Abstract
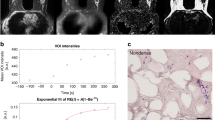
Despite the technological improvement of radiologic, endoscopic and nuclear imaging, the accuracy of diagnostic procedures for tumors can be limited whenever a mass-forming lesion is identified. This is true also because bioptical sampling cannot be properly guided into the lesions so as to puncture neoplastic tissue and to avoid necrotic areas. Under these circumstances, invasive and expensive procedures are still required to obtain diagnosis which is mandatory to plan the most appropriate therapeutic strategy. In order to test if electrical impedance spectroscopy may be helpful in providing further evidence for cancer detection, resistivity measurements were taken on 22 mice, 11 wild‐type and 11 sparc–/– (knock out for the protein SPARC: secreted protein acidic and rich in cysteine), bearing mammary carcinomas, by placing a needle-probe into tumor, peritumoral and contralateral healthy fat areas. Tumor resistivity was significantly lower than both peritumoral fat and contralateral fat tissues. Resistivity in sparc–/– mice was lower than wild-type animals. A significant frequency dependence of resistivity was present in tissues analyzed. We conclude that accurate measurements of resistivity may allow to discriminate between tissues with different pathological and/or structural characteristics. Therefore, resistivity measurements could be considered for in vivo detection and differential diagnosis of tumor masses.



Similar content being viewed by others
References
Aslakson CJ, Miller FR (1992) Selective events in the metastatic process defined by analysis of the sequential dissemination of subpopulations of a mouse mammary tumor. Cancer Res 52:1399–1405
Blad B, Baldetorp B (1996) Impedance spectra of tumour tissue in comparison with normal tissue; a possible clinical application for electrical impedance tomography. Physiol Meas 17:A105–A115
Brown BH, Smallwood RH (1999) Medical physics and biomedical engineering. IOP, London
DeVita, Hellman, and Rosenberg’s cancer: Principles and practice of oncology by Vincent T. DeVita Jr. MD (Editor), Theodore S. Lawrence MD Ph.D. (Editor), Steven A. Rosenberg MD Ph.D. (Editor), Ronald A. DePinho MD (Editor), Robert A. Weinberg Ph.D. (Editor). Ninth Edition, Lippincott, Philadelphia 2011
Dunn OJ (1964) Multiple comparisons using rank sums. Technometrics 6:241–252
Faupel M, Vanel D, Barth V, Davies R, Fentiman IS, Holland R, Lamarque JL, Sacchini V, Schreer I (1997) Electropotential evaluation as a new technique for diagnosis breast lesions. Eur J Radiol 24:33–38
Fricke H, Morse S (1926) The electrical capacity of tumors of the breast. J Cancer Res 10:340–376
Geddes LA, Baker LE (1967) The specific resistance of biological material—A compendium of data for the biomedical engineer and physiologist. Med Biol Eng 5:271–293
Haemmerich D, Staellin ST, Tsai JZ et al (2003) In vivo electrical conductivity of hepatic tumours. Physiol Meas 24:251–260
Halter RJ, Kim Y-J (2014) Toward microendoscopic electrical impedance tomography for intraoperative surgical margin assessment. IEEE Trans Biomed Eng 61(11):2779–2786
Holder DS (2005) Electrical impedance tomography: methods, history and applications. Department of Medical Physics and Bioengineering, University College London, London
Hope TA, Iles SE (2004) Technology review: the use of electric impedance scanning in the detection of breast cancer. Breast Cancer Res 6:69–74
Ivorra A (2003) Bioimpedance monitoring for physicians: an overview. Centre Nacional de Microelectrònica Biomedical Applications Group, pp. 1–35
Ivorra A, Gomez R, Noguera N, Villa R, Sola A, Palacios L, Hotter G, Aguilo J (2009) Minimally invasive silicon probe for electrical impedance measurements in small animals. Biosens Bioelectron 391:391–399
Karki B, Wi H, McEwan A, Kwon H, Oh IT, Woo EJ, Seo JK (2014) Evaluation of a multi-electrode bioimpedance spectroscopy tensor probe to detect the anisotropic conductivity spectra of biological tissues. Meas Sci Technol 25:1–11
Keshtkar A, Salehnia Z, Keshtkar A, Shokouhi B (2012) Bladder cancer detection using electrical impedance technique (tabriz mark 1). Pathol Res Int 2012:470101
Kimura S, Morimoto T, Uyama T, Monden Y, Kinouchi Y, Iritani T (1994) Application of electrical impedance analysis for diagnosis of a pulmonary mass. Chest 105:1679–1682
Kruskal WH, Wallis WA (1952) Use of ranks in one-criterion variance analysis. J Am Stat Assoc 47(260):583–621
Lee BR, Roberts WW, Smith DG, Ko HW, Epstein JI, Lecksell K, Partin AW (1999) Bioimpedance: novel use of a minimally invasive technique for cancer localization in the intact prostate. The Prostate 39:213–218
Mann HB, Whitney DR (1947) On a test of whether one of two random variables is stochastically larger than the other. Ann Math Stat 18:50–60
Mauri G, Chiodoni C, Parenza M, Arioli I, Tripodo C, Colombo MP (2013) Ultrasound-guided intra-tumor injection of combined immunotherapy cures mice from orthotopic prostate cancer. Cancer Immunol Immunother 62:1811–1819
Mishra V, Bouayad H, Schned A, Heaney J, Halter RJ (2012) Electrical impedance spectroscopy for prostate cancer diagnosis. In: 34th Annual international conference of the IEEE EMBS, pp 3258–3261
Nie J, Sage EH (2009) SPARC functions as an inhibitor of adipogenesis. J Cell Commun Signal 3:247–254
Pethig R (1984) Dielectric properties of biological materials: biophysical and medical applications. IEEE Trans Electr Insul 19:453–474
Prakash S, Karnes MP, Sequin EK, West JD, Hitchcock CL, Nichols SD, Bloomston M, Abdel-Misih SR, Schmidt CR, Martin EW Jr, Povoski SP, Subramanian VV (2015) Ex vivo electrical impedance measurements on excised hepatic tissue from human patients with metastatic colorectal cancer. Physiol Meas 36:315–328
Sangaletti S, Stoppacciaro A, Guiducci C, Torrisi MR, Colombo MP (2003) Leukocyte, rather than tumor-produced SPARC, determines stroma and collagen type IV deposition in mammary carcinoma. J Exp Med 198:1475–1485
Schellinga MWM, Vanhouette D, Swinnen M, Cleutjens JP, Debets J, van Leeuwen REW, d’Hooge J, de Werf FV, Carmeliet P, Pinto YM, Sage EH, Heymans S (2008) Absence of SPARC results in increased cardiac rupture and dysfunction after acute myocardial infarction. JEM 1:113–123
Subbhuraam VS, Ng EY, Kaw G, Acharya UR, Chong BK (2012) Evaluation of the efficiency of biofield diagnostic system in breast cancer detection using clinical study results and classifiers. J Med Syst 36(1):15–24
Tukey J (1949) Comparing individual means in the analysis of variance. Biometrics 5(2):99–114
Yan Q, Sage EH (1999) SPARC a matricellular glycoprotein with important biological functions. J Histochem Cytochem 47:1495–1506
Yu D, Jun D, Qing Y, Jianxun Z (2015) Development of a noninvasive electrical impedance probe for minimally invasive tumor localization. Physiol Meas 36:1795–1799
Zou Y, Guo Z (2003) A review of electrical impedance techniques for breast cancer detection. Med Eng Phys 25:79–90
Acknowledgments
We thank Bioengineering Laboratories S.r.l. and in particular Dr. Francesco Greco for the precious support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest, financial or otherwise, are declared by the authors.
Rights and permissions
About this article
Cite this article
Meroni, D., Mauri, G., Bovio, D. et al. Healthy and tumoral tissue resistivity in wild-type and sparc–/– animal models. Med Biol Eng Comput 54, 1949–1957 (2016). https://doi.org/10.1007/s11517-016-1489-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11517-016-1489-6