Abstract
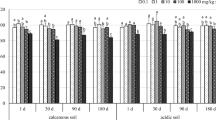
Zinc oxide nanoparticles (ZnO NPs) are used in an array of products and processes, ranging from personal care products to antifouling paints, textiles, food additives, antibacterial agents and environmental remediation processes. Soils are an environment likely to be exposed to manmade nanoparticles due to the practice of applying sewage sludge as a fertiliser or as an organic soil improver. However, understanding on the interactions between soil properties, nanoparticles and the organisms that live within soil is lacking, especially with regards to soil bacterial communities. We studied the effects of nanoparticulate, non-nanoparticulate and ionic zinc (in the form of zinc chloride) on the composition of bacterial communities in soil with a modified pH range (from pH 4.5 to pH 7.2). We observed strong pH-dependent effects on the interaction between bacterial communities and all forms of zinc, with the largest changes in bacterial community composition occurring in soils with low and medium pH levels (pH 4.8 and 5.9). The high pH soil (pH 7.2) was less susceptible to the effects of zinc exposure. At the highest doses of zinc (2500 mg/kg dw soil), both nano and non-nano particulate zinc applications elicited a similar response in the soil bacterial community, and this differed significantly to the ionic zinc salt treatment. The results highlight the importance of considering soil pH in nanotoxicology studies, although further work is needed to determine the exact mechanisms controlling the toxicity and fate and interactions of nanoparticles with soil microbial communities.






Similar content being viewed by others
References
Albanese A, Tang PS, Chan WCW (2012) The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu Rev Biomed Eng 14:1–16
Baek YW, An YJ (2011) Microbial toxicity of metal oxide nanoparticles (CuO, NiO, ZnO, and Sb2O3) to Escherichia coli, Bacillus subtilis, and Streptococcus aureus. Sci Total Environ 409:1603–1608
Bartram AK, Jiang XP, Lynch MDJ, Masella AP, Nicol GW, Dushoff J, Neufeld JD (2014) Exploring links between pH and bacterial community composition in soils from the Craibstone experimental farm. FEMS Microbiol Ecol 87:403–415
Berg JM, Romoser A, Banerjee N, Zebda R, Sayes CM (2009) The relationship between pH and zeta potential of similar to 30 nm metal oxide nanoparticle suspensions relevant to in vitro toxicological evaluations. Nanotoxicology 3:276–283
Blackwood CB, Hudleston D, Zak DR, Buyer JS (2007) Interpreting ecological diversity indices applied to terminal restriction fragment length polymorphism data: insights from simulated microbial communities. Appl Environ Microbiol 73:5276–5283
Brayner R, Ferrari-Iliou R, Brivois N, Djediat S, Benedetti MF, Fievet F (2006) Toxicological impact studies based on Escherichia coli bacteria in ultrafine ZnO nanoparticles colloidal medium. Nano Lett 6:866–870
Cho WS, Duffin R, Thielbeer F, Bradley M, Megson IL, MacNee W, Poland CA, Tran CL, Donaldson K (2012) Zeta potential and solubility to toxic ions as mechanisms of lung inflammation caused by metal/metal oxide nanoparticles. Toxicol Sci 126:469–477
Choi O, Hu ZQ (2008) Size dependent and reactive oxygen species related nanosilver toxicity to nitrifying bacteria. Environ Sci Technol 42:4583–4588
Collins D, Luxton T, Kumar N, Shah S, Walker VK, Shah V (2012) Assessing the impact of copper and zinc oxide nanoparticles on soil: a field study. PLos One 7:e42663
Coradeghini R, Gioria S, Garcia CP, Nativo P, Franchini F, Gilliland D, Ponti J, Rossi F (2013) Size-dependent toxicity and cell interaction mechanisms of gold nanoparticles on mouse fibroblasts. Toxicol Lett 217:205–216
Coutris C, Joner EJ, Oughton DH (2012) Aging and soil organic matter content affect the fate of silver nanoparticles in soil. Sci Total Environ 420:327–333
Dimkpa CO, Calder A, Britt DW, McLean JE, Anderson AJ (2011) Responses of a soil bacterium, Pseudomonas chlororaphis O6 to commercial metal oxide nanoparticles compared with responses to metal ions. Environ Pollut 159:1749–1756
Duster TA, Fein JB (2014) Comparison of the aggregation behavior of TiO2 nanoparticles exposed to fulvic acid and Bacillus subtilis exudates. Water Air Soil Pollut 225:2189
Fang J, Shan XQ, Wen B, Lin JM, Owens G (2009) Stability of titania nanoparticles in soil suspensions and transport in saturated homogeneous soil columns. Environ Pollut 157:1101–1109
Fernandez-Calvino D, Rousk J, Brookes PC, Baath E (2011) Bacterial pH-optima for growth track soil pH, but are higher than expected at low pH. Soil Biol Biochem 43:1569–1575
Fierer N, Jackson RB (2006) The diversity and biogeography of soil bacterial communities. Proc Natl Acad Sci U S A 103:626–631
Ge YG, Schimel JP, Holden PA (2011) Evidence for negative effects of TiO2 and ZnO nanoparticles on soil bacterial communities. Environ Sci Technol 45:1659–1664
Ge Y, Schimel JP, Holden PA (2012) Identification of soil bacteria susceptible to TiO2 and ZnO nanoparticles. Appl Environ Microbiol 78:6749–6758
Ge Y, Priester JH, De Werfhorst LCV, Schimel JP, Holden PA (2013) Potential mechanisms and environmental controls of TiO2 nanoparticle effects on soil bacterial communities. Environ Sci Technol 47:14411–14417
Ge Y, Priester JH, Van de Werfhorst LC, Walker SL, Nisbet RM, An YJ, Schimel JP, Gardea-Torresdey JL, Holden PA (2014) Soybean plants modify metal oxide nanoparticle effects on soil bacterial communities. Environ Sci Technol 48:13489–13496
Gliga AR, Skoglund S, Wallinder IO, Fadeel B, Karlsson HL (2014) Size-dependent cytotoxicity of silver nanoparticles in human lung cells: the role of cellular uptake, agglomeration and Ag release. Part Fibre Toxicol 11:11
Gottschalk F, Sonderer T, Scholz RW, Nowack B (2009) Modeled environmental concentrations of engineered nanomaterials (TiO2, ZnO, Ag, CNT, Fullerenes) for different regions. Environ Sci Technol 43:9216–9222
Gou N, Onnis-Hayden A, Gu AZ (2010) Mechanistic toxicity assessment of nanomaterials by whole-cell-array stress genes expression analysis. Environ Sci Technol 44:5964–5970
Griffiths B, Philippot L (2013) Insights into the resistance and resilience of the soil microbial community. FEMS Microbiol Rev 37:112–129
Griffiths RI, Thomson BC, James P, Bell T, Bailey M, Whiteley AS (2011) The bacterial biogeography of British soils. Environ Microbiol 13:1642–1654
Heggelund LR, Diez-Ortiz M, Lofts S, Lahive E, Jurkschat K, Wojnarowicz J, Cedergreen N, Spurgeon D, Svendsen C (2014) Soil pH effects on the comparative toxicity of dissolved zinc, non-nano and nano ZnO to the earthworm Eisenia fetida. Nanotoxicology 8:559–572
Hernandez-Sierra JF, Ruiz F, Pena DC, Martinez-Gutierrez F, Martinez AE, Guillen Ade J, Tapia-Perez H, Castanon GM (2008) The antimicrobial sensitivity of Streptococcus mutans to nanoparticles of silver, zinc oxide, and gold. Nanomed Nanotechnol Biol Med 4:237–240
Hua J, Vijver MG, Richardson MK, Ahmad F, Peijnenburg WJGM (2014) Particle-specific toxic effects of differently shaped zinc oxide nanoparticles to Zebrafish Embryos (Danio rerio). Environ Toxicol Chem 33:2859–2868
Jiang W, Mashayekhi H, Xing BS (2009) Bacterial toxicity comparison between nano- and micro-scaled oxide particles. Environ Pollut 157:1619–1625
Jones N, Ray B, Ranjit KT, Manna AC (2008) Antibacterial activity of ZnO nanoparticle suspensions on a broad spectrum of microorganisms. FEMS Microbiol Lett 279:71–76
Kemmitt SJ, Wright D, Goulding KWT, Jones DL (2006) pH regulation of carbon and nitrogen dynamics in two agricultural soils. Soil Biol Biochem 38:898–911
Klaine SJ, Alvarez PJJ, Batley GE, Fernandes TF, Handy RD, Lyon DY, Mahendra S, McLaughlin MJ, Lead JR (2008) Nanomaterials in the environment: behavior, fate, bioavailability, and effects. Environ Toxicol Chem 27:1825–1851
Kroll A, Behra R, Kaegi R, Sigg L (2014) Extracellular polymeric substances (EPS) of freshwater biofilms stabilize and modify CeO2 and Ag nanoparticles. PLos One 9:e110709
Kumar A, Pandey AK, Singh SS, Shanker R, Dhawan A (2011a) Engineered ZnO and TiO2 nanoparticles induce oxidative stress and DNA damage leading to reduced viability of Escherichia coli. Free Radic Biol Med 51:1872–1881
Kumar A, Pandey AK, Singh SS, Shanker R, Dhawan A (2011b) Cellular uptake and mutagenic potential of metal oxide nanoparticles in bacterial cells. Chemosphere 83:1124–1132
Lombi E, Donner E, Taheri S, Tavakkoli E, Jamting AK, McClure S, Naidu R, Miller BW, Scheckel KG, Vasilev K (2013) Transformation of four silver/silver chloride nanoparticles during anaerobic treatment of wastewater and post-processing of sewage sludge. Environ Pollut 176:193–197
Lopes S, Ribeiro F, Wojnarowicz J, Lojkowski W, Jurkschat K, Crossley A, Soares AMVM, Loureiro S (2014) Zinc oxide nanoparticles toxicity to Daphnia magna: size-dependent effects and dissolution. Environ Toxicol Chem 33:190–198
Lu X, Weakley AT, Aston DE, Rasco BA, Wang S, Konkel ME (2012) Examination of nanoparticle inactivation of Campylobacter jejuni biofilms using infrared and Raman spectroscopies. J Appl Microbiol 113:952–963
Ma R, Levard C, Marinakos SM, Cheng YW, Liu J, Michel FM, Brown GE, Lowry GV (2012) Size-controlled dissolution of organic-coated silver nanoparticles. Environ Sci Technol 46:752–759
Ma R, Levard C, Judy JD, Unrine JM, Durenkamp M, Martin B, Jefferson B, Lowry GV (2014) Fate of zinc oxide and silver nanoparticles in a pilot wastewater treatment plant and in processed biosolids. Environ Sci Technol 48:104–112
McDonald D, Price MN, Goodrich J, Nawrocki EP, DeSantis TZ, Probst A, Andersen GL, Knight R, Hugenholtz P (2012) An improved Greengenes taxonomy with explicit ranks for ecological and evolutionary analyses of bacteria and archaea. ISME J 6:610–618
Miao AJ, Zhang XY, Luo ZP, Chen CS, Chin WC, Santschi PH, Quigg A (2010) Zinc oxide engineered nanoparticles dissolution and toxicity to marine phytoplankton. Environ Toxicol Chem 29:2814–2822
Mortimer M, Kasemets K, Heinlaan M, Kurvet I, Kahru A (2008) High throughput kinetic Vibrio fischeri bioluminescence inhibition assay for study of toxic effects of nanoparticles. Toxicol in Vitro 22:1412–1417
Mu H, Chen YG (2011) Long-term effect of ZnO nanoparticles on waste activated sludge anaerobic digestion. Water Res 45:5612–5620
Mu H, Chen YG, Xiao ND (2011) Effects of metal oxide nanoparticles (TiO2, Al2O3, SiO2 and ZnO) on waste activated sludge anaerobic digestion. Bioresour Technol 102:10305–10311
Mu H, Zheng X, Chen YG, Chen H, Liu K (2012) Response of anaerobic granular sludge to a shock load of zinc oxide nanoparticles during biological wastewater treatment. Environ Sci Technol 46:5997–6003
Musee N, Thwala M, Nota N (2011) The antibacterial effects of engineered nanomaterials: implications for wastewater treatment plants. J Environ Monit 13:1164–1183
Nair S, Sasidharan A, Rani VVD, Menon D, Nair S, Manzoor K, Raina S (2009) Role of size scale of ZnO nanoparticles and microparticles on toxicity toward bacteria and osteoblast cancer cells. J Mater Sci Mater Med 20:235–241
Negi H, Agarwal T, Zaidi MGH, Goel R (2012) Comparative antibacterial efficacy of metal oxide nanoparticles against Gram negative bacteria. Ann Microbiol 62:765–772
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Henry M, Stevens H, Wagner H (2013) Vegan: Community Ecology Package
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing Vienna, Austria
Read DS, Gweon HS, Bowes MJ, Newbold LK, Field D, Bailey MJ, Griffiths RI (2015) Catchment-scale biogeography of riverine bacterioplankton. ISME J 9:516–526
Roesch LF, Fulthorpe RR, Riva A, Casella G, Hadwin AKM, Kent AD, Daroub SH, Camargo FAO, Farmerie WG, Triplett EW (2007) Pyrosequencing enumerates and contrasts soil microbial diversity. ISME J 1:283–290
Rousk J, Baath E, Brookes PC, Lauber CL, Lozupone C, Caporaso JG, Knight R, Fierer N (2010) Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J 4:1340–1351
Rousk J, Ackermann K, Curling SF, Jones DL (2012) Comparative toxicity of nanoparticulate CuO and ZnO to soil bacterial communities. PLos One 7:e34197
Schwegmann H, Feitz AJ, Frimmel FH (2010) Influence of the zeta potential on the sorption and toxicity of iron oxide nanoparticles on S. cerevisiae and E. coli. J Colloid Interface Sci 347:43–48
Suzuki M, Rappé MS, Giovannoni SJ (1998) Kinetic bias in estimates of coastal picoplankton community structure obtained by measurements of small-subunit rRNA gene PCR amplicon length heterogeneity. Appl Environ Microbiol 64:4522–4529
Tourinho PS, van Gestel CAM, Lofts S, Svendsen C, Soares AMVM, Loureiro S (2012) Metal-based nanoparticles in soil: fate, behavior, and effects on soil invertebrates. Environ Toxicol Chem 31:1679–1692
Waalewijn-Kool PL, Ortiz MD, van Gestel CAM (2012) Effect of different spiking procedures on the distribution and toxicity of ZnO nanoparticles in soil. Ecotoxicology 21:1797–1804
Waalewijn-Kool PL, Ortiz MD, Lofts S, van Gestel CAM (2013) The effect of pH on the toxicity of zinc oxide nanoparticles to Folsomia candida in amended field soil. Environ Toxicol Chem 32:2349–2355
Waalewijn-Kool PL, Rupp S, Lofts S, Svendsen C, van Gestel CAM (2014) Effect of soil organic matter content and pH on the toxicity of ZnO nanoparticles to Folsomia candida. Ecotoxicol Environ Saf 108:9–15
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267
Zhang HZ, Chen B, Banfield JF (2010) Particle size and pH effects on nanoparticle dissolution. J Phys Chem C 114:14876–14884
Zheng XO, Wu R, Chen YG (2011) Effects of ZnO nanoparticles on wastewater biological nitrogen and phosphorus removal. Environ Sci Technol 45:2826–2832
Acknowledgements
The NanoFATE Project CP-FP 247739 (2010–2014) under the 7th Framework Programme of the European Commission (FP7-NMP-ENV-2009, Theme 4) coordinated by C. Svendsen; www.nanofate.eu and DEFRA project CB0460 are acknowledged for financial support. Dr. M. Diez-Ortiz was supported by a Marie Curie Intra-European Fellowship within the 7th European Community Framework Programme (call reference FP7-PEOPLE-2010-IEF, 273207 Nano-Ecotoxicity).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Daniel. S. Read and Marianne Matzke are joint first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure S1
(PDF 103 kb)
Supplementary Figure S2
(PDF 96 kb)
Table S1
(PDF 25 kb)
Rights and permissions
About this article
Cite this article
Read, D.S., Matzke, M., Gweon, H.S. et al. Soil pH effects on the interactions between dissolved zinc, non-nano- and nano-ZnO with soil bacterial communities. Environ Sci Pollut Res 23, 4120–4128 (2016). https://doi.org/10.1007/s11356-015-4538-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-015-4538-z