Abstract
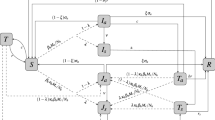
Sulfadoxine-pyrimethamine (SP) has been one of the most widely used antimalarial treatments world-wide, and is also used prophylactically in vulnerable populations. In this paper, we develop a mathematical model which allows us to infer the time distribution of SP protection from drug-trial data. Fitting our model to data from a controlled field study in Mali, we find that SP provided protection from malaria for an average of 37.9 days in this pediatric population. We demonstrate that the duration of SP protection is not well described by an exponential distribution, and in fact has a much narrower dispersal about the mean; the best-fit standard deviation predicted by our model was only 17.0 days, as opposed to 41.8 days for the exponential model. We estimate the monthly entomological inoculation rate and the basic reproductive number for malaria in this population, and demonstrate that extremely high SP treatment rates would be necessary to maintain an effective reproductive number below one throughout a single rainy season. These results have implications for further efforts to model the impact of SP treatment, or for investigations of the optimal timing of prophylactic SP.








Similar content being viewed by others
References
Aguas, R., White, L. J., Snow, R. W., & Gomes, M. G. M. (2008). Prospects for malaria eradication in Sub-Saharan Africa. PLoS ONE, 3, 1–6.
Anderson, R. M., & May, R. M. (1991). Infectious diseases of humans: dynamics and control. Oxford: Oxford University Press.
Arino, J., Ducrot, A., & Zongo, P. (2012). A metapopulation model for malaria with transmission-blocking partial immunity in hosts. J. Math. Biol., 64, 423–448.
Auger, P., Kouokam, E., Sallet, G., Tchuente, M., & Tsanou, B. (2008). The Ross–Macdonald model in a patchy environment. Math. Biosci., 216, 123–131.
Barnes, K. I., Little, F., Smith, P. J., Evans, A., Watkins, W. M., & White, N. J. (2006). Sulfadoxine-pyrimethamine pharmacokinetics in malaria: pediatric dosing implications. Clin. Pharmacol. Ther., 67, 582–596.
Beier, J. C. (1998). Malaria parasite development in mosquitoes. Annu. Rev. Entomol., 43, 519–543.
Bell, D. J., Nyirongo, S. K., Mukaka, M., Zijlstra, E. E., Plowe, C. V., Molyneux, M. E., Ward, S. A., & Winstanley, P. A. (2008). Sulfadoxine-pyrimethamine-based combinations for malaria: a randomised blinded trial to compare efficacy, safety and selection of resistance in Malawi. PLoS ONE, 3, e1578.
Burkot, T. R., & Graves, P. M. (1995). The value of vector-based estimates of malaria transmission. Ann. Trop. Med. Parasitol., 89, 125–134.
Centers for Disease Control and Prevention (2010). Malaria fact sheets. http://www.cdc.gov/malaria/about/facts.html.
Centers for Disease Control and Prevention (2012). CDC health information for international travel 2012: the yellow book. Oxford: Oxford University Press.
Chitnis, N., Cushing, J. M., & Hyman, J. M. (2006). Bifurcation analysis of a mathematical model for malaria transmission. SIAM J. Appl. Math., 67, 24–45.
Chitnis, N., Smith, T., & Steketee, R. (2008). A mathematical model for the dynamics of malaria in mosquitoes feeding on a heterogeneous host population. J. Biol. Dyn., 2, 259–285.
Chitnis, N., Hardy, D., & Smith, T. (2012). A periodically-forced mathematical model for the seasonal dynamics of malaria in mosquitoes. Bull. Math. Biol., 74, 1098–1124. doi:10.1007/s11538-011-9710-0.
Cintron-Arias, A., Castillo-Chavez, C., Bettencourt, L. M. A., Lloyd, A. L., & Banks, H. T. (2009). The estimation of the effective reproductive number from disease outbreak data. Math. Biosci. Eng., 6, 261–282.
Coulibaly, D., Diallo, D. A., Thera, M. A., Dicko, A., Guindo, A. B., Kone, A. K., Cissoko, Y., Coulibaly, S., Djimde, A., Lyke, K., Doumbo, O. K., & Plowe, C. V. (2002). Impact of preseason treatment on incidence of falciparum malaria and parasite density at a site for testing malaria vaccines in Bandiagara, Mali. Am. J. Trop. Med. Hyg., 67, 604–610.
Dembele, B., Friedman, A., & Yakubu, A. (2010). Mathematical model for optimal use of sulfadoxine-pyrimethamine as a temporary malaria vaccine. Bull. Math. Biol., 72, 914–930.
Dicko, A., Toure, S. O., Traore, M., Sagara, I., Toure, O. B., Sissoko, M. S., Diallo, A. T., Rogier, C., Salomon, R., de Sousa, A., & Doumbo, O. K. (2011). Increase in epi vaccines coverage after implementation of intermittent preventive treatment of malaria in infant with sulfadoxine-parimethamine in the district of Kolokani, Mali: results from a cluster randomized control trial. BMC Public Health, 11, 1–7.
Diekmann, O., Heesterbeek, J. A. P., & Metz, J. A. J. (1990). On the definition and the computation of the basic reproduction ratio R 0 in models for infectious diseases in heterogeneous populations. J. Math. Biol., 28, 365–382.
Dietz, K., Molineaux, L., & Thomas, A. A. (1974). Malaria model tested in the African Savannah. Bull. World Health Organ., 50, 347–357.
Drakeley, C., Schellenberg, D., Kihonda, J., Sousa, C. A., Arez, A. P., Lopes, D., Lines, J., Mshinda, H., Lengeler, C., Schellenberg, J. A., Tanner, M., & Alonso, P. (2003). An estimation of the entomological inoculation rate for Ifakara: a semi-urban area in a region of intense malaria transmission in Tanzania. Trop. Med. Int. Health, 8, 767–774.
Ducrot, A., Sirima, S. B., Somé, B., & Zongo, P. (2009). A mathematical model for malaria involving differential susceptibility, exposedness and infectivity of human host. J. Biol. Dyn., 3, 574–598.
Egan, A., Crawley, J., & Schellenberg, D. (2005). Ipti consortium. Intermittent preventive treatment for malaria control in infants: moving towards evidence-based policy and public health action. Trop. Med. Int. Health, 10, 815–817.
Farrington, C. P., & Whitaker, H. J. (2003). Estimation of effective reproduction numbers for infectious diseases using serological survey data. Biostatistics, 4, 626–632.
Filipe, J. A. N., Riley, E. M., Drakeley, C. J., Sutherland, C. J., & Ghani, A. C. (2007). Determination of the processes driving the acquisition of immunity to malaria using a mathematical transmission model. PLoS Comput. Biol., 3, e255.
Griffin, J. T., Hollingsworth, T. D., Okell, L. C., Churcher, T. S., White, M., Hinsley, W., Bousema, T., Drakely, C. J., Ferguson, N. M., Basáñez, M. G., & Ghani, A. C. (2010). Reducing Plasmodium falciparum malaria transmission in Africa: a model-based evaluation of intervention strategies. PLoS Med., 7, e1000324.
De Groote, H., Douro-Kpindou, O., & Togo, T. (2003). Biological control of locusts and grasshoppers in the Dogon area: a participatory rural appraisal. LUBILOSA Socioecon. Working Paper, 98, 1–17.
Kelly-Hope, L. A., & Mckenzie, F. (2009). The multiplicity of malaria transmission: a review of entomological inoculation rate measurements and methods across Sub-Saharan Africa. Malar. J., 8, 1–16.
Koella, J. C., & Antia, R. (2003). Epidemiological models for the spread of anti-malarial resistance. Malar. J., 2, 1–11.
Lou, Y., & Zhao, X. Q. (2010). A climate-based malaria transmission model with structured vector population. SIAM J. Appl. Math., 70, 2023–2044.
Lyke, K. E., Dicko, A., Kone, A., Coulibaly, D., Guindo, A., Cissoko, Y., Traoré, K., Plowe, C. V., & Doumbo, O. K. (2004). Incidence of severe Plasmodium falciparum malaria as a primary endpoint for vaccine efficacy trials in Bandiagara, Mali. Vaccine, 22, 3169–3174.
Macdonald, G. (1957). The epidemiology and control of malaria. London: Oxford University Press.
Maire, N., Smith, T., Ross, A., Owusu-Agyei, S., Dietz, K., & Molineaux, L. (2006). A model for natural immunity to asexual blood stages of Plasmodium falciparum malaria in endemic areas. Am. J. Trop. Med. Hyg., 75, 19–31.
Ruan, S., Xiaob, D., & Beier, J. C. (2008). On the delayed Ross–Macdonald model for malaria transmission. Bull. Math. Biol., 70, 1089–1114.
Shaukat, A. M., Breman, J. G., & Mackenzie, F. E. (2010). Using the entomological inoculation rate to assess the impact of vector control on malaria parasite transmission and elimination. Malar. J., 9, 122.
Smith, D. L., Dushoff, J., Snow, R. W., & Hay, S. I. (2005a). The entomological inoculation rate and Plasmodium falciparum infection in African children. Nature, 438, 492–495.
Smith, D. L., Dushoff, J., & Mckenzie, F. E. (2005b). The risk of a mosquito-borne infection in a heterogeneous environment. PLoS Biol., 2, 1957–1964.
Smith, D. L., Mckenzie, F. E., Snow, R. W., & Hay, S. I. (2007). Revisiting the basic reproductive number for malaria and its implications for malaria control. PLoS Biol., 5, 531–542.
Smith, D. L., Drakeley, C. J., Chiyaka, C., & Hay, S. I. (2010). A quantitative analysis of transmission efficiency versus intensity for malaria. Nat. Commun., 1, 108. doi:10.1038/ncomms1107.
Sogoba, N., Doumbia, S., Vounatsou, P., Baber, I., Keita, M., Maiga, M., Traore, S. F., Toure, A., Dolo, G., Smith, T., & Ribeiro, J. M. C. (2007). Monitoring of larval habitats and mosquito densities in the Sudan Savanna of Mali: implications for malaria vector control. Am. J. Trop. Med. Hyg., 72, 82–88.
ter Kuile, F. O., van Eijk, A. M., & Filler, S. J. (2007). Effect of sulfadoxine-pyrimethamine resistance on the efficacy of intermittent preventive therapy for malaria control during pregnancy. J. Am. Med. Assoc., 297, 2603–2616.
van den Driessche, P., & Watmough, J. (2002). Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math. Biosci., 180, 29–48.
World Health Organization (2010). WHO policy recommendation on intermittent preventive treatment during infancy with sulphadoxine pyrimethamine (SP-IPTi) for Plasmodium falciparum malaria control in Africa. http://www.who.int/malaria/news/WHO_policy_recommendation_IPTi_032010.pdf.
Wyse, A. P. P., Bevilacqua, L., & Rafikov, M. (2007). Simulating malaria model for different treatment intensities in a variable environment. Ecol. Model., 206, 322–330.
Acknowledgements
The authors are grateful to two referees for their insightful comments, and to the Natural Sciences and Engineering Research Council of Canada for funding.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Akbari, S., Vaidya, N.K. & Wahl, L.M. The Time Distribution of Sulfadoxine-Pyrimethamine Protection from Malaria. Bull Math Biol 74, 2733–2751 (2012). https://doi.org/10.1007/s11538-012-9775-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-012-9775-4