Abstract
Background
The European Organisation for Research and Treatment of Cancer Quality of Life Core Questionnaire (EORTC QLQ-C30) was evaluated for its psychometric properties in a sample of cancer patients from the culturally distinct South Asian subcontinent, which accounts for a significant proportion of the global cancer burden.
Methods
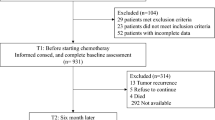
Psychometric testing assessed the hypothesised scale structure, internal consistency, content and construct validity, and acceptability of the Sinhala version of the QLQ-C30 independently in two heterogeneous groups of cancer patients at pretreatment (N = 489) and during treatment (N = 343).
Results
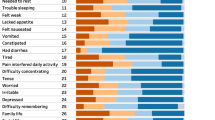
Qualitative feedback from an expert panel assessing content validity recommended measuring family support during illness as an additional, culturally-relevant dimension of health-related quality of life (HRQL). Compliance was high (100%), with little missing data (0.11%). Multitrait scaling results supported the scale structure of the QLQ-C30, with the exception of the cognitive functioning scale, which was also the only scale that did not meet the 0.70 internal consistency criteria in either sample. Interscale correlations were of a moderate size, with conceptually related scales showing higher correlations. All scales were able to discriminate clearly between pre- and current treatment patients (P < 0.01), although results were less consistent when comparing groups formed on the basis of age and disease stage.
Conclusions
Overall psychometric results confirmed the QLQ-C30 as a reliable and valid questionnaire for assessing HRQL of cancer patients in Sri Lanka.



Similar content being viewed by others
References
World Health Organization. (2001). The World health report 2000, health systems: Improving performance. Geneva: WHO.
Ministry of Healthcare. (2006). Annual Health Bulletin 2002. Colombo: Ministry of Healthcare.
Aaronson, N. K. (1995). Quality of life and clinical trials. Lancet, 346, 1–2.
Aaronson, N. K., Ahmedzai, S., Bergman, B., et al. (1993). The European Organisation for Research and Treatment Of Cancer QLQ-C30: A quality-of-life instrument for use in international clinical trials in oncology. Journal of the National Cancer Institute, 85, 365–376.
Young, T., De Haes, H., Curran, D., et al. (2002). On behalf of the European Organisation for Research and Treatment of Cancer (EORTC) quality of life group and quality of life unit. Guidelines for assessing quality of life in EORTC clinical trials (version 2). Brussels: EORTC.
Chie, W. C., Chang, K. J., Huang, C. S., & Kuo, W. H. (2003). Quality of life of breast cancer patients in Taiwan: Validation of the Taiwan Chinese version of the EORTC QLQ-C30 and EORTC QLQ-BR23. Psycho-Oncology, 12(7), 729–735.
Chie, W. C., Hong, R. L., Lai, C. C., Ting L. L., & Hsu M. M. (2003). Quality of life in patients of nasopharyngeal carcinoma: Validation of the Taiwanese Chinese version of the EORTC QLQ-C30 and the EORTC QLQ-H&N35. Quality of Life Research, 12(1), 93–98.
Chie, W. C., Yang, C. H., Hsu, C., & Yang, P. C. (2004). Quality of life of lung cancer patients: Validation of the Taiwan Chinese version of the EORTC QLQ-C30 and QLQ-LC13. Quality of Life Research, 13(1), 257–262.
Kobayashi, K., Takeda, F., Teramukai, S., et al. (1998). A cross-validation of the European Organisation for Research and Treatment of Cancer (EORTC QLQ-C30) for Japanese with lung cancer. European Journal of Cancer, 34(6), 810–815.
Zhao, H., & Kanda, K. (2000). Translation and validation of the Standard Chinese version of the EORTC QLQ-C30. Quality of Life Research, 9, 129–137.
Yun, Y. H., Park, Y. S., Lee, S., et al. (2004). Validation of the Korean version of the EORTC QLQ-C30. Quality of Life Research, 13(4), 863–868.
Jayasekara, H. D. M. H., & Rajapaksa, L. C. (2006). An assessment of quality-of-life and satisfaction with care in patients diagnosed with some common cancers (MD Thesis). Colombo: Postgraduate Institute of Medicine.
Cull, A., Sprangers, M. A. G., Bjordal, K., & Aaronson, N. (1998). On behalf of the European Organisation for Research and Treatment of Cancer quality of life study group. EORTC quality of life study group translation procedure. Brussels: EORTC.
World Health Organization. (1992). International statistical classification of diseases and related health problems (10th revision). Geneva: WHO.
Tabachnik, B. J., & Fidel, L. S. (1993). Using multivariate statistics. London: Harper & Row.
Fayers, P. M., Aaronson, N. K., Bjordal, K., Groenvold, M., Curran, D., & Bottomley, A. (2001). On behalf of the European Organisation for Research and Treatment of Cancer quality of life study group. The EORTC QLQ-C30 scoring manual (3rd ed.). Brussels: EORTC.
Hays, R. D., Hayashi, T., Carson, S., & Ware, J. E. (1988). User’s guide for the multitrait analysis program (MAP). Santa Monica, Carlifornia: Rand Corporation.
Cronbach, L. J. (1951). Coefficient alpha and the internal structure of tests. Psychometrika, 16, 297–334.
Nunnally, J. C., & Bernstein, I. H. (1994). Psychometric theory. USA: McGraw-Hill Inc.
Kerlinger, F. N. (1978). Foundations of behavioural research. New York: McGraw-Hill.
Arraras, J. I., Arias, F., Tejedor, M., et al. (2002). The EORTC QLQ-C30 (version 3.0) quality-of-life questionnaire: Validation study for Spain with head and neck cancer patients. Psycho- Oncology, 11(3), 249–256.
Bjordal, K., & Kaasa, S. (1992). Psychometric validation of the EORTC core quality of life questionnaire, 30-item version and a diagnosis-specific module for head and neck cancer patients. Acta Oncologica, 31(3), 311–321.
Bjordal, K., Hammerlid, E., Ahlner-Elmqvist, M., et al. (1999). Quality of life in head and neck cancer patients: Validation of the European Organisation for Research and Treatment of Cancer quality of life questionnaire-H&N35. Journal of Clinical Oncology, 17(3), 1008–1119.
Montazeri, A., Harirchi, I., Vahdani, M., et al. (1999). The European Organisation for Research and Treatment of Cancer (EORTC QLQ-C30): Translation and validation study of the Iranian version. Supportive Care in Cancer, 7(6), 400–406.
Montazeri, A., Harirchi, I., Vahdani, M., et al. (2000). The EORTC breast cancer-specific quality of life questionnaire (EORTC QLQ-BR23): Translation and validation study of the Iranian version. Quality of Life Research, 9(2), 177–184.
Apolone, G., Filiberti, A., Cifani, S., Ruggiata, R., & Mosconi, P. (1998). Evaluation of the EORTC QLQ-C30 questionnaire: A comparison with SF-36 health survey in a cohort of Italian long-survival cancer patients. Annals of Oncology, 9(5), 549–557.
Dinshaw, K. A., Shastri, S. S., Kurkure, A. P., & Nandakumar, A. (2006). Cancer awareness, prevention and control: Strategies for South Asia. Geneva: International Union Against Cancer (UICC).
Acknowledgements
The authors thank all patients who participated in this study, the officials of the Ministry of Healthcare, and the management and staff of the following institutions for their cooperation: National Cancer Institute, National Hospital of Sri Lanka, Colombo South Teaching Hospital, Sri Jayawardenepura General Hospital, Castle Street Hospital for Women, De Zoysa Maternity Hospital and Dental Institute of Colombo. The dedication of the following persons during the translation procedure is gratefully appreciated: Wasantha Gunathunga, Anil Samaranayake, Thusitha Malalasekara, Mohan Seevaratnam, Steve Cockeram, and Karen West (Translation Coordinator at EORTC QLU). Neil Thalagala is acknowledged for his statistical assistance. The study was funded by the World Health Organization Country Office for Sri Lanka through Agreement for Performance of Work no. SE/SRL OSD 001/RB 04/EC 1/P1/A2.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jayasekara, H., Rajapaksa, L.C. & Aaronson, N.K. Quality of life in cancer patients in South Asia: psychometric properties of the Sinhala version of the EORTC QLQ-C30 in cancer patients with heterogeneous diagnoses. Qual Life Res 17, 783–791 (2008). https://doi.org/10.1007/s11136-008-9345-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-008-9345-2