Abstract
Because the detrimental effects of chromium (Cr) to higher plants have been poorly investigated, the present study was undertaken to verify the toxic attributes of hexavalent chromium [Cr(VI)] to plant mitotic microtubules (MTs), to determine any differential disruption of MTs during mitosis of taxonomically related species and to clarify the relationship between the visualized chromosomal aberrations and the Cr(VI)-induced MT disturbance. For this purpose, 5-day-old uniform seedlings of Vicia faba, Pisum sativum, Vigna sinensis and Vigna angularis, all belonging to the Fabaceae family, were exposed to 250 μM Cr(VI) supplied as potassium dichromate (K2Cr2O7) for 24, 72 and 120 h and others in distilled water serving as controls. Root tip samples were processed for tubulin immunolabelling (for MT visualization) and DNA fluorescent staining (for chromosomal visualization). Microscopic preparations of cell squashes were then examined and photographed by confocal laser scanning microscopy (CLSM). Cr(VI) halted seedling growth turning roots brown and necrotic. Severe chromosomal abnormalities and differential disturbance of the corresponding MT arrays were found in all mitotic phases. In particular, in V. faba MTs were primarily depolymerized and replaced by atypical tubulin conformations, whereas in P. sativum, V. sinensis and V. angularis they became bundled in a time-dependent manner. In P. sativum, the effects were milder compared to those of the other species, but in all cases MT disturbance adversely affected the proper aggregation of chromosomes on the metaphase plate, their segregation at anaphase and organization of the new nuclei at telophase. Cr(VI) is very toxic to seedling growth. The particular effect depends on the exact stage the cell is found at the time of Cr(VI) entrance and is species-specific. Mitotic MT arrays are differentially deranged by Cr(VI) in the different species examined, even if they are taxonomically related, while their disturbance underlies chromosomal abnormalities. Results furthermore support the view that MTs may constitute a reliable, sensitive and universal subcellular marker for monitoring heavy metal toxicity.






Similar content being viewed by others
Notes
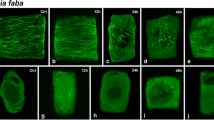
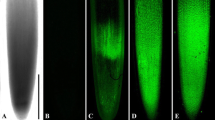
The confocal laser scanning microscopy (CLSM) figures were obtained from seedlings treated with 250 μM Cr(VI) for 24 h, except for Fig. 5g that was treated for 72 h, and of equivalent controls. All CLSM figures are projections of serial optical sections unless otherwise stated. Both tubulin (microtubules—MTs) immunolocalization (green) and DNA staining (red) are shown, either single or merged. When figures of the same cell are used, they are identified by the same letter and numbered consecutively. Cells are placed with their longitudinal axis vertical in accordance with the normal orientation of the root.
References
Adamakis I-DS, Eleftheriou EP, Rost TL (2008) Effects of sodium tungstate on the ultrastructure and growth of pea (Pisum sativum) and cotton (Gossypium hirsutum) seedlings. Environ Exp Bot 63:416–425
Adamakis I-DS, Panteris E, Eleftheriou EP (2010a) Tungsten affects the cortical microtubules of Pisum sativum root cells: experiments on tungsten-molybdenum antagonism. Plant Biol 12:114–124
Adamakis I-DS, Panteris E, Eleftheriou EP (2010b) The cortical microtubules are a universal target of tungsten toxicity among land plant taxa. J Biol Res (Thessaloniki) 13:59–66
Adamakis I-DS, Panteris E, Eleftheriou EP (2011) The fatal effect of tungsten on Pisum sativum L. root cells: indications for endoplasmic reticulum stress-induced programmed cell death. Planta 34:21–34
Barceló J, Poschenriender C, Ruano A, Gunse B (1985) Leaf water potential in Cr(VI) treated bean plants (Phaseolus vulgaris L). Plant Physiol Suppl 77:163–164
Bianchi A, Corradi MG, Tirillini B, Albasini A (1998) Effects of hexavalent chromium on Mentha aquatica L. J Herbs Spices Med Plants 5:3–12
Bishnoi NR, Dua A, Gupta VK, Sawhney SK (1993) Effect of chromium on seed germination, seedling growth and yield of peas. Agric Ecosyst Environ 47:47–57
Botsou F, Karageorgis AP, Dassenakis E, Scoullos M (2011) Assessment of heavy metal contamination and mineral magnetic characterization of the Asopos River sediments (Central Greece). Marine Poll Bull 62:547–563
Degrassi F, Rizzoni M (1982) Micronucleus test in Vicia faba root tips to detect mutagen damage in fresh-water pollution. Mut Res 97:19–23
Dho S, Camusso W, Mucciarelli M, Fusconi A (2010) Arsenate toxicity on the apices of Pisum sativum L. seedling roots: effects on mitotic activity, chromatin integrity and microtubules. Environ Exp Bot 69:17–23
Dovgalyuk A, Kalynyak T, Blume YB (2003) Heavy metals have a different action from aluminium in disrupting microtubules in Allium cepa meristematic cells. Cell Biol Intern 27:193–195
EC Council Directive (98/83/EC) of 3 November 1998 on the quality of water intended for human consumption. Official J Eur Comm L330/32
Economou-Eliopoulos M, Megremi I, Vasilatos C (2011) Factors controlling the heterogeneous distribution of Cr(VI) in soil, plants and groundwater: evidence from the Assopos basin, Greece. Chem Erde 71:39–52
Eleftheriou EP, Adamakis I-DS, Melissa P (2012) Effects of hexavalent chromium on microtubule organization, ER distribution and callose deposition in root tip cells of Allium cepa L. Protoplasma 249:401–416
Eleftheriou EP, Adamakis I-DS, Fatsiou M, Panteris E (2013) Hexavalent chromium disrupts mitosis by stabilizing microtubules in Lens culinaris Moench. root tip cells. Physiol Plant 147:169–180
Eun S-O, Youn HS, Lee Y (2000) Lead disturbs microtubule organization in the root meristem of Zea mays. Physiol Plant 110:356–365
Frantzios G, Galatis B, Apostolakos P (2000) Aluminium effects on microtubule organization in dividing root-tip cells of Triticum turgidum. I. Mitotic cells. New Phytol 145:211–224
Frantzios G, Galatis B, Apostolakos P (2001) Aluminium effects on microtubule organization in dividing root-tip cells of Triticum turgidum. II. Cytokinetic cells. J Plant Res 114:157–170
Fusconi A, Gallo C, Camusso W (2007) Effects of cadmium on root apical meristems of Pisum sativum L.: cell viability, cell proliferation and microtubule pattern as suitable markers for assessment of stress pollution. Mut Res 632:9–19
Hepler P, Hush JM (1996) Behavior of microtubules in living plant cells. Plant Physiol 112:455–461
Hu Y, Liu X, Bai J, Shih K, Zeng EY, Cheng H (2013) Assessing heavy metal pollution in the surface soils of a region that had undergone three decades of intense industrialization and urbanization. Environ Sci Pollut Res 20:6150–6159
Jamal SN, Iqbal MZ, Athar M (2006) Effect of aluminum and chromium on the germination and growth of two Vigna species. Int J Environ Sci Tech 3:53–58
Kimbrough DE, Cohen Y, Winer AM, Creelman L, Mabuni C (1999) A critical assessment of chromium in the environment. Crit Rev Environ Sci Technol 29:1–46
Kirkillis CG, Pasias IN, Miniadis-Meimaroglou S, Thomaidis NS, Zabetakis I (2012) Concentration levels of trace elements in carrots, onions, and potatoes cultivated in Asopos region, Central Greece. Anal Lett 45:551–562
Knasmüller S, Gottmann E, Steinkellner H, Fomin A, Pickl C, Paschke A, Göd R, Kundi M (1998) Detection of genotoxic effects of heavy metal contaminated soils with plant bioassays. Mut Res 420:37–48
Labra M, Grassi F, Imazio S, Di Fabio T, Citterio S, Sgorbati S, Agradi E (2004) Genetic and DNA-methylation changes induced by potassium dichromate in Brassica napus L. Chemosphere 54:1049–1058
Liu D, Xue P, Meng Q, Zou J, Gu J, Jiang W (2009) Pb/Cu effects on the organization of microtubule cytoskeleton in interphase and mitotic cells of Allium sativum L. Plant Cell Rep 28:695–702
Liu J, Zhang X-H, Tran H, Wang D-Q, Zhu Y-N (2013) Heavy metal contamination and risk assessment in water, paddy soil, and rice around an electroplating plant. Environ Sci Pollut Res 18:1623–1632
Ma T-H, Xu Z, Xu C, McConnell H, Rabago EV, Arreola GA, Zhang H (1995) The improved Allium/Vicia root tip micronucleus assay for clastogenicity of environmental pollutants. Mut Res 334:185–195
Malea P, Adamakis I-DS, Kevrekidis T (2013) Microtubule integrity and cell viability under metal (Cu, Ni and Cr) stress in the seagrass Cymodocea nodosa. Chemosphere 93:1035–1042
Mallick S, Sinam G, Kumar Mishra R, Sinha S (2010) Interactive effects of Cr and Fe treatments on plants growth, nutrition and oxidative status in Zea mays L. Ecotoxicol Environ Safety 73:987–995
Morejohn LC (1991) The molecular pharmacology of plant tubulin and microtubules. In: Lloyd CW (ed) The cytoskeletal basis of plant growth and form. Academic, London, pp 29–43
Oliveira H (2012) Chromium as an environmental pollutant: insights on induced plant toxicity. J Bot 2012: ID375843, 8p
Parr PD, Taylor FG Jr (1982) Germination and growth effects of hexavalent chromium in Orocol TL (a corrosion inhibitor) on Phaseolus vulgaris. Environ Int 7:197–202
Peralta JR, Gardea-Torresdey JL, Tiemann KJ, Gomez E, Arteaga S, Rascon E, Parsons JG (2001) Uptake and effects of five heavy metals on seed germination and plant growth in alfalfa (Medicago sativa L.). Bull Environ Contam Toxicol 66:727–734
Perdiz D, Mackeh R, Pous C, Baillet A (2011) The ins and outs of tubulin acetylation: more than just a post-translational modification? Cell Signal 23:763–771
Qian X (2004) Mutagenic effects of chromium trioxide on root tip cells of Vicia faba. J Zhejiang Univ (Sci) 5:1570–1576
Rodriguez E, Azevedo R, Fernandes P, Santos C (2011) Cr(VI) induces DNA damage, cell cycle arrest and polyploidization: a flow cytometric and comet assay study in Pisum sativum. Chem Res Toxicol 24:1040–1047
Rout GR, Samantaray S, Das P (1997) Differential chromium tolerance among eight mungbean cultivars grown in nutrient culture. J Plant Nutr 20:473–483
Samantary S (2002) Biochemical responses of Cr-tolerant and Cr-sensitive mung bean cultivars grown on varying levels of chromium. Chemosphere 47:1065–1072
Singh HP, Mahajan P, Kaur S, Batish DR, Kohli RK (2013) Chromium toxicity and tolerance in plants. Environ Chem Lett 13:229–254
Speranza A, Ferri P, Battistelli M, Falcieri E, Crinelli R, Scoccianti V (2007) Both trivalent and hexavalent chromium strongly alter in vitro germination and ultrastructure of kiwifruit pollen. Chemosphere 66:1165–1174
Speranza A, Taddei AR, Gambelini G, Ovidi E, Scoccianti V (2009) The cell wall of kiwifruit pollen tubes is a target for chromium toxicity: alterations to morphology, callose pattern and arabinogalactan protein distribution. Plant Biol 11:179–193
Truta E, Mihai C, Gherghel D, Vochita G (2014) Assessment of the cytogenetic damage induced by chromium short-term exposure in root tip meristems of barley seedlings. Water Air Soil Poll 225:1993. doi:10.1007/s11270-014-1933-x
Tziritis E, Kelepertzis E, Korres G, Perivolaris D, Repani S (2012) Hexavalent chromium contamination in groundwaters of Thiva basin. Cent Greece Bull Environ Contam Toxicol. doi:10.1007/s00128-012-0831-4
van der Vaart B, Akhmanova A, Straube A (2009) Regulation of microtubule dynamic instability. Biochem Soc Trans 37:1007–1013
Vázquez MD, Poschenrieder C, Barceló J (1987) Chromium VI induced structural and ultrastructural changes in bush bean plants (Phaseolus vulgaris L.). Ann Bot 59:427–438
Wang H (1999) Clastogenicity of chromium contaminated soil samples evaluated by Vicia root-micronucleus assay. Mutat Res 426:147–149
Wick SM (1985) Immunofluorescence microscopy of tubulin and microtubule arrays in plant cells. III. Transition between mitotic/cytokinetic and interphase microtubule arrays. Cell Biol Intern Rep 9:357–371
Xu P, Liu D, Jiang W (2009) Cadmium effects on the organization of microtubular cytoskeleton in interphase and mitotic cells of Allium sativum. Biol Plant 53:387–390
Zeid IM (2001) Responses of Phaseolus vulgaris to chromium and cobalt treatments. Biol Plant 44:111–115
Acknowledgments
This research was financially supported by the Department of Botany, School of Biology, Aristotle University of Thessaloniki, Greece. We are grateful to Dr. Anastasia Tsingotjidou, Faculty of Veterinary Medicine, Aristotle University of Thessaloniki, Greece, for generously providing access to the Nikon D-Eclipse C1 CLSM, and to Dr. Takahiro Hamada (Kyoto University, Japan) for cordially providing seeds of V. angularis.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues.
Rights and permissions
About this article
Cite this article
Eleftheriou, E.P., Michalopoulou, V.A. & Adamakis, ID.S. Aberration of mitosis by hexavalent chromium in some Fabaceae members is mediated by species-specific microtubule disruption. Environ Sci Pollut Res 22, 7590–7599 (2015). https://doi.org/10.1007/s11356-014-3880-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3880-x