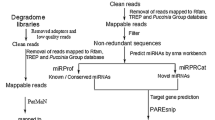
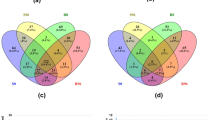
Abstract
Bacterial wilt (BW) disease is a widespread disease in peanut (Arachis hypogaea L.), reducing peanut production because of high susceptibility of most cultivated species. However, Arachis glabrata, a perennial wild relative, has high resistance to soil-borne diseases. MicroRNAs (miRNAs) have been characterized as important factors in plant growth, development, and biotic/abiotic stress responses. However, their roles in response to pathogen are still unknown in peanut. Here, we constructed four miRNA libraries from A. hypogaea and A. glabrata and identified 155 known miRNAs, 31 novel miRNAs, and 173 plausible candidates of novel miRNAs. After infected with BW, the expression of many known miRNA was induced or inhibited in both cultivated and wild-type peanuts. Many miRNAs displayed differential expression patterns in these two species, and the expression of some miRNAs was confirmed by quantitative real-time PCR (qRT-PCR) analysis. A total of 309 targets were predicted, among which 74 of them were confirmed by degradome sequencing. More than 10 % of the targets were defense response genes, such as hypersensitive-induced response protein, leucine-rich repeat (LRR) receptor-like serine/threonine-protein kinase, GRAS, aquaporin, lipid transfer protein, ARF, MYB transcription factors, and MLP-like protein. Our results indicate that diverse set of miRNAs of cultivated and wild peanut species is responsive to BW infection and suggests a possible role in peanut disease resistance.







Similar content being viewed by others
References
Addo-Quaye C, Eshoo TW, Bartel DP, Axtell MJ (2008) Endogenous siRNA and miRNA targets identified by sequencing of the Arabidopsis degradome. Curr Biol 18:758–762. doi:10.1016/j.cub.2008.04.042
Arenas-Huertero C, Pérez B, Rabanal F, Blanco-Melo D, De la Rosa C, Estrada-Navarrete G, Sanchez F, Covarrubias AA, Reyes JL (2009) Conserved and novel miRNAs in the legume Phaseolus vulgaris in response to stress. Plant Mol Biol 70:385–401. doi:10.1007/s11103-009-9480-3
Baltensperger DD, Prine GM, Dunn RA (1986) Root-knot nematode resistance in Arachis glabrata. Peanut Sci 13:78–80
Banks DJ (1969) Breedlng for northern root knolneode Melordogyne hapla reslstance in peanuts. J Am Peanut Res Educ Assoc 1:23–28
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 2004(116):281–297
Bazzini AA, Hopp HE, Beachy RN, Asurmendi S (2007) Infection and coaccumulation of tobacco mosaic virus proteins alter microRNA levels, correlating with symptom and plant development. PNAS 104:12157–12162
Borel C, Antonarakis SE (2008) Functional genetic variation of human miRNAs and phenotypic consequences. Mamm Genome 19:503–509. doi:10.1007/s00335-008-9137-6
Chen XM (2004) A microRNA as a translational repressor of APETALA2 in Arabidopsis flower development. Science 303:2022–2025
Chen H, Li Z, Xiong L (2012) A plant microRNA regulates the adaptation of roots to drought stress. FEBS Lett 586:1742–1747. doi:10.1016/j.febslet.2012.05.013
Chi X, Yang Q, Chen X, Wang J, Pan L, Chen M, Yang Z, He Y, Liang X, Yu S (2011) Identification and characterization of microRNAs from peanut (Arachis hypogaea L.) by high-throughput sequencing. PLoS One 6:e27530
Dai X, Zhao PX (2011) psRNATarget: a plant small RNA target analysis server. Nucleic Acids Res 39:W155–W159
Dortay H, Gruhn N, Pfeifer A, Schwerdtner M, Schmuelling T, Heyl A (2008) Toward an interaction map of the two-component signaling pathway of Arabidopsis thaliana. J Proteome Res 7:3649–3660
Feng H, Duan X, Zhang Q, Li X, Wang B, Huang L, Wang X, Kang Z (2013) The target gene of tae-miR164, a novel NAC transcription factor from the NAM subfamily, negatively regulates resistance of wheat to stripe rust. Mol Plant Pathol 16. doi: 10.1111/mpp.12089
French ER, Gutarra L, Aley P, Elphinstone J (1995) Culture media for Ralstonia solanacearum isolation, identification and maintenance. Fitopatologia 30:126–130
Gao P, Bai X, Yang L, Lv D, Li Y, Cai H, Ji W, Guo D, Zhu Y (2010) Over-expression of osa-MIR396c decreases salt and alkali stress tolerance. Planta 231:991–1001. doi:10.1007/s00425-010-1104-2
Gao P, Bai X, Yang L, Lv D, Pan X, Li Y, Cai H, Ji W, Chen Q, Zhu Y (2011) osa-MIR393: a salinity- and alkaline stress-related microRNA gene. Mol Biol Rep 38:237–242. doi:10.1007/s11033-010-0100-8
German MA, Pillay M, Jeong DH, Hetawal A, Luo S, Janardhanan P, Kannan V, Rymarquis LA, Nobuta K, German R, De Paoli E, Lu C, Schroth G, Meyers BC, Green PJ (2008) Global identification of microRNA-target RNA pairs by parallel analysis of RNA ends. Nat Biotechnol 26:941–946. doi:10.1038/nbt1417
Gong J, Tong Y, Zhang HM, Wang K, Hu T, Shan G, Sun J, Guo AY (2012) Genome-wide identification of SNPs in microRNA genes and the SNP effects on microRNA target binding and biogenesis. Hum Mutat 33(1):254–263. doi:10.1002/humu.21641
Griffiths-Jones S, Saini HK, van Dongen EAJ (2008) miRBase: tools for microRNA genomics. Nucleic Acids Res 36:D154–D158
Guo L, Lu Z (2010) Global expression analysis of miRNA gene cluster and family based on isomiRs from deep sequencing data. Comput Biol Chem 34:165–171. doi:10.1016/j.compbiolchem.2010.06.001
Hafner M, Landgraf P, Ludwig J, Rice A, Ojo T, Lin C, Holoch D, Lim C, Tuschl T (2008) Identification of microRNAs and other small regulatory RNAs using cDNA library sequencing. Methods 44:3–12
Herbert TT, Stalker HT (1981) Resistance to peanut stunt virus in cultivated and wild Arachis species. Peanut Sci 8:45–57
Jones-Rhoades MW, Bartel DP (2004) Computational identification of plant microRNAs and their targets, including a stress-induced miRNA. Mol Cell 14:787–799
Kantar M, Lucas SJ, Budak H (2011) miRNA expression patterns of Triticum dicoccoides in response to shock drought stress. Planta 233:471–484. doi:10.1007/s00425-010-1309-4
Katawczik M, Mila AL (2012) Plant age and strain of Ralstonia solanacearum affect the expression of resistance of tobacco cultivars to granville wilt. Tob Sci 49:8–13
Lan Y, Su N, Shen Y, Zhang R, Wu F, Cheng Z, Wang J, Zhang X, Guo X, Lei C, Wang J, Jiang L, Mao L, Wan J (2012) Identification of novel MiRNAs and MiRNA expression profiling during grain development in indica rice. BMC Genomics 13:264, 21
Lavia GI, Ortiz AM, Robledo G, Fernández A, Seijo G (2011) Origin of triploid Arachis pintoi (Leguminosae) by autopolyploidy evidenced by FISH and meiotic behaviour. Ann Bot 108:103–111. doi:10.1093/aob/mcr108
Li F, Pignatta D, Bendix C, Brunkard JO, Cohn MM, Tung J, Sun H, Kumar P, Baker B (2012) MicroRNA regulation of plant innate immune receptors. PNAS 109:1790–1795. doi:10.1073/pnas.1118282109
Li Y, Lu Y, Shi Y, Wu L, Xu Y, Huang F, Guo X, Zhang Y, Fang J, Zhao Q, Zhang H, Xu P, Zhou J, Wu X, Wang P, Wang W (2014) Multiple rice microRNAs are involved in immunity against the blast fungus Magnaporthe oryzae. Plant Physiol 164:1077–1092
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Lu LF, Liston A (2009) MicroRNA in the immune system, microRNA as an immune system. Immunology 127:291–298
Lu S, Sun YH, Amerson H, Chiang VL (2007) MicroRNAs in loblolly pine (Pinus taeda L.) and their association with fusiform rust gall development. Plant J 51:1077–1098
Meyers BC, Axtell MJ, Bartel B, Bartel DP, Baulcombe D, Bowman JL, Cao X, Carrington JC, Chen X, Green PJ, Griffiths-Jones S, Jacobsen SE, Mallory AC, Martienssen RA, Poethig RS, Qi Y, Vaucheret H, Voinnet O, Watanabe Y, Weigel D, Zhu JK (2008) Criteria for annotation of plant MicroRNAs. Plant Cell 20:3186–3190. doi:10.1105/tpc.108.064311
Milla SR, Isleib TG, Stalker HT (2005) Taxonomic relationships among Arachis sect. Arachis species as revealed by AFLP markers. Genome 48:1–11
Moldovan D, Spriggs A, Yang J, Pogson BJ, Dennis ES, Wilson IW (2010) Hypoxia-responsive microRNAs and trans-acting small interfering RNAs in Arabidopsis. J Exp Bot 61:165–177
Naqvi AR, Haq QM, Mukherjee SK (2010) MicroRNA profiling of tomato leaf curl New Delhi virus (tolcndv) infected tomato leaves indicates that deregulation of mir159/319 and mir172 might be linked with leaf curl disease. Virol J 7:281. doi:10.1186/1743-422X-7-281
Ortiz MA, Guillermo Seijo J, Fernandez A, Lavia GI (2011) Meiotic behavior and pollen viability of tetraploid Arachis glabrata and A. nitida species (Section Rhizomatosae, Leguminosae): implications concerning their polyploid nature and seed set production. Plant Syst Evol 292:73–78
Padmanabhan C, Zhang X, Jin H (2009) Host small RNAs are big contributors to plant innate immunity. Curr Opin Plant Biol 12:465–472
Palatnik JF, Allen E, Wu X, Schommer C, Schwab R, Carrington JC, Weigel D (2003) Control of leaf morphogenesis by microRNAs. Nature 425:257–263
Pande S, Narayana Rao J (2001) Resistance of wild Arachis species to late leaf spot and rust in greenhouse trials. Plant Dis 85:851–855
Pantaleo V, Szittya G, Moxon S, Miozzi L, Moulton V, Dalmay T, Burgyan J (2010) Identification of grapevine microRNAs and their targets using high-throughput sequencing and degradome analysis. Plant J 62:960–976. doi:10.1111/j.0960-7412.2010.04208.x
Rajagopalan R, Vaucheret H, Trejo J, Bartel DP (2006) A diverse and evolutionarily fluid set of microRNAs in Arabidopsis thaliana. Genes Dev 20:3407–3425
Ratnaparkhe MB, Wang X, Li J, Compton RO, Rainville LK, Lemke C, Kim C, Tang H, Paterson AH (2011) Comparative analysis of peanut NBS-LRR gene clusters suggests evolutionary innovation among duplicated domains and erosion of gene microsynteny. New Phytol 192:164–178. doi:10.1111/j.1469-8137.2011.03800.x
Rhoades MW, Reinhart BJ, Lim LP, Burge CB, Bartel B, Bartel DP (2002) Prediction of plant microRNAs targets. Cell 110:513–520
Seijo G, Lavia GI, Fernández A, Krapovickas A, Ducasse DA, Bertioli DJ, Moscone EA (2007) Genomic relationships between the cultivated peanut (Arachis hypogaea, Leguminosae) and its relatives revealed by double GISH. Am J Bot 94:1963–1971
Shivaprasad PV, Chen HM, Patel K, Bond DM, Santos BA, Baulcombe DC (2012) A microRNA superfamily regulates nucleotide binding site-leucine-rich repeats and other mRNAs. Plant Cell 24:859–874
Singh AK, Smartt J (1998) The genome donors of the groundnut/ peanut (Arachis hypogaea L.) revisited. Genet Resour Crop Evol 45:113–118
Subrahmanyam P, Moss JP, Rao VR (1983) Resistance to peanut rust in wild Arachis species. Plant Dis 67:209–212
Subrahmanyam P, Ghanekar AM, Nolt BL, Reddy DVR, McDonald D (1985) Resistance to groundnut diseases in wild Arachis species. In Proceedings of the International Workshop on Cytogenetics of Arachis. 49–55
Subramanian S, Fu Y, Sunkar R, Barbazuk WB, Zhu JK, Yu O (2008) Novel and nodulation-regulated microRNAs in soybean roots. BMC Genomics 9:160
Sunkar R, Zhu JK (2004) Novel and stress-regulated microRNAs and other small RNAs from Arabidopsis. Plant Cell 16:2001–2019
Wang YX (2013) Characterization of a novel Medicago sativa NAC transcription factor gene involved in response to drought stress. Mol Biol Rep 40:6451–6458. doi:10.1007/s11033-013-2760-7
Wang CT, Wang XZ, Tang YY, Chen DZ, Cui FG, Zhang JC, Yu SL (2009) Field screening of groundnut genotypes for resistance to baterial wilt in Shandong province in China. J SAT Agric Res 7:1–7
Weech MH, Chapleau M, Pan L, Ide C, Bede JC (2008) Caterpillar saliva interferes with induced Arabidopsis thaliana defence responses via the systemic acquired resistance pathway. J Exp Bot 59:2437–2448. doi:10.1093/jxb/ern108
Xin M, Wang Y, Yao Y, Xie C, Peng H, Ni Z, Sun Q (2010) Diverse set of microRNAs are responsive to powdery mildew infection and heat stress in wheat (Triticum aestivum L.). BMC Plant Biol 10:123
Yao YY, Guo GG, Ni ZF, Sunkar R, Du JK, Zhu JK, Sun QX (2007) Cloning and characterization of microRNAs from wheat (Triticum aestivum L.). Genome Biol 8:R96
Zhai J, Jeong DH, De Paoli E, Park S, Rosen BD, Li Y, González AJ, Yan Z, Kitto SL, Grusak MA, Jackson SA, Stacey G, Cook DR, Green PJ, Sherrier DJ, Meyers BC (2011) MicroRNAs as master regulators of the plant NB-LRR defense gene family via the production of phased, trans-acting siRNAs. Genes Dev 25:2540–2553. doi:10.1101/gad.177527.111
Zhang L, Chia JM, Kumari S, Stein JC, Liu Z, Narechania A, Maher CA, Guill K, McMullen MD, Ware D (2009) A genome-wide characterization of microRNA genes in maize. PLoS Genet 5(11):e1000716
Zhang W, Gao S, Zhou X, Chellappan P, Chen Z, Zhou X, Zhang X, Fromuth N, Coutino G, Coffey M, Jin H (2011a) Bacteria-responsive microRNAs regulate plant innate immunity by modulating plant hormone networks. Plant Mol Biol 75:93–105
Zhang X, Zou Z, Gong P, Zhang J, Ziaf K, Li H, Xiao F, Ye Z (2011b) Over-expression of microRNA169 confers enhanced drought tolerance to tomato. Biotechnol Lett 33:403–409. doi:10.1007/s10529-010-0436-0
Zhang Q, Zhao CZ, Li M, Sun M, Liu Y, Xia H, Sun MN, Li AQ, Li CS, Zhao SZ, Hou L, Picimbon JF, Wang XJ, Zhao YX (2013) Genome-wide identification of Thellungiella salsuginea microRNAs with putative roles in the salt stress response. BMC Plant Biol 13:180. doi:10.1186/1471-2229-13-180
Zhao CZ, Xia H, Frazier TP, Yao YY, Bi YP, Li AQ, Li MJ, Li CS, Zhang BH, Wang XJ (2010) Deep sequencing identifies novel and conserved microRNAs in peanuts (Arachis hypogaea L.). BMC Plant Biol 10:3
Zuker M (2003) Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res 31:3406–3415
Acknowledgments
This work is supported by the National Natural Science Foundation of China (31101427), grants from the Initial Special Research for 973 Program of China (2012CB126313), Ministry of Science and Technology of China (2013AA102602, 2011BAD35B04, 2012BAD33B07), Shandong Province Taishan Scholar Foundation (tshw20100416), Shandong Province Germplasm Innovation and Utilization Project and grants from Shandong Province (ZR2010CZ002, ZR2010CQ008, 201103023), Young Talents Training Program of Shandong Academy of Agricultural Sciences.
Conflict of Interests
The authors declare that they have no competing interests.
Authors’ contributions
XW and XZ designed the study. CZ, HX, TC, YY, SZ, LH, YZ, and CL carried out most of the experiments and data analysis and wrote the material and method part of the manuscript. XW and XZ wrote the manuscript, made the figures, and finalized the tables.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Chuanzhi Zhao and Han Xia contributed equally to this paper.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
Hairpin structures of known miRNAs. (PDF 1103 kb)
Fig. S2
Hairpin structures of novel miRNAs. (PDF 665 kb)
Table S1
High quality reads generated from four peanut miRNA libraries. (DOC 37 kb)
Table S2
Detail information of known miRNA families of wild and cultivated peanut. (XLS 81 kb)
Table S3
Known miRNA variants identified in wild and cultivated peanut. (XLS 149 kb)
Table S4
The plausible candidates of novel miRNAs identified in wild and cultivated peanut. (XLS 99 kb)
Table S5
Predicted target genes of known and novel miRNAs. (XLS 77 kb)
Rights and permissions
About this article
Cite this article
Zhao, C., Xia, H., Cao, T. et al. Small RNA and Degradome Deep Sequencing Reveals Peanut MicroRNA Roles in Response to Pathogen Infection. Plant Mol Biol Rep 33, 1013–1029 (2015). https://doi.org/10.1007/s11105-014-0806-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-014-0806-1