Abstract
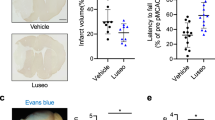
Honokiol, a constituent of Magnolia obovata, has various pharmacological effects, including protection against cerebral ischemia. However, few studies have been conducted to evaluate the possible neuroprotective effects of honokiol against cerebral ischemia. We recently reported that cerebral ischemic neuronal damage could be triggered by glucose intolerance that develops after the onset of ischemic stress (i.e., post-ischemic glucose intolerance). In addition, suppression of post-ischemic glucose intolerance significantly ameliorated ischemic neuronal damage. Here, we investigated the effects of honokiol on the development of post-ischemic glucose intolerance and neuronal damage. Mice were subjected to middle cerebral artery occlusion (MCAO) for 2 h. The development of post-ischemic glucose intolerance on day 1 and neuronal damage on day 3 after MCAO were significantly reduced by intraperitoneal administration of honokiol (10 mg/kg) compared with the vehicle-treated group. Honokiol did not affect serum insulin or adiponectin levels. However, honokiol significantly decreased the expression of phosphoenolpyruvate carboxykinase and increased the expression of 5′-AMP-activated protein kinase (AMPK) on day 1 after MCAO, compared with the vehicle-treated MCAO group. The results of this study suggest that honokiol could prevent post-ischemic glucose intolerance in an AMPK-dependent manner, which may be involved in the neuroprotective effects of honokiol against cerebral ischemia.






Similar content being viewed by others
Change history
03 March 2020
This article [1] has been retracted at the request of the corresponding author because an Investigation Committee established by Kobe Gakuin University (Kobe, Japan) has found numerous discrepancies between the raw data and the data presented in Figs.��6b, d. Statistical analysis of the raw data showed no significant difference between conditions. Authors S. Harada, K. Nakamoto, W. Fujita-Hamabe, H.-H. Chen, M.-H. Chan, and S. Tokuyama agree with this retraction. Authors M. Kishimoto and M. Kobayashi could not be reached for comment about this retraction.
References
World Health Organization (2000) Guidelines for the regulatory assessment of medicinal products for use in self-medication. WHO, Geneva
Suzuki N (2008) Clinical application of dietary supplements. Nippon Yakurigaku Zasshi 131:252–257
Fujita M, Itokawa H, Sashida Y (1973) Studies on the components of Magnolia obovata Thunb. 3. Occurrence of magnolol and honokiol in M. obovata and other allied plants. Yakugaku Zasshi 93:429–434
Liou KT, Shen YC, Chen CF, Tsao CM, Tsai SK (2003) Honokiol protects rat brain from focal cerebral ischemia-reperfusion injury by inhibiting neutrophil infiltration and reactive oxygen species production. Brain Res 992:159–166
Irie T, Miyamoto E, Kitagawa K, Maruyama Y, Inoue K, Inagaki C (2001) An anxiolytic agent, dihydrohonokiol-B, inhibits ammonia-induced increases in the intracellular Cl(−) of cultured rat hippocampal neurons via GABA(c) receptors. Neurosci Lett 312:121–123
Liu B, Hattori N, Zhang NY, Wu B, Yang L, Kitagawa K, Xiong ZM, Irie T, Inagaki C (2005) Anxiolytic agent, dihydrohonokiol-B, recovers amyloid beta protein-induced neurotoxicity in cultured rat hippocampal neurons. Neurosci Lett 384:44–47
Lin YR, Chen HH, Ko CH, Chan MH (2005) Differential inhibitory effects of honokiol and magnolol on excitatory amino acid-evoked cation signals and NMDA-induced seizures. Neuropharmacology 49:542–550
Fukuyama Y, Nakade K, Minoshima Y, Yokoyama R, Zhai H, Mitsumoto Y (2002) Neurotrophic activity of honokiol on the cultures of fetal rat cortical neurons. Bioorg Med Chem Lett 12:1163–1166
Zhai H, Nakade K, Oda M, Mitsumoto Y, Akagi M, Sakurai J, Fukuyama Y (2005) Honokiol-induced neurite outgrowth promotion depends on activation of extracellular signal-regulated kinases (ERK1/2). Eur J Pharmacol 516:112–117
Chen CM, Liu SH, Lin-Shiau SY (2007) Honokiol, a neuroprotectant against mouse cerebral ischaemia, mediated by preserving Na+, K+-ATPase activity and mitochondrial functions. Basic Clin Pharmacol Toxicol 101:108–116
Kagansky N, Levy S, Knobler H (2001) The role of hyperglycemia in acute stroke. Arch Neurol 58:1209–1212
Toni D, De Michele M, Fiorelli M, Bastianello S, Camerlingo M, Sacchetti ML, Argentino C, Fieschi C (1994) Influence of hyperglycaemia on infarct size and clinical outcome of acute ischemic stroke patients with intracranial arterial occlusion. J Neurol Sci 123:129–133
Ennis SR, Keep RF (2007) Effect of sustained-mild and transient-severe hyperglycemia on ischemia-induced blood–brain barrier opening. J Cereb Blood Flow Metab 27:1573–1582
Folbergrova J, Memezawa H, Smith ML, Siesjo BK (1992) Focal and perifocal changes in tissue energy state during middle cerebral artery occlusion in normo- and hyperglycemic rats. J Cereb Blood Flow Metab 12:25–33
Berger L, Hakim AM (1986) The association of hyperglycemia with cerebral edema in stroke. Stroke 17:865–871
Widmer H, Abiko H, Faden AI, James TL, Weinstein PR (1992) Effects of hyperglycemia on the time course of changes in energy metabolism and pH during global cerebral ischemia and reperfusion in rats: correlation of 1H and 31P NMR spectroscopy with fatty acid and excitatory amino acid levels. J Cereb Blood Flow Metab 12:456–468
Dietrich WD, Alonso O, Busto R (1993) Moderate hyperglycemia worsens acute blood–brain barrier injury after forebrain ischemia in rats. Stroke 24:111–116
Harada S, Fujita WH, Shichi K, Tokuyama S (2009) The development of glucose intolerance after focal cerebral ischemia participates in subsequent neuronal damage. Brain Res 1279:174–181
Kahn BB, Alquier T, Carling D, Hardie DG (2005) AMP-activated protein kinase: ancient energy gauge provides clues to modern understanding of metabolism. Cell Metab 1:15–25
Ronnett GV, Ramamurthy S, Kleman AM, Landree LE, Aja S (2009) AMPK in the brain: its roles in energy balance and neuroprotection. J Neurochem 109(Suppl 1):17–23
Hayashi T, Hirshman MF, Kurth EJ, Winder WW, Goodyear LJ (1998) Evidence for 5′ AMP-activated protein kinase mediation of the effect of muscle contraction on glucose transport. Diabetes 47:1369–1373
Merrill GF, Kurth EJ, Hardie DG, Winder WW (1997) AICA riboside increases AMP-activated protein kinase, fatty acid oxidation, and glucose uptake in rat muscle. Am J Physiol 273:E1107–E1112
Choi SS, Cha BY, Iida K, Sato M, Lee YS, Teruya T, Yonezawa T, Nagai K, Woo JT (2011) Honokiol enhances adipocyte differentiation by potentiating insulin signaling in 3T3-L1 preadipocytes. J Nat Med 65:424–430
Alonso-Castro AJ, Zapata-Bustos R, Dominguez F, Garcia-Carranca A, Salazar-Olivo LA (2011) Magnolia dealbata Zucc and its active principles honokiol and magnolol stimulate glucose uptake in murine and human adipocytes using the insulin-signaling pathway. Phytomedicine 18:926–933
Harada S, Hamabe W, Kamiya K, Satake T, Yamamoto J, Tokuyama S (2009) Preventive effect of Morinda citrifolia fruit juice on neuronal damage induced by focal ischemia. Biol Pharm Bull 32:405–409
Shichi K, Fujita-Hamabe W, Harada S, Mizoguchi H, Yamada K, Nabeshima T, Tokuyama S (2011) Involvement of matrix metalloproteinase-mediated proteolysis of neural cell adhesion molecule in the development of cerebral ischemic neuronal damage. J Pharmacol Exp Ther 338:701–710
Harada S, Fujita-Hamabe W, Tokuyama S (2010) The importance of regulation of blood glucose levels through activation of peripheral 5′-AMP-activated protein kinase on ischemic neuronal damage. Brain Res 1351:254–263
Harada S, Fujita-Hamabe W, Tokuyama S (2011) Effect of orexin-A on post-ischemic glucose intolerance and neuronal damage. J Pharmacol Sci 115:155–163
Hall ED, Braughler JM (1989) Central nervous system trauma and stroke. II. Physiological and pharmacological evidence for involvement of oxygen radicals and lipid peroxidation. Free Radic Biol Med 6:303–313
Flamm ES, Demopoulos HB, Seligman ML, Poser RG, Ransohoff J (1978) Free radicals in cerebral ischemia. Stroke 9:445–447
Kitagawa K, Matsumoto M, Oda T, Niinobe M, Hata R, Handa N, Fukunaga R, Isaka Y, Kimura K, Maeda H et al (1990) Free radical generation during brief period of cerebral ischemia may trigger delayed neuronal death. Neuroscience 35:551–558
Yamauchi T, Kamon J, Minokoshi Y, Ito Y, Waki H, Uchida S, Yamashita S, Noda M, Kita S, Ueki K, Eto K, Akanuma Y, Froguel P, Foufelle F, Ferre P, Carling D, Kimura S, Nagai R, Kahn BB, Kadowaki T (2002) Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat Med 8:1288–1295
Mihaylova MM, Vasquez DS, Ravnskjaer K, Denechaud PD, Yu RT, Alvarez JG, Downes M, Evans RM, Montminy M, Shaw RJ (2011) Class IIa histone deacetylases are hormone-activated regulators of FOXO and mammalian glucose homeostasis. Cell 145:607–621
Chiu JH, Ho CT, Wei YH, Lui WY, Hong CY (1997) In vitro and in vivo protective effect of honokiol on rat liver from peroxidative injury. Life Sci 61:1961–1971
Taira J, Ikemoto T, Mimura K, Hagi A, Murakami A, Makino K (1993) Effective inhibition of hydroxyl radicals by hydroxylated biphenyl compounds. Free Radic Res Commun 19(Suppl 1):S71–S77
Teng CM, Chen CC, Ko FN, Lee LG, Huang TF, Chen YP, Hsu HY (1988) Two antiplatelet agents from Magnolia officinalis. Thromb Res 50:757–765
Lo YC, Teng CM, Chen CF, Chen CC, Hong CY (1994) Magnolol and honokiol isolated from Magnolia officinalis protect rat heart mitochondria against lipid peroxidation. Biochem Pharmacol 47:549–553
Son HJ, Lee HJ, Yun-Choi HS, Ryu JH (2000) Inhibitors of nitric oxide synthesis and TNF-alpha expression from Magnolia obovata in activated macrophages. Planta Med 66:469–471
Acknowledgments
Parts of this study were supported by grants-in-aid and by special coordination funds from grants-in-aid for Scientific Research (C) (22500683) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article has been retracted at the request of the corresponding author because an Investigation Committee established by Kobe Gakuin University (Kobe, Japan) has found numerous discrepancies between the raw data and the data presented in figures 6B and 6D. Statistical analysis of the raw data showed no significant difference between conditions. Authors S. Harada, K. Nakamoto, W. Fujita-Hamabe, H.-H. Chen, M.-H. Chan, and S. Tokuyama agree with this retraction. Authors M. Kishimoto and M. Kobayashi could not be reached for comment about this retraction.
About this article
Cite this article
Harada, S., Kishimoto, M., Kobayashi, M. et al. RETRACTED ARTICLE: Honokiol suppresses the development of post-ischemic glucose intolerance and neuronal damage in mice. J Nat Med 66, 591–599 (2012). https://doi.org/10.1007/s11418-011-0623-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-011-0623-x