Abstract
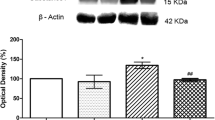
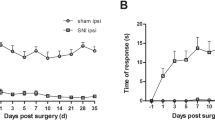
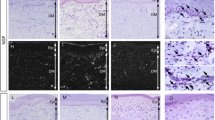
ATP is a ligand of P2X family purinoceptors, and exogenous ATP administration evokes pain behaviors. To date, there is a lack of systematic studies to address relationships between endogenous ATP and neuropathic pain. In this report, we took advantage of a mouse model of resiniferatoxin (RTX)-induced neuropathic pain to address the role of endogenous ATP in neuropathic pain. After RTX administration, endogenous ATP markedly increased in dorsal root ganglia (DRGs) (p < 0.01) and skin tissues (p < 0.001). The excessive endogenous ATP was removed by apyrase, an ATP hydrolyzing enzyme, administration via either a lumbar puncture route (p < 0.001) or an intraplantar injection (p < 0.001), which led to the normalization of neuropathic pain. In addition, intraplantar treatment with apyrase caused mechanical analgesia. Linear analyses showed that the densities of P2X3(+) neurons (r = −0.72, p < 0.0001) and P2X3(+) dermal nerves (r = −0.72, p < 0.0001) were inversely correlated with mechanical thresholds. Moreover, the contents of endogenous ATP in skin tissues were linearly correlated with P2X3(+) dermal nerves (r = 0.80, p < 0.0001) and mechanical thresholds (r = −0.80, p < 0.0001). In summary, this study demonstrated that enhanced purinergic signalling due to an increase in endogenous ATP after RTX-induced nerve injury contributed to the development of neuropathic pain. The data in this report provide a new therapeutic strategy for pain control by targeting the endogenous ligand of purinergic signalling.






Similar content being viewed by others
References
Burnstock G (2009) Purines and sensory nerves. Handb Exp Pharmacol 194:333–392
Burnstock G (2009) Purinergic receptors and pain. Curr Pharm Des 15(15):1717–1735
Burnstock G (2009) Purinergic mechanosensory transduction and visceral pain. Mol Pain 5:69
Tsuchihara T, Ogata S, Nemoto K, Okabayashi T, Nakanishi K, Kato N, Morishita R, Kaneda Y, Uenoyama M, Suzuki S, Amako M, Kawai T, Arino H (2009) Nonviral retrograde gene transfer of human hepatocyte growth factor improves neuropathic pain-related phenomena in rats. Mol Ther 17(1):42–50
Xu GY, Shenoy M, Winston JH, Mittal S, Pasricha PJ (2008) P2X receptor-mediated visceral hyperalgesia in a rat model of chronic visceral hypersensitivity. Gut 57(9):1230–1237
Banerjee B, Medda BK, Schmidt J, Zheng Y, Zhang Z, Shaker R, Sengupta JN (2009) Altered expression of P2X3 in vagal and spinal afferents following esophagitis in rats. Hist Cell Biol 132(6):585–597
Hori K, Ozaki N, Suzuki S, Sugiura Y (2010) Upregulations of P2X(3) and ASIC3 involve in hyperalgesia induced by cisplatin administration in rats. Pain 149(2):393–405
Oliveira MC, Pelegrini-da-Silva A, Tambeli CH, Parada CA (2009) Peripheral mechanisms underlying the essential role of P2X3,2/3 receptors in the development of inflammatory hyperalgesia. Pain 141(1–2):127–134
Hansen RR, Nasser A, Falk S, Baldvinsson SB, Ohlsson PH, Bahl JM, Jarvis MF, Ding M, Heegaard AM (2012) Chronic administration of the selective P2X3, P2X2/3 receptor antagonist, A-317491, transiently attenuates cancer-induced bone pain in mice. Eur J Pharmacol 688(1–3):27–34
Kaan TK, Yip PK, Patel S, Davies M, Marchand F, Cockayne DA, Nunn PA, Dickenson AH, Ford AP, Zhong Y, Malcangio M, McMahon SB (2010) Systemic blockade of P2X3 and P2X2/3 receptors attenuates bone cancer pain behaviour in rats. Brain 133(9):2549–2564
Tsuda M, Koizumi S, Kita A, Shigemoto Y, Ueno S, Inoue K (2000) Mechanical allodynia caused by intraplantar injection of P2X receptor agonist in rats: involvement of heteromeric P2X2/3 receptor signaling in capsaicin-insensitive primary afferent neurons. J Neurosci 20(15):RC90
Shinoda M, Ozaki N, Sugiura Y (2008) Involvement of ATP and its receptors on nociception in rat model of masseter muscle pain. Pain 134(1–2):148–157
Matsuka Y, Ono T, Iwase H, Mitrirattanakul S, Omoto KS, Cho T, Lam YY, Snyder B, Spigelman I (2008) Altered ATP release and metabolism in dorsal root ganglia of neuropathic rats. Mol Pain 4:66
Fujita M, Andoh T, Sasaki A, Saiki I, Kuraishi Y (2010) Involvement of peripheral adenosine 5′-triphosphate and P2X purinoceptor in pain-related behavior produced by orthotopic melanoma inoculation in mice. Eur J Neurosci 31(9):1629–1636
Cook SP, McCleskey EW (2002) Cell damage excites nociceptors through release of cytosolic ATP. Pain 95(1–2):41–47
Hsieh YL, Chiang H, Tseng TJ, Hsieh ST (2008) Enhancement of cutaneous nerve regeneration by 4-methylcatechol in resiniferatoxin-induced neuropathy. J Neuropathol Exp Neurol 67(2):93–104
Hsieh YL, Chiang H, Lue JH, Hsieh ST (2012) P2X3-mediated peripheral sensitization of neuropathic pain in resiniferatoxin-induced neuropathy. Exp Neurol 235(1):316–325
Zimmermann M (1983) Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16(2):109–110
Fairbanks CA (2003) Spinal delivery of analgesics in experimental models of pain and analgesia. Adv Drug Deliv Rev 55(8):1007–1041
Yegutkin GG (2008) Nucleotide- and nucleoside-converting ectoenzymes: important modulators of purinergic signalling cascade. Biochim Biophys Acta 1783(5):673–694
Burnstock G, Knight GE (2004) Cellular distribution and functions of P2 receptor subtypes in different systems. Int Rev Cytol 240:31–304
Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL (1994) Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 53(1):55–63
Courchesne SL, Karch C, Pazyra-Murphy MF, Segal RA (2011) Sensory neuropathy attributable to loss of Bcl-w. J Neurosci 31(5):1624–1634
Tschachler E, Reinisch CM, Mayer C, Paiha K, Lassmann H, Weninger W (2004) Sheet preparations expose the dermal nerve plexus of human skin and render the dermal nerve end organ accessible to extensive analysis. J Invest Dermatol 122(1):177–182
Chen X, Molliver DC, Gebhart GF (2010) The P2Y2 receptor sensitizes mouse bladder sensory neurons and facilitates purinergic currents. J Neurosci 30(6):2365–2372
Malin SA, Davis BM, Koerber HR, Reynolds IJ, Albers KM, Molliver DC (2008) Thermal nociception and TRPV1 function are attenuated in mice lacking the nucleotide receptor P2Y2. Pain 138(3):484–496
Moriyama T, Iida T, Kobayashi K, Higashi T, Fukuoka T, Tsumura H, Leon C, Suzuki N, Inoue K, Gachet C, Noguchi K, Tominaga M (2003) Possible involvement of P2Y2 metabotropic receptors in ATP-induced transient receptor potential vanilloid receptor 1-mediated thermal hypersensitivity. J Neurosci 23(14):6058–6062
Lakshmi S, Joshi PG (2005) Co-activation of P2Y2 receptor and TRPV channel by ATP: implications for ATP induced pain. Cell Mol Neurobiol 25(5):819–832
Wang H, Wang DH, Galligan JJ (2010) P2Y2 receptors mediate ATP-induced resensitization of TRPV1 expressed by kidney projecting sensory neurons. Am J Physiol Regul Integr Comp Physiol 298(6):R1634–1641
Ando RD, Mehesz B, Gyires K, Illes P, Sperlagh B (2010) A comparative analysis of the activity of ligands acting at P2X and P2Y receptor subtypes in models of neuropathic, acute and inflammatory pain. Br J Pharmacol 159(5):1106–1117
Burnstock G (2006) Historical review: ATP as a neurotransmitter. Trends Pharmacol Sci 27(3):166–176
Grafe P, Schaffer V, Rucker F (2006) Kinetics of ATP release following compression injury of a peripheral nerve trunk. Purinergic Signal 2(3):527–536
Mizumoto N, Mummert ME, Shalhevet D, Takashima A (2003) Keratinocyte ATP release assay for testing skin-irritating potentials of structurally diverse chemicals. J Invest Dermatol 121(5):1066–1072
Mizumoto N, Kumamoto T, Robson SC, Sevigny J, Matsue H, Enjyoji K, Takashima A (2002) CD39 is the dominant Langerhans cell-associated ecto-NTPDase: modulatory roles in inflammation and immune responsiveness. Nat Med 8(4):358–365
Attal N, Cruccu G, Baron R, Haanpaa M, Hansson P, Jensen TS, Nurmikko T (2010) EFNS guidelines on the pharmacological treatment of neuropathic pain: 2010 revision. Eur J Neurol 17(9):1113–e1188
O'Connor AB, Dworkin RH (2009) Treatment of neuropathic pain: an overview of recent guidelines. Am J Med 122(10 Suppl):S22–S32
Acknowledgements
This work was supported by grants from the National Science Council (NSC100-2320-B-037-018) and Kaohsiung Medical University Research Foundation (KMU-Q098015, KMU-Q099005, and KMU-M100004).
Conflicts of interests
The authors declare that they have no conflicts of interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lin, CL., Fu, YS., Hsiao, TH. et al. Enhancement of purinergic signalling by excessive endogenous ATP in resiniferatoxin (RTX) neuropathy. Purinergic Signalling 9, 249–257 (2013). https://doi.org/10.1007/s11302-012-9347-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11302-012-9347-y