipalpis (Diptera: Psychodidae) in visceral leishmaniasis endemic urban areas in southeastern Brazil. PLoS Negl Trop Dis 7(9):e2443
Chapman LA, Dyson L, Courtenay O, Chowdhury R, Bern C, Medley GF, Hollingsworth TD (2015) Quantification of the natural history of visceral leishmaniasis and consequences for control. Parasites Vectors 8(1):1–13
Chappuis F, Sundar S, Hailu A, Ghalib H, Rijal S, Peeling RW, Alvar J, Boelaert M (2007) Visceral leishmaniasis: what are the needs for diagnosis, treatment and control? Nat Rev Microbiol 5(11):873–882
Claborn DM (2010) The biology and control of leishmaniasis vectors. J Glob Infect Dis 2(2):127
Dye C (1996) The logic of visceral leishmaniasis control. Am J Trop Med Hyg 55(2):125–130
Dye C, Wolpert DM (1988) Earthquakes, influenza and cycles of Indian kala-azar. Trans R Soc Trop Med Hyg 82(6):843–850
ELmojtaba IM, Mugisha JYT, Hashim MHA (2010) Mathematical analysis of the dynamics of visceral leishmaniasis in the Sudan. Appl Math Comput 217:2567–2578
ELmojtaba IM, Mugisha JYT, Hashim MHA (2013) Vaccination model for visceral leishmaniasis with infective immigrants. Math Methods Appl Sci 36(2):216–226
European centre for disease prevention and control (2014) Phlebotomine sandflies. http://ecdc.europa.eu/en/healthtopics/vectors/sanflies/Pages/sandflies.aspx
Fialho RF, Schall JJ (1995) Thermal ecology of a malarial parasite and its insect vector: consequences for the parasite’s transmission success. J Anim Ecol 64(5):553–562
Garba SM, Gumel AB, Bakar MRA (2008) Backward bifurcations in dengue transmission dynamics. Math Biosci 215:11–25
Gelman A, Carlin J, Stern HS, Rubin DB (1995) Bayesian data analysis. Chapman & Hall, Boca Raton
Heffernan JM, Smith RJ, Wahl LM (2005) Perspectives on the basic reproductive ratio. J R Soc Interface 2:281–293
Islam S, Kenah E, Bhuiyan MAA, Rahman KM, Goodhew B, Ghalib CM et al (2013) Clinical and immunological aspects of Post–Kala–Azar Dermal leishmaniasis in Bangladesh. Am J Trop Med Hyg 89(2):345–353
Joshi AB, Das ML, Akhter S, Chowdhury R, Mondal D, Kumar V, Das P, Kroeger A, Boelaert M, Petzold M (2009) Chemical and environmental vector control as a contribution to the elimination of visceral leishmaniasis on the Indian subcontinent: cluster randomized controlled trials in Bangladesh, India and Nepal. BMC Med 7(1):1
Kamgang JC, Sallet G (2008) Computation of threshold conditions for epidemiological models and global stability of the disease-free equilibrium (DFE). Math Biosci 213:1–12
Kealey A, Smith R (2010) Neglected tropical diseases: infection, modeling, and control. J Health Care Poor Unders 21(1):53–69
LaSalle JP (1968) Stability theory for ordinary differential equations. J Differ Equ 4(1):57–65
Lenhart S, Workman JT (2007) Optimal control applied to biological models. CRC Press, Boca Raton
Magill AJ, Grogl M, Gasser RA Jr, Sun W, Oster CN (1993) Visceral infection caused by leishmania tropica in veterans of operation desert storm. N Engl J Med 328(19):1383–1387
Marino S, Hogue IB, Ray CJR, Kirschner DE (2008) A methodology for performing global uncertainty and sensitivity analysis in systems biology. J Theor Biol 254:178–196
Meheus F, Balasegaram M, Olliaro P, Sundar S, Rijal S, Faiz MA, Boelaert M (2010) Cost-effectiveness analysis of combination therapies for visceral leishmaniasis in the Indian subcontinent. PLoS Negl Trop Dis 4(9):e818
Moncaz A, Faiman R, Kirstein O, Warburg A (2012) Breeding sites of Phlebotomus sergenti, the sand fly vector of cutaneous leishmaniasis in the Judean Desert. PLoS Negl Trop Dis 6(7):e1725
Moore EM, Lockwood DN (2010) Treatment of visceral leishmaniasis. J Glob Infect Dis 2(2):151–158
Morrison AC, Ferro C, Pardo R, Torres M, Devlin B, Wilson ML, Tesh RB (1995) Seasonal abundance of Lutzomyia longipalpis (Dipteral psychodidae) at an endemic focus of visceral leishmaniasis in Colombia. J Med Entomol 32(4):538–548
Mubayi A, Castillo-Chavez C, Chowell G, Kribs-Zaleta C, Siddiqui NA, Kumar N, Das P (2010) Transmission dynamics and underreporting of Kala–Azar in the Indian state of Bihar. J Theor Biol 262(1):177–185
Nawaratna SK, Weilgama DJ, Rajapaksha K (2009) Cutaneous leishmaniasis in Sri Lanka: a study of possible animal reservoirs. Int J Infect Dis 13(4):513–517
Rock KS, le Rutte EA, de Vlas SJ, Adams ER, Medley GF, Hollingsworth TD (2015) Uniting mathematics and biology for control of visceral leishmaniasis. Trends Parasitol 31(6):251–259
Sardar T, Mukhopadhyay S, Bhowmick AR, Chattopadhyay J (2013) An optimal cost effectiveness study on Zimbabwe cholera seasonal data from 2008–2011. PLoS ONE 8(12):e81231
Sardar T, Sasmal SK, Chattopadhyay J (2016) Estimating dengue type reproduction numbers for two provinces of Sri Lanka during the period 2013–14. Virulence 7(2):187–200
Sobol IM (1990) On sensitivity estimation for nonlinear mathematical models. Mat Modelirovanie 2(1):112–118
Sobol IM (2001) Global sensitivity indices for nonlinear mathematical models and their Monte Carlo estimates. Math Comput Simul 55(1):271–280
Srinivasan R, Panicker KN (1993) Laboratory observations on the biology of the phlebotomid sandfly, Phlebotomus papatasi (Scopoli, 1786). Southeast Asian J Trop Med Public Health 24(3):536–539
Stauch A, Sarkar RR, Picado A, Ostyn B, Sundar S (2011) Visceral leishmaniasis in the Indian subcontinent: modelling epidemiology and control. PLoS Negl Trop Dis 5:e1405
Stauch A, Duerr HP, Dujardin JC, Vanaerschot M, Sundar S (2012) Treatment of visceral leishmaniasis: model-based analyses on the spread of antimony-resistant L. donovani in Bihar, India. PLoS Negl Trop Dis 6:e1973
Stauch A, Duerr HP, Picado A, Ostyn B, Sundar S, Rijal S, Boelaert M, Dujardin JC, Eichner M (2014) Model-based investigations of different vector-related intervention strategies to eliminate visceral leishmaniasis on the Indian subcontinent. PLoS Negl Trop Dis 8(4):e2810
Stockdale L, Newton R (2013) A review of preventative methods against human leishmaniasis infection. PLoS Negl Trop Dis 7(6):e2278
Subramanian A, Singh V, Sarkar RR (2015) Understanding visceral leishmaniasis disease transmission and its control: a study based on mathematical modeling. Mathematics 3:913–944
Walker K (2000) Cost-comparison of DDT and alternative insecticides for malaria control. Med Vet Entomol 14(4):345–354
W.H.O. (2013) Crude birth and death rate Data by country. www.who.int/gho/data/view.main.CBDR2040 [Online Accessed 10/2/2016]
W.H.O. (2014) Summary of visceral leishmaniasis (VL) or Kala azar (KA) outbreak situation in South Sudan. http://www.who.int/hac/crises/ssd/sitreps/south_sudan_kala_azar_september2014.pdf [Online Accessed 10/2/2016]
Zijlstra EE, El-Hassan AM, Ismael A, Ghalib HW (1994) Endemic kala–azar in eastern sudan: a longitudinal study on the incidence of clinical and subclinical infection and post–kala–azar dermal leishmaniasis. Am J Trop Med Hyg 51(6):826–836
Acknowledgements
The authors are grateful to the Editor-in-chief Prof. Reinhard Laubenbacher and the learned reviewers for their comments and suggestions on the earlier version of this paper. The comments immensely improve the standard of the manuscript. Special thanks to the learned first reviewer of the manuscript for providing a rigorous review. Indrajit Ghosh was supported by the research fellowship from University Grant Commission (UGC), Government of India. Dr. Tridip Sardar was supported by the postdoctoral fellowship from National Board of Higher Mathematics (NBHM), Government of India. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix 1
Proof of Lemma 1
System (1)–(5) can be rewritten in the following form:
where \(X = (S_\mathrm{H}, A_\mathrm{H}, K_\mathrm{H}, D_\mathrm{H}, P_\mathrm{H}, R_\mathrm{H}, S_\mathrm{A}, I_\mathrm{A}, F_\mathrm{NA}, S_{V}, I_{V})^\mathrm{T}\) and
where \(Q_\mathrm{H}=b p_\mathrm{H}^{v} \frac{I_{V}}{N_\mathrm{H}}\), \(a_{9}=\displaystyle r \kappa _{F}\left( 1- \frac{F_\mathrm{NA}}{C}\right) \), \(a_{10}=\displaystyle -\mu _{F} - b p_{v}^{H} \frac{(K_\mathrm{H} + P_\mathrm{H} + \lambda A_\mathrm{H})}{N_\mathrm{H}} - b p_{v}^{A}\frac{I_\mathrm{A}}{N_\mathrm{A}}.\)
The vector \(\displaystyle B = (\pi _\mathrm{H},0,0,0,0,0,\pi _\mathrm{A},0,0,0,0)^\mathrm{T}\) is positive. Note that A(X) has all off-diagonal entries nonnegative, i.e., A(X) is a Metzler matrix for all \(X \in \mathbb {R}_{+}^{11}\), since \(B \ge 0\) system (1)–(5) is positively invariant in \(\mathbb {R}_{+}^{11}\) (Abate et al. 2009). Therefore, any trajectory of the system (1)–(5) starting from an initial state in \(\mathbb {R}_{+}^{11}\) remains trapped forever in \(\mathbb {R}_{+}^{11}\).
Adding the first six equations of the model system (1)–(5), we get
Hence, \( 0 \le N_\mathrm{H}(t) \le \frac{\varPi _\mathrm{H}}{\mu _\mathrm{H}} + \left( N_\mathrm{H}(0) - \frac{\varPi _\mathrm{H}}{\mu _\mathrm{H}}\right) e^{-\mu _\mathrm{H} t}\). Therefore, as \(t \rightarrow \infty \), \( 0 \le N_\mathrm{H}(t) \le \frac{\varPi _\mathrm{H}}{\mu _\mathrm{H}}\) and, for any \(t>0\), \( 0 \le N_\mathrm{H}(t) \le Z_1\) , where \(Z_1 = \max \left\{ \frac{\varPi _\mathrm{H}}{\mu _\mathrm{H}},N_\mathrm{H}(0)\right\} \).
By adding the equations in the \(S_\mathrm{A}\) and \(I_\mathrm{A}\) compartments of system (1)–(5), we get
By similar arguments, we have, for any \(t>0\), \( 0 \le N_\mathrm{A}(t) \le Z_2\), where \(Z_2 = \max \left\{ \frac{\varPi _\mathrm{A}}{\mu _\mathrm{A}},N_\mathrm{A}(0)\right\} \).
Assume that \(Z_3=\max \left\{ C,F_\mathrm{NA}(0)\right\} \). Then \(F_\mathrm{NA} \le Z_3\). Adding the equations in \(S_{V}\) and \(I_{V}\) compartments of system (1)–(5), we get:
which implies \( 0 \le (S_{V} + I_{V}) \le \frac{\eta Z_3}{\mu _{F}}\).
Therefore, all mathematically and biologically feasible solutions of the model system (1)–(5) enter the region \(\varSigma \); i.e, \(\varSigma \) is attracting. Hence, it is now sufficient to study the dynamical properties of the model (1)– (5) in \(\varSigma \). \(\square \)
Appendix 2
Proof of Theorem 1
Assuming \(M = S_{V} + I_{V}\), we consider the subsystem of the system (1)–(5) as follows:
To study the global stability of \(E_{0}\), we consider the following Lyapunov function for subsystem (17) in \(\varSigma \):
The total derivative of V is given by
since \(R_{C} \le 1\), \(\displaystyle \dot{V} \le 0\).
Also, \(\dot{V} = 0\) if and only if \(F_\mathrm{NA} = M = 0\). Applying Lasalle’s invariance principle (LaSalle 1968), we get \(F_\mathrm{NA} \rightarrow 0\) and \(M \rightarrow 0\) as \(t \rightarrow \infty \).
Again, \(S_{V} \ge 0\), \( I_{V} \ge 0\) and \(M = S_{V} + I_{V} \rightarrow 0\) as \(t \rightarrow \infty \), so it follows that \(S_{V} \rightarrow 0\) and \(I_{V} \rightarrow 0\).
Substituting \(F_\mathrm{NA} = S_{V} = I_{V} = 0\) in the original system (1)–(5), we get
\((S_\mathrm{H}, A_\mathrm{H}, K_\mathrm{H}, D_\mathrm{H}, P_\mathrm{H}, R_\mathrm{H}, S_\mathrm{A}, I_\mathrm{A}) \rightarrow \left( \frac{\pi _\mathrm{H}}{\mu _\mathrm{H}},0,0,0,0,0,\frac{\pi _\mathrm{A}}{\mu _\mathrm{A}},0\right) \) as \(t \rightarrow \infty \).
As \(\varSigma \) is positively invariant and attracting, the trivial vector-free equilibrium \(E_{0}\) is globally asymptotically stable in \(\varSigma \) if \(R_{C} \le 1\). \(\square \)
Appendix 3
Proof of Theorem 2
Let us consider the bounded set
The compartments of the uninfected individuals of the system (1)–(5) can be written in the following way
with \( X = (S_\mathrm{H}, R_\mathrm{H}, S_\mathrm{A}, F_\mathrm{NA}, S_{V})^\mathrm{T}\).
From system (18), it is clear that \((S_\mathrm{H}, R_\mathrm{H}, S_\mathrm{A}) \rightarrow \left( \frac{\pi _\mathrm{H}}{\mu _\mathrm{H}},0,\frac{\pi _\mathrm{A}}{\mu _\mathrm{A}}\right) \) as \( t \rightarrow \infty \).
The remaining two compartments of the system (18)) are
Clearly, the non-trivial equilibrium \( \bigg (C\left( 1 - \frac{1}{R_{C}}\right) ,\frac{\eta C}{\mu _{F}}\left( 1 - \frac{1}{R_{C}}\right) \bigg )\) is a stable node of the system (19). Also, a direct application of Bendixon’s nonexistence criterion shows that system (19) contains no closed path. Therefore, the equilibrium \( (C(1 - \frac{1}{R_{C}}),\frac{\eta C}{\mu _{F}}(1 - \frac{1}{R_{C}}))\) of the system (19) is globally asymptotically stable. Thus, \(E_{1}\) is globally asymptotically stable for system (18).
Therefore, for our model system (1)–(5) the first two conditions are satisfied. Furthermore, in G, \(A_{2}^{0}\) can be written as follows:
with \( \displaystyle M_{1} = \displaystyle bp_{v}^{H}\frac{\eta C\pi _\mathrm{H}}{\mu _{F}(\mu _\mathrm{H}+\delta )}\).
We can write \(A_{2}^{0} = Q + P\), where Q and P have the following forms:
and
with \( \displaystyle M_{1} = \displaystyle bp_{v}^{H}\frac{\eta C\pi _\mathrm{H}}{\mu _{F}(\mu _\mathrm{H}+\delta )}\)
Q is Metzler stable and \(P \ge 0\). Therefore, using Proposition 3.3 of Kamgang and Sallet (2008), we get \(\alpha (A_{2}^{0}) \le 0 \Leftrightarrow \rho (-PQ^{-1})\le 1\).
After a few calculations, we get \(\rho (-PQ^{-1})\le 1\), giving \( R_{*} \le 1\), with
From the expression (20), it is clear that \( R_{*}^{2} > R_{0}^{2}\). Therefore, if \(R_{C} > 1\) and \(R_{0}< R_{*} < 1\), then the non-trivial disease-free equilibrium \(E_{1}\) is globally asymptotically stable in G. \(\square \)
Appendix 4
Proof of Theorem 3
Putting the values of \(I_{V}^{*}\) and \(N_\mathrm{H}^{*}\) in the expression
we get a quadratic equation in \(\lambda _\mathrm{H}\):
where
When \(\delta = 0\) and \(\beta = 0 \), \(\displaystyle N_\mathrm{H}^{*}= \frac{\varPi _\mathrm{H}}{\mu _\mathrm{H}}\). The expression of \(\lambda _\mathrm{H}\) takes the form
Since \(F_\mathrm{NA}^{*} > 0\) for \(R_{C}>1\), it follows that \(\lambda _\mathrm{H}>0\) if and only if \(R_{0}>1\). Hence Case 1 follows.
Similarly, when \(\delta = 0\) and \(\beta > 0 \), the expression of \(\lambda _\mathrm{H}\) is obtained as follows
which is positive if \(R_{0}>1\) and \(\beta F_{1} < 1\) or if \(R_{0}>1\) and \(\displaystyle \beta F_{1}> 1 + \frac{\mu _{F} \varPi _\mathrm{H} R_{0}^{2}}{b p_\mathrm{H}^{v} \eta F_\mathrm{NA}^{*}}\). This proves Case 2.
Finally, when \(\delta > 0\) and \(\beta > 0 \),
Note that \(\lambda _\mathrm{H}\) always possesses a positive solution if \(C > 0\) and \(A < 0\) or if \(C < 0\) and \(A > 0\). Clearly, \(C > 0\) if \(R_{0}>1\) and \(A < 0\) if \(\sigma< \beta F_{1} < 1 - \frac{\delta (1-\sigma )}{( \alpha _{1}+\delta +\mu _\mathrm{H})}\).
Also \(A < 0\) if \(R_{0}^{2} < \displaystyle \frac{b p_\mathrm{H}^{v} \eta F_\mathrm{NA}^{*}}{\mu _{F} \varPi _\mathrm{H}} \left[ \frac{\delta (1-\sigma ) }{ \alpha _1 + \delta + \mu _\mathrm{H}} +(\beta F_{1} -1) \right] \).
This proves Case 3a(i) and Case 3a(ii).
Again \(C < 0\) if \(R_{0}<1\) and \(A > 0\) if \(1 - \frac{\delta (1-\sigma ) }{ \alpha _1 + \delta + \mu _\mathrm{H}}< \beta F_{1} < \displaystyle 1+ \frac{\mu _{F} \varPi _\mathrm{H} R_{0}^{2}}{b p_\mathrm{H}^{v} \eta F_\mathrm{NA}^{*}}\); i.e, if Case 3a(iii) is satisfied.
If \(B^{2}>4AC\), then, depending on the sign of B, the cases when two endemic equilibria can occur are given by \(B<0,A>0,C>0\) and \(B>0,A<0,C<0\). It is clear from the expressions of A, B and C that the above two inequalities hold only when Case 3b(i) and Case 3b(ii) are satisfied, respectively. This completes the proof. \(\square \)
Appendix 5
Proof of Theorem 4
For \(\delta =0\), putting the values of \(I_{V}^{*}\) and \(N_\mathrm{H}^{*}\) in the expression
we get a quadratic equation in \(\lambda _\mathrm{H}\):
where
when \(\delta =0\) and \(\beta =0\), it is clear that model (1)–(5) has exactly one endemic equilibrium whenever \(R_{0}>1\) and has two endemic equilibria whenever \(B_{0}>0\), \(R_{0} < 1\) and \(B_{0}^{2}>4A_{0}C_{0}\). Hence Case 1 follows.
For \(\delta =0\) and \(\beta >0\), it follows that \(\lambda _\mathrm{H}\) always possesses a positive solution if \(C_{0} > 0\) and \(A_{0} < 0\) or if \(C_{0} < 0\) and \(A_{0} > 0\). From the expressions of \(A_{0}\), \(B_{0}\) and \(C_{0}\), we find that \(A_{0} < 0\) if \(\beta F_{1}<1\) and \(C_{0} > 0\) if \(R_{0}>1\).
Again \(A_{0} > 0\) if \(\displaystyle \beta F_{1}> 1 + \frac{1}{\mu _\mathrm{H}+p_{v}^{A}}\left( \frac{\mu _{F}^{2} \varPi _\mathrm{H} R_{0}^{2}}{b p_\mathrm{H}^{v} \eta F_\mathrm{NA}^{*}}-\frac{bp_\mathrm{A}^{v}p_{v}^{A}\varPi _\mathrm{H}}{\varPi _\mathrm{H}p_\mathrm{H}^{v}}\right) \) and \(C_{0} < 0\) if \(R_{0}^{2}< 1\).
This proves Case 2a. If \(B_{0}^{2}>4A_{0}C_{0}\), model (1)–(5) possesses two endemic equilibria whenever \(B_{0}<0\), \(A_{0}>0\), \(C_{0}>0\) or \(B_{0}>0\), \(A_{0}<0\), \(C_{0}<0\). It is clear from the expressions of \(A_{0}\), \(B_{0}\) and \(C_{0}\) that the above two inequalities hold only when Case 2b(i) and Case 2b(ii) are satisfied, respectively. This completes the proof. \(\square \)
Rights and permissions
About this article
Cite this article
Ghosh, I., Sardar, T. & Chattopadhyay, J. A Mathematical Study to Control Visceral Leishmaniasis: An Application to South Sudan. Bull Math Biol 79, 1100–1134 (2017). https://doi.org/10.1007/s11538-017-0274-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-017-0274-5