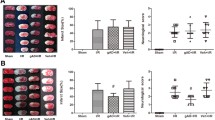
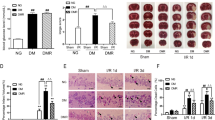
Abstract
Diabetes mellitus substantially increases the risk of stroke and enhances brain’s vulnerability to ischemia insult. In a previous study, Chikusetsu saponin IVa (CHS) pretreatment was proved to protect the brain from cerebral ischemic in normal stroke models. Whether CHS could attenuate cerebral ischemia/reperfusion (I/R) injury in diabetic mice and the possible underlying mechanism are still unrevealed. Male C57BL/6 mice were injected streptozotocin to induce diabetes. After that, the mice were pretreated with CHS for 1 month, and then, focal cerebral ischemia was induced following 24-h reperfusion. The neurobehavioral scores, infarction volumes, and some cytokines in the brain were measured. Apoptosis was analyzed by caspase-3, Bax, and Bcl-2 expression. Downstream molecules of adiponectin (APN) were investigated by Western blotting. The results showed that CHS reduced infarct size, improved neurological outcomes, and inhibited cell injury after I/R. In addition, CHS pretreatment increased APN level and enhanced neuronal AdipoR1, adenosine monophosphate-activated protein kinase (AMPK), and glycogen synthase kinase 3 beta (GSK-3β) expression in a concentration-dependent manner in diabetic mice, and these effects were abolished by APN knockout (KO). In vitro test, CHS treatment also alleviated PC12 cell injury and apoptosis, evidenced by reduced tumor necrosis factor alpha (TNF-α), malondialdehyde (MDA) and caspase-3 expression, and Bax/Bcl-2 ratio in I/R injured cells. Moreover, CHS enhanced AdipoR1, AMPK, and GSK-3β expression in a concentration-dependent manner. Likewise, short interfering RNA (sinRNA) knockdown of liver kinase B1 (LKB1), an upstream kinase of AMPK, reduced the ability of CHS in protecting cells from I/R injury. Furthermore, this LKB1-dependent cellular protection resulted from AdipoR1 and APN activation, as supported by the experiment using sinRNA knockdown of AdipoR1 and APN. Thus, CHS protected brain I/R in diabetes through AMPK-mediated phosphorylation of GSK-3β downstream of APN-LKB1 pathway.









Similar content being viewed by others
References
Kim YM, Namkoong S, Yun YG, Hong HD, Lee YC, Ha KS, Lee H, Kwon HJ, Kwon YG, Kim YM (2007) Water extract of Korean red ginseng stimulates angiogenesis by activating the PI3K/Akt-dependent ERK1/2 and eNOS pathways in human umbilical vein endothelial cells. Biol Pharm Bull 30(9):1674–1679
Tuttle KR, McGill JB, Haney DJ, Lin TE, Anderson PW (2007) Kidney outcomes in long-term studies of ruboxistaurin for diabetic eye disease. Clin J Am Soc Nephrol 2(4):631–636
International Diabetes Federation (2012) IDF diabetes atlas, fifth edition, update 2012. http://www.idf.org/worlddiabetesday/toolkit/gp/factsfigures. Accessed 11 Dec 2013
Melamed E (1976) Reactive hyperglycemia in patients with acute stroke. J Neurol Sci 29(2–4):267–275
Capes SE, Hunt D, Malmberg K, Pathak P, Gerstein HC (2001) Stress hyperglycemia and prognosis of stroke in nondiabetic and diabetic patients: a systematic overview. Stroke 32:2426–2432
Luitse MJ, Biessels GJ, Rutten GE, Kappelle LJ (2012) Diabetes, hyperglycaemia, and acute ischaemic stroke. Lancet Neurol 11(3):261–271
Rehni AK, Nautiyal N, Perez-Pinzon MA, Dave KR (2014) Hyperglycemia/hypoglycemia-induced mitochondrial dysfunction and cerebral ischemic damage in diabetics. Metab Brain Dis. doi:10.1007/s11011-014-9538-z
Chen BH, Jiang DY, Tang LS (2006) Advanced glycation end-products induce apoptosis involving the signaling pathways of oxidative stress in bovine retinal pericytes. Life Sci 79:1040–1048
Mooradian AD (1997) Central nervous system complications of diabetes mellitus—a perspective from the blood-brain barrier. Brain Res Brain Res Rev 23(3):210–218
Duckrow RB, Beard DC, Brennan RW (1985) Regional cerebral blood flow decreases during hyperglycemia. Ann Neurol 17:267–272
OuYang YB, Mellergård P, Kristián T, Kristiánova V, Siesjö BK (1994) Influence of acid-base changes on the intracellular calcium concentration of neurons in primary culture. Exp Brain Res 101(2):265–271
Siesjö BK, Bendek G, Koide T, Westerberg E, Wieloch T (1985) Influence of acidosis on lipid peroxidation in brain tissue in vitro. J Cereb Blood Flow Metab 5:253–258
Yorek MA (2003) The role of oxidative stress in diabetic vascular and neural disease. Free Radic Res 37(5):471–480
Rizk NN, Rafols J, Dunbar JC (2005) Cerebral ischemia induced apoptosis and necrosis in normal and diabetic rats. Brain Res 1053(1–2):1–9
Ichikawa H, Kuriki A, Kinno R, Katoh H, Mukai M, Kawamura M (2012) Occurrence and clinicotopographical correlates of brainstem infarction in patients with diabetes mellitus. J Stroke Cerebrovasc Dis 21(8):890–897
Misra P, Chakrabarti R (2007) The role of AMP kinase in diabetes. Indian J Med Res 125(3):389–398
Mukherjee P, Mulrooney TJ, Marsh J, Blair D, Chiles TC, Seyfried TN (2008) Differential effects of energy stress on AMPK phosphorylation and apoptosis in experimental brain tumor and normal brain. Mol Cancer 7:37
Atherton PJ, Babraj J, Smith K, Singh J, Rennie MJ, Wackerhage H (2005) Selective activation of AMPK-PGC-1alpha or PKB-TSC2-mTOR signaling can explain specific adaptive responses to endurance or resistance training-like electrical muscle stimulation. FASEB J 19:786–788
Arita Y, Kihara S, Ouchi N, Takahashi M, Maeda K, Miyagawa J (1999) Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochem Biophys Res Commun 257:79–83
Ahima RS (2006) Metabolic actions of adipocyte hormones: focus on adiponectin. Obesity (Silver Spring) 14(Suppl 1):9S–15S
Thundyil J, Tang SC, Okun E, Shah K, Karamyan VT, Li YI (2010) Evidence that adiponectin receptor 1 activation exacerbates ischemic neuronal death. Exp Transl Stroke Med 2(1):15
Dash PK, Johnson D, Clark J, Orsi SA, Zhang M, Zhao J, Grill RJ, Moore AN, Pati S (2011) Involvement of the glycogen synthase kinase-3 signaling pathway in TBI pathology and neurocognitive outcome. PLoS One 6(9):e24648
Simao F, Matte A, Pagnussat AS, Netto CA, Salbego CG (2012) Resveratrol prevents CA1 neurons against ischemic injury by parallel modulation of both GSK-3beta and CREB through PI3-K/Akt pathways. Eur J Neurosci 36(7):2899–2905
Gross ER, Hsu AK, Gross GJ (2007) Diabetes abolishes morphine-induced cardioprotection via multiple pathways upstream of glycogen synthase kinase-3beta. Diabetes 56(1):127–136
Horike N, Sakoda H, Kushiyama A, Ono H, Fujishiro M, Kamata H et al (2008) AMP-activated protein kinase activation increases phosphorylation of glycogen synthase kinase 3beta and thereby reduces cAMP-responsive element transcriptional activity and phosphoenolpyruvate carboxykinase C gene expression in the liver. J Biol Chem 283(49):33902–339010
Lee JM, Seo WY, Song KH, Chanda D, Kim YD, Kim DK (2010) AMPK-dependent repression of hepatic gluconeogenesis via disruption of CREB.CRTC2 complex by orphan nuclear receptor small heterodimer partner. J Biol Chem 285(42):32182–32191
Cui J, Li YW, Jia N, Song XM, Duan JL, Weng Y et al (2013) Insulin-secretagogue activity of eleven plant extracts and twelve pure compounds isolated from Aralia taibaiensis. Life Sci 92(2):131–136
Wang Q, Peng Y, Chen S, Gou X, Hu B, Du J, Lu Y, Xiong L (2009) Pretreatment with electroacupuncture induces rapid tolerance to focal cerebral ischemia through regulation of endocannabinoid system. Stroke 40(6):2157–2164
Hata R, Mies G, Wiessner C, Fritze K, Hesselbarth D, Brinker G, Hossmann KA (1998) A reproducible model of middle cerebral artery occlusion in mice: hemodynamic, biochemical, and magnetic resonance imaging. J Cereb Blood Flow Metab 18(4):367–375
Baird TA, Parsons MW, Barber PA, Butcher KS, Desmond PM, Tress BM et al (2002) The influence of diabetes mellitus and hyperglycaemia on stroke incidence and outcome. J Clin Neurosci 9(6):618–626
Williams LS, Rotich J, Qi R, Fineberg N, Espay A, Bruno A, Fineberg SE, Tierney WR (2002) Effects of admission hyperglycemia on mortality and costs in acute ischemic stroke. Neurology 59(1):67–71
Xu Y, Wang L, He J, Bi Y, Li M, Wang T, Jiang Y, Dai M, Lu J, Xu M (2013) Prevalence and control of diabetes in Chinese adults. J Am Med Assoc 310(9):948–959
Remuzzi G, Schieppati A, Ruggenenti P (2002) Clinical practice. Nephropathy in patients with type 2 diabetes. N Engl J Med 346(15):1145–1151
Scheen AJ (2010) Central nervous system: a conductor orchestrating metabolic regulations harmed by both hyperglycaemia and hypoglycaemia. Diabetes Metab 36(Suppl 3):S31–S38
Kamada H, Yu F, Nito C, Chan PH (2007) Influence of hyperglycemia on oxidative stress and matrix metalloproteinase-9 activation after focal cerebral ischemia/reperfusion in rats: relation to blood-brain barrier dysfunction. Stroke 38(3):1044–1049
California Acute Stroke Pilot Registry (CASPR) Investigators (2005) Prioritizing interventions to improve rates of thrombolysis for ischemic stroke. Neurology 64(4):654–659
Weng Y, Yu L, Cui J, Zhu YR, Guo C, Wei G, Duan JL, Yin Y (2014) Antihyperglycemic, hypolipidemic and antioxidant activities of total saponins extracted from Aralia taibaiensis in experimental type 2 diabetic rats. J Ethnopharmacol 152(3):553–560
Serlin Y, Levy J, Shalev H (2011) Vascular pathology and blood-brain barrier disruption in cognitive and psychiatric complications of type 2 diabetes mellitus. Cardiovasc Psychiatry Neurol 2011:609202
King TD, Song L, Jope RS (2006) AMP-activated protein kinase (AMPK) activating agents cause dephosphorylation of Akt and glycogen synthase kinase-3. Biochem Pharmacol 71:1637–1647
Steffens S, Mach F (2008) Adiponectin and adaptive immunity: linking the bridge from obesity to atherogenesis. Circ Res 102(2):140–142
Goldstein BJ, Scalia R (2007) Adipokines and vascular disease in diabetes. Curr Diabetes Rep 7:25–33
Tao L, Gao E, Jiao X, Yuan Y, Li S, Christopher TA et al (2007) Adiponectin cardioprotection after myocardial ischemia/reperfusion involves the reduction of oxidative/nitrative stress. Circulation 115(11):1408–1416
Cheng CF, Lian WS, Chen SH, Lai PF, Li HF (2012) Protective effects of adiponectin against renal ischemia-reperfusion injury via prostacyclin-PPAR- alpha-heme oxygenase-1 signaling pathway. J Cell Physiol 227(1):239–249
Zhang CHZ, Liao Y, Li Q, Chen MG, Zhao Q, Deng RH (2013) Recombinant adiponectin ameliorates liver ischemia reperfusion injury via activating the AMPK/eNOS pathway. PLoS One 8(6):e66382
Kos K, Harte AL, da Silva NF, Tonchev A, Chaldakov G, James S, Snead DR, Hoggart B, O’ Hare JP, McTernan PG, Kumar S (2007) Adiponectin and resistin in human cerebrospinal fluid and expression of adiponectin receptors in the human hypothalamus. J Clin Endocrinol Metab 92(3):1129–1136
Yamauchi T, Kamon J, Ito Y, Tsuchida A, Yokomizo T, Kita S (2003) Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature 423(6941):762–769
Guillod-Maximin E, Roy AF, Vacher CM, Aubourg A, Bailleux V, Lorsignol A et al (2009) Adiponectin receptors are expressed in hypothalamus and colocalized with proopiomelanocortin and neuropeptide Y in rodent arcuate neurons. J Endocrinol 200(1):93–105
Nishimura M, Izumiya Y, Higuchi A, Shibata R, Qiu J, Kudo C et al (2008) Adiponectin prevents cerebral ischemic injury through endothelial nitric oxide synthase dependent mechanisms. Circulation 117(2):216–223
Kadowaki T, Yamauchi T (2005) Adiponectin and adiponectin receptors. Endocr Rev 26(3):439–451
Cai XJ, Chen L, Li L, Feng M, Li X, Zhang K et al (2010) Adiponectin inhibits lipopolysaccharide-induced adventitial fibroblast migration and transition to myofibroblasts via AdipoR1-AMPK-iNOS pathway. Mol Endocrinol 24(1):218–228
Ronnett GV, Ramamurthy S, Kleman AM, Landree LE, Aja S (2009) AMPK in the brain: its roles in energy balance and neuroprotection. J Neurochem 109:17–23
Shaw RJ, Kosmatka M, Bardeesy N, Hurley RL, Witters LA, DePinho RA (2004) The tumor suppressor LKB1 kinase directly activates AMP-activated kinase and regulates apoptosis in response to energy stress. Proc Natl Acad Sci 101:3329–3335
Cline GW, Johnson K, Regittnig W, Perret P, Tozzo E, Xiao L et al (2002) Effects of a novel glycogen synthase kinase-3 inhibitor on insulin-stimulated glucose metabolism in Zucker diabetic fatty (fa/fa) rats. Diabetes 51(10):2903–2910
Xi J, Wang H, Mueller RA, Norfleet EA, Xu Z (2009) Mechanism for resveratrol-induced cardioprotection against reperfusion injury involves glycogen synthase kinase 3beta and mitochondrial permeability transition pore. Eur J Pharmacol 604(1–3):111–116
Collino M, Thiemermann C, Mastrocola R, Gallicchio M, Benetti E, Miglio G et al (2008) Treatment with the glycogen synthase kinase-3beta inhibitor, TDZD-8, affects transient cerebral ischemia/reperfusion injury in the rat hippocampus. Shock 30(3):299–307
Park H, Kam T, Kim Y, Choi H, Gwon Y, Kim C (2012) Neuropathogenic role of adenylate kinase-1 in Ab-mediated tau phosphorylation via AMPK and GSK3b. Hum Mol Genet 21(12):2725–2737
Malhi H, Gores GJ, Lemasters JJ (2006) Apoptosis and necrosis in the liver: a tale of two deaths? Hepatology 43(2 Suppl 1):S31–S44
Acknowledgments
This work was supported by the National Science Foundation of China (Nos. 81173514, 81303264, 81403134, 81403135, 81403182, and 81470174) and Excellent Civil Service Training Fund of Forth Military Medical University (No. 4138C4IDK6).
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jialin Duan, Ying Yin, Jia Cui, Jiajia Yan, and Yanrong Zhu have equally contributed to this work.
Rights and permissions
About this article
Cite this article
Duan, J., Yin, Y., Cui, J. et al. Chikusetsu Saponin IVa Ameliorates Cerebral Ischemia Reperfusion Injury in Diabetic Mice via Adiponectin-Mediated AMPK/GSK-3β Pathway In Vivo and In Vitro. Mol Neurobiol 53, 728–743 (2016). https://doi.org/10.1007/s12035-014-9033-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-9033-x