Abstract
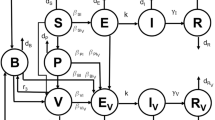
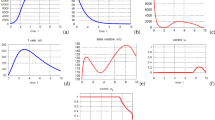
We modify the classical virus dynamics model by incorporating an immune response with fixed or fluctuating vaccination frequencies and dosages to obtain a system of impulsive differential equations for the virus dynamics of both the wild-type and mutant strains. This model framework permits us to obtain precise conditions for the virus elimination, which are much more feasible compared with existing results, which require frequent vaccine administration with large dosage. We also consider the corresponding impulsive optimal control problem to describe when and how much of the vaccine should be administered in order to maximize levels of healthy CD4+ T cells and immune response cells. A gradient-based optimization method is applied to obtain the optimal schedule numerically. For a case study when the CTL vaccine is administered in a period of one year, our numerical studies support the optimal vaccination schedule consisting of vaccine administration three times, with the first dosage strong (to boost the immune system), followed by a second dosage shortly after (to strengthen the immune response) and then the third and final dosage long after (to ensure the immune system can handle viruses rebound).





Similar content being viewed by others
References
Agur, Z., Cojocaru, L., et al. (1993). Pulse mass measles vaccination across age cohorts. Proc. Natl. Acad. Sci. USA, 90, 11698–11702.
Bacaër, N. (2007). Approximation of the basic reproduction number R 0 for vector-borne diseases with a periodic vector population. Bull. Math. Biol., 69, 1067–1091.
Barles, G. (1985). Deterministic impulsive control problems. SIAM J. Control Optim., 23, 419–432.
Barouch, D. H., Kunstman, J., et al. (2002). Eventual AIDS vaccine failure in a rhesus monkey by viral escape from cytotoxic T lymphocytes. Nature, 415, 335–339.
Bensoussan, A., & Lions, J.-L. (1984). Impulse control and quasi-variational inequalities. Philadelphia: Wiley.
Cappuccio, A., Castiglione, F., & Piccoli, B. (2007). Determination of the optimal therapeutic protocol in cancer immunotherapy. Math. Biosci., 209, 1–13.
Castiglione, F., & Piccoli, B. (2006). Optimal control in a model of dendritic cell transfection cancer immunotherapy. Bull. Math. Biol., 68, 255–274.
Castiglione, F., & Piccoli, B. (2007). Cancer immunotherapy, mathematical modelling, and optimal control. J. Theor. Biol., 247, 713–732.
Christian, L. A., & Rob, J. D. (2008). Dynamics of immune escape during HIV/SIV infection. PLoS Comput. Biol., 4(7), e1000103.
Culshaw, R. V., Ruan, S. G., & Spiteri, R. J. (2004). Optimal HIV treatment by maximising immune response. J. Math. Biol., 48, 545–562.
Currie, J. R., Visawapoka, U., et al. (2006). CTL epitope distribution patterns in the Gag and Nef proteins of HIV-1 from subtype A infected subjects in Kenya: use of multiple peptide sets increases the detectable breadth of the CTL response. BMC Immunol., 7(8).
De Quadros, C. A., Andrus, J. K., & Olive, J. M. (1991). Eradication of poliomyelitis: progress in the Americas. Pediatr. Infect. Dis. J., 10(3), 222–229.
Fister, K. R., Lenhart, S., & McNally, J. S. (1998). Optimizing chemotherapy in an HIV model. Electron. J. Differ. Equ., 32, 1–12.
Fleming, W. H., & Rishel, R. W. (1975). Deterministic and stochastic optimal control. New York: Springer.
Jennings, L. S., Fisher, M. E., Teo, K. L., & Goh, C. J. (1991). MISER3: solving optimal control problems—an update. Adv. Eng. Softw. Workstn., 13, 190–196.
Kirschner, D. E., Lenhart, S., & Serbin, S. (1997). Optimal control of the chemotherapy of HIV. J. Math. Biol., 35, 775–792.
Klausner, R. D., Fauci, A. S., et al. (2003). The need for a global HIV vaccine enterprise. Science, 300, 2036–2039.
Konrad, B. P., Vaidya, N. K., & Smith, R. J. (2011). Modelling mutation to a cytotoxic T-lymphocyte HIV vaccine. Math. Popul. Stud., 18, 122–149.
Li, X., & Yong, J. (1995). Optimal control theory for infinite-dimensinal systems. Boston: Birkhäuser.
Liu, Y., Teo, K. L., et al. (1998). On a class of optimal control problems with state jumps. J. Optim. Theory Appl., 98(1), 65–82.
Lou, J., Lou, Y. J., & Wu, J. H. (2011). Threshold virus dynamics with impulsive antiretroviral drug effects. J. Math. Biol. doi:10.1007/s00285-011-0474-9.
Luo, L., Li, Y., Chang, J. S., et al. (1998). Induction of V3-specific cytotoxic T lymphocyte responses by HIV gag particles carrying multiple immunodominant V3 epitopes of gp120. Virology, 240, 316–325.
Marino, S., Hogue, I. B., et al. (2008). A methodology for performing global uncertainty and sensitivity analysis in systems biology. J. Theor. Biol., 254, 178–196.
McMichael, A. J., & Hanke, T. (2003). HIV vaccines 1983–2003. Nat. Med., 9, 874–880.
Nowak, M. A., & McLean, A. R. (1991). A mathematical model of vaccination against HIV to prevent the development of AIDS. Proc. Biol. Sci., 246, 141–146.
Piccoli, B., & Castiglione, F. (2006). Optimal vaccine scheduling in cancer immunotherapy. Physica A, 370(2), 672–680.
Pontryagin, L., Boltyanskii, V., et al. (1986). The mathematical theory of optimal process. New York: Gordon & Breach.
Rong, L. B., Feng, Z. L., & Perelson, A. S. (2007). Emergence of HIV-1 drug resistance during antiretroviral treatment. Bull. Math. Biol., 69, 2027–2060.
Rossio, J. L., Esser, M. T., et al. (1998). Inactivation of human immunodeficiency virus type 1 infectivity with preservation of conformational and functional integrity of virion surface proteins. J. Virol., 72, 7992–8001.
Sabin, A. B. (1991). Measles, killer of millions in developing countries: strategies of elimination and continuing control. Eur. J. Epidemiol., 7, 1–22.
Shulgin, B., Stone, L., & Agur, Z. (1998). Pulse vaccination strategy in the SIR epidemic model. Bull. Math. Biol., 60, 1123–1148.
Smith, R. J., & Schwartz, E. J. (2008). Predicting the potential impact of a cytotoxic T-lymphocyte HIV vaccine: how often should you vaccinate and how strong should the vaccine be? Math. Biosci., 212, 180–187.
Smith, S. M. (2004). HIV CTL escape: at what cost? Retrovirology, 1(8).
Wahl, L. M., & Nowak, M. A. (2000). Adherence and drug resistance: predictions for therapy outcome. Proc. R. Soc. Lond. B, 267, 835–843.
Wang, W. D., & Zhao, X.-Q. (2008). Threshold dynamics for compartmental epidemic models in periodic environments. J. Dyn. Differ. Equ., 20, 699–717.
Wein, L. M., Zenios, S. A., & Nowak, M. A. (1997). Dynamic multidrug therapies for HIV: a control theoretic approach. J. Theor. Biol., 185, 15–29.
Wodarz, D., & Lloyd, A. L. (2004). Immune responses and the emergence of drug-resistant virus strains in vivo. Proc. R. Soc. Lond. B, 271, 1101–1109.
Yang, Y. P., & Xiao, Y. N. (2010). The effects of population dispersal and pulse vaccination on disease control. Math. Comput. Model., 52, 1591–1604.
Yang, Y. P., & Xiao, Y. N. (2012). Threshold dynamics for compartmental epidemic models with impulses. Nonlinear Anal., Real World Appl., 13, 224–234.
Zhang, F., & Zhao, X.-Q. (2007). A periodic epidemic model in a patchy environment. J. Math. Anal. Appl., 325, 496–516.
Zhao, X.-Q. (2003). Dynamical systems in population biology. New York: Springer.
Acknowledgements
Y. Yang acknowledges China Scholarship Council for the financial support while visiting York University. The authors are supported by the National Mega-project of Science Research (2012ZX10001-001), by the National Natural Science Foundation of China (NSFC 11171268), and by the Fundamental Research Funds for the Central Universities (08143042) (YX). The authors are also supported by the Canada Research Chair Program, the Natural Sciences and Engineering Research Council of Canada, the Mitacs/Mprime (JW), as well as the International Development Research Center (Ottawa, Canada, 104519-010) (YX, JW).
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Proof of Lemma 3.1
By the continuity of solutions with respect to the initial values, ∀ϵ>0 there exists δ 0>0 such that for all x 0∈X 0 with ∥x 0−M 1∥≤δ 0, there holds ∥u(t,x 0)−u(t,M 1))∥≤ϵ,∀t∈[0,τ]. We further claim that Eq. (5) holds. Assume, by contradiction, that (5) does not hold. Then we have
for some x 0∈X 0. Without loss of generality, we assume that d(P m(x 0),M 1)<δ 0, for all m≥0. It follows that
For any t≥0, let t=mτ+t′, where t′∈[0,τ] and \(m=[\frac{t}{\tau}]\) is the greatest integer less than or equal to \(\frac{t}{\tau}\). Thus, we get
Note that (T(t),T w (t),V w (t),T r (t),V r (t),C(t))=u(t,x 0). It then follows that T w (t)<ϵ,V w (t)<ϵ,T r (t)<ϵ,V r (t)<ϵ,∀t≥0. Then from the first and last equations of (1), we have

Consider an auxiliary system
For any ϵ>0, system (20) admits a globally asymptotically stable solution \((\hat{T}(0,\epsilon), C^{*}(t,\epsilon))\), where \(\hat{T}(0,\epsilon)=\lambda/(\delta_{T}+\epsilon \beta_{w}+\epsilon \beta_{r}), C^{*}(t,\epsilon)= \tilde{C}e^{(2\epsilon\alpha-\delta_{C})(t-n\tau)}/ (1-e^{(2\epsilon\alpha-\delta_{C})\tau}), t\in (i\tau, (i+1)\tau]\). Then for any ξ>0, there exists t 3>0 such that \(\hat{T}(t,\epsilon)\geq \hat{T}(0,\epsilon)-\xi, \hat{C}(t,\epsilon)\leq C^{*}(t,\epsilon)+\xi\) for t≥t 3, \((\hat{T}(t,\epsilon), \hat{C}(t,\epsilon))\) is any solution of Eq. (20). Note that \(\hat{T}(0,\epsilon) \rightarrow T_{0}, C^{*}(t,\epsilon)\rightarrow C^{*}(t)\) as ϵ→0. Then for any \(\bar{\eta}>0\) there exists \(\bar{\epsilon}>0\) such that \(\hat{T}(0,\epsilon)\geq T_{0}-\bar{\eta}, C^{*}(t,\epsilon)\leq C^{*}(t)+\bar{\eta}\) for \(\epsilon<\bar{\epsilon}\). It follows that for t≥t 3 and ϵ small enough (\(\epsilon<\bar{\epsilon}\))

It follows from Eq. (20) and comparison principles that for t≥t 3 and ϵ small enough,
Consider the fourth and the fifth equations in system (1) with the nonnegativity of the solutions, there holds for t≥t 3
Consider the corresponding comparison differential equations of system (21),
By Zhang and Zhao (2007) (Lemma 2.1), we know that there exists a positive, τ-periodic function p(t)=(p 1(t),p 2(t)), such that \(e^{\mu_{2}t}p(t)\) is a solution of system (22), where \(\mu_{2}=\frac{1}{\tau}\ln\rho(\varPhi_{F_{r}(\eta)-V_{r}(\eta)}(\tau))\). Since \(\rho(\varPhi_{F_{r}(\eta)-V_{r}(\eta)}(\tau))\) is continuous for small η and \(R_{0}^{r}>1\) indicates that \(\rho(\varPhi_{F_{r}-V_{r}}(\tau))>1\), we can choose η small enough such that \(\rho(\varPhi_{F_{r}(\eta)-V_{r}(\eta)}(\tau))>1\), that is μ 2>0. Let t=nτ>t 3, and n be nonnegative integer, we get

For any negative initial values (T I (t 3),V I (t 3))T of system (21), there exits a sufficiently small z ∗>0, such that (T r (t 3),V r (t 3))T≥z ∗(p 1(0),p 2(0))T. By the comparison theorem, we have \((T_{r}(t),V_{r}(t))^{T}\geq z_{*}e^{\mu_{2}(t-t_{3})}(\hat{T}_{r}(t-t_{3}),\hat{V}_{r}(t-t_{3}))^{T}\), for all t≥t 3. Thus, we obtain T r (nτ)→∞,V r (nτ)→∞, as n→∞, a contradiction. Hence, Eq. (5) holds. This completes the proof. □
Rights and permissions
About this article
Cite this article
Yang, Y., Xiao, Y. & Wu, J. Pulse HIV Vaccination: Feasibility for Virus Eradication and Optimal Vaccination Schedule. Bull Math Biol 75, 725–751 (2013). https://doi.org/10.1007/s11538-013-9831-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-013-9831-8