Abstract
Purpose
An empirical pharmacodynamic model was developed to assess the effect of recombinant human erythropoietin (rHu-EPO) treatment on the reticulocyte production rate and lifespan distribution.
Materials and Methods
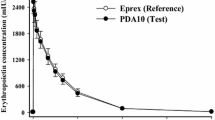
Single doses of rHu-EPO at levels 20, 40, 60, 90, 120, and 160 kIU were administered to healthy volunteers (n = 8 per dose level). Erythropoietin plasma concentrations as well as hematologic responses were measured up to 42 days. The hematological data were used to determine explicit relationships between reticulocyte and red blood cell counts (RBC) and the reticulocytes’ production rate and lifespan distribution.
Results
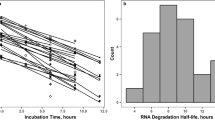
The parameter estimates obtained by simultaneous fitting of the model to the reticulocyte and RBC data revealed that rHu-EPO transiently increased the reticulocyte lifespan from the baseline value of 1.7 days to 3.4 days and the effect lasted for 8.3 days. The dose dependent increase in the reticulocyte production had the maximal value of 77.5 109 cells/l/day and was followed by a rebound that was less than 9% of the baseline value. Both reticulocyte and RBC responses were preceded by a dose-independent lag time of 1.7 days.
Conclusions
The effect of rHu-EPO on the reticulocyte production rate and lifespan distribution was characterized. The results of the present study can be further utilized in building more mechanistic pharmacodynamic models of rHu-EPO stimulatory effects.






Similar content being viewed by others
Abbreviations
- χ(t):
-
Jump function
- C EPO :
-
rHu-EPO plasma concentration
- CF:
-
Conversion factor for reticulocytes
- Δk R(t):
-
Change of k R(t) from the baseline value
- Δk R1 :
-
Increase in reticulocyte production rate due to rHu-EPO
- Δk R2 :
-
Decrease in reticulocyte production rate following exposure to rHu-EPO
- ΔRET(t):
-
Change of the reticulocyte count from the baseline value
- ΔRBC(t):
-
Change of RBC count from the baseline value
- ΔT :
-
Increase in the reticulocyte lifespan due to rHu-EPO treatment
- ΔT 1 :
-
Duration of the rHu-EPO effect on reticulocyte lifespan distribution
- ΔT 2 :
-
Duration of the rebound in reticulocyte production
- δ(τ):
-
Dirac delta function
- D :
-
Dose
- ε :
-
Residual error
- E :
-
Effect
- E 0 :
-
Baseline effect
- E max :
-
Maximal effect
- ED50 :
-
Dose eliciting 50% of the maximal effect
- INT(z):
-
The integral part of the number z
- kout(t):
-
Reticulocyte elimination rate
- kR(t):
-
Reticulocyte production rate
- k R0 :
-
Baseline reticulocyte production rate
- k R1 :
-
Reticulocyte production rate during rHu-EPO stimulation
- k R2 :
-
Reticulocyte production rate during the rebound
- ℓ(t,τ):
-
Probability density function for reticulocyte lifespan distribution at time t
- ℓ0(τ):
-
Time independent probability density function for reticulocyte lifespan distribution
- MRBC:
-
Mature red blood cells; mature red blood cell count
- MRBC0 :
-
Baseline mature red blood cell count
- N :
-
Integer part of the ratio t/T RET0
- p.d.f.:
-
Probability density function
- RBC:
-
Red blood cells
- RBC0 :
-
Baseline red blood cell count
- RET:
-
Reticulocytes; reticulocyte count
- RET0 :
-
Baseline reticulocyte count
- RHu-EPO:
-
Recombinant human erythropoietin
- σ :
-
Standard deviation of the residual error
- τ :
-
Reticulocyte lifespan
- T 0 :
-
Lag time between rHu-EPO administration and its effect
- T 1 :
-
Time at which the effect of rHu-EPO on the reticulocyte lifespan distribution stops
- T 2 :
-
Time at which the rebound ends
- T RET :
-
Reticulocyte lifespan
- T RET0 :
-
Baseline reticulocyte lifespan
- T RBC :
-
Red blood cell lifespan
- t max :
-
Observed reticulocyte count peak time
- UIR(t):
-
Unit impulse response
References
J. W. Fisher. Erythropoietin: physiology and pharmacology update. Exp. Biol. Med. 228:1–14 (2003).
W. K. Cheung, B. L. Goon, M. C. Guilfoyle, and M. C. Wacholtz. Pharmacokinetics and pharmacodynamics of recombinant human erythropoietin after single and multiple subcutaneous doses to healthy subjects. Clin. Pharmacol. Ther. 64:412–423 (1998).
N. Hayashi, K. P. Zuideveld, P. Jordan, and R. Gieschke. A mechanism-based PK/PD model predicts the time course of hematological responses for epoetin beta. PAGE 12 (2003) Abstr 396, (www.page-meeting.org/?abstract=396).
R. Ramakrishnan, W. K. Cheung, M. C. Wacholtz, N. Minton, and W. J. Jusko. Pharmacokinetic and pharmacodynamic modeling of recombinant human erythropoietin after single and multiple doses in healthy volunteers. J. Clin. Pharmacol. 44:991–1002 (2004).
D. E. Uehlinger, F. A. Gotch, and L. B. Sheiner. A pharmacodynamic model of erythropoietin therapy for uremic anemia. Clin. Pharmacol. Therap. 51:76–89 (1992).
A. C. Heatherington, D. Henry, R. Patel, N. S. Tchekmedyian, R. Berg, M. Austin, G. Rossi, and J. Glaspy. The impact of timing of chemotherapy relative to darbepoetin alfa (DA) on the pharmacokinetics (PK) and hematologic effects. Clin. Pharm. Ther. 75:P60 (2004).
A. Major, C. Bauer, C. Breyman, A. Huch, and R. Huch. rh-Erythropoietin stimulates immature reticulocyte release in man. Br. J. Haematol. 87:605–608 (1994).
N. H. Al-Huniti, J. A. Widness, R. L. Schmidt, and P. Veng-Pedersen. Pharmacodynamic analysis of changes in reticulocyte subtype distribution in phlebotomy-induced stress erythropoiesis. J. Pharmacokinet. Pharmacodyn. 32:359–376 (2005).
A. Rescigno, and G. Segre. Drug and Tracer Kinetics. Blaisdell, Waltham, MA, 1966.
M. Loeffler, K. Pantel, H. Wulff, and H. E. Wichmann. A mathematical model of erythropoiesis in mice and rats, Part 1: structure of the model. Cell Tissue Kinet. 22:13–30 (1989).
W. Krzyzanski, R. Ramakrishnan, and W. J. Jusko. Basic pharmacodynamic models for agents that alter production of natural cells. J. Pharmacokin. Biopharm. 27:467–489 (1999).
W. Krzyzanski, S. Woo, and W. J. Jusko. Pharmacodynamic models for agents that alter productions of natural cells with various distributions of lifespans. J. Pharmacokinet. Pharmacodyn. 33:125–166 (2006).
S. Neelakantan, J. Widness, R. Schmidt, P. and Veng-Pedersen. Novel deconvolution method for model-independent direct analysis of cellular production and transformation rates. AAPS J. 7 S2: Abstract T2330 (2005).
L. Ratnasingam. An open-label, randomized, parallel-design study to investigate the pharmacokinetic and pharmacodynamic profiles of single subcutaneously administered doses of epoetin alfa in healthy male volunteers. Protocol EPO-PHI-380. Data on file, Johnson & Johnson Pharmaceutical Research and Development, (2001).
S. L. Beal and L. B. Sheiner. NONMEM Users Guide. San Francisco: NONMEM Project Group, Univ. Calif. (1989).
B. Efron, and R. Tibshirani. An Introduction to the Bootstrap, Chapman and Hall, London, UK (1993).
R. Ramakrishnan, W. K. Cheung F. Farrell, L. Joffee, and W. J. Jusko. Pharmacokinetic and pharmacodynamic modeling of recombinant human erythropoietin after intravenous and subcutaneous dose administration in cynomologous monkeys. J. Pharmacol. Exp. Ther. 306:1–8 (2003).
W. Krzyzanski, W. J. Jusko, M. C. Wacholtz, N. Minton, and W. K. Cheung. Pharmacokinetic and pharmacodynamic modeling of recombinant human erythropoietin after multiple subcutaneous doses in healthy subjects. Eur. J. Pharm. Sci. 26:295–306 (2005).
S. H. Chapel, P. Veng-Pedersen, R. L. Schmidt, and J. A. Widness. A pharmacodynamic analysis of erythropoietin-stimulated reticulocyte response in phlebotomized sheep. J. Pharmacol. Exp. Ther. 295:346–351 (2000).
P. Veng-Pedersen, S. Chapel, P. R. L. Schmidt, N. H. Al-Huniti, R. T. Cook, and J. A. Widness. An integrated pharmacodynamic analysis of erythropoietin, reticulocyte, and hemoglobin responses in acute anemia. Pharm. Res. 19:1630–1635 (2002).
N. I. Berlin and P. D. Berk. The biological life of the red cell. In: D. M. Surgenor (ed.), The Red Blood Cell. Vol. II. Academic Press, New York, 1975.
L. Rice, W. Ruiz, T. Driscoll, C. E. Whitley, R. Tapa, D. L. Hachey, G. F. Gonzales, and C. P. Alfrey. Neocytolysis on descent from altitude: a newly recognized mechanism for the control of red cell mass. Ann. Intern. Med. 134:652–656 (2001).
N. A. Noble, Q. P. Xu, L. L. Hoge. Reticulocytes II: reexamination of the in vivo survival of stress reticulocytes. Blood 75:1877–1882 (1990).
A. Ganzoni, R. S. Hillman, and C. A. Finch, Maturation of the macroreticulocyte. Br. J. Haematol. 16:119–135 (1969).
S. E. Come, S. B. Shohet, and S. H. Robinson. Surface remodeling vs. whole-cell hemolysis of reticulocytes produced with erythroid stimulation or iron deficiency anemia. Blood 44:817–830 (1974)
Acknowledgements
This study was supported by Johnson & Johnson Pharmaceutical Research & Development, A Division of Janssen Pharmaceutica, NV, Beerse, Belgium, and in part by the National Institute of General Medical Sciences, National Institutes of Health Grant GM 57980.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A
Derivation of the Precursor–successor Relationship Eq. 12
If the fixed lifespan T RET0 is assumed for all reticulocytes, then the RBC production is the delayed T RET0 reticulocyte production rate (11):
Thus, similarly to Eq. 11, one can write
Changing the variables in the above integral \( s = z - T_{{{\text{RET0}}}} \) leads to
The interval of integration in Eq. 27, \( t - T_{{{\text{RET0}}}} > s > t - T_{{{\text{RET0}}}} - T_{{{\text{RBC}}}} \), can be partitioned to the following subintervals: \( t - T_{{{\text{RET}}}} > s > t - 2T_{{{\text{RET}}}} ,\,t - 2T_{{{\text{RET}}}} > s > t - 3T_{{{\text{RET}}}} , \ldots ,t - NT_{{{\text{RET}}}} > s > t - {\left( {N + 1} \right)}T_{{{\text{RET0}}}} \), where N =INT(t/T RET0) is the integer describing how many times t is bigger than T RET0. One can now decompose the integral in Eq. 27 into a sum of integrals over these subintervals and a remainder:
Equation 11 implies that for any integer i
Since for t/T RET < N + 1, the upper limit in the reminder integral in Eq. 28 is less than 0, and for t ≤ T RBC the lower limit in this integral is less than 0 as well. We assume that prior to erythropoietin treatment the reticulocyte production was at the baseline level, k R(s) = k R0 for s < 0. Then the reminder integral in Eq. 28 is equal to
Taking into account the baseline Eqs. 4 and 5 yields
One can now combine Eqs. 28, 29, 30, 31, and obtain
Because RBC is the sum of MRBC and RET, then
and Eq. 12 follows.
APPENDIX B
Derivation of equation for RET(t)
Taking into account Eq. 16 for the reticulocyte conversion rate k out(t) one can integrate both sides of Eq. 1 from 0 to t and obtain
Changing the variables in the second integral \( s = z - T_{{{\text{RET0}}}} \) and in the third integral \( s = z - T_{{{\text{RET}}}} \) yields
Since for \( t < 0{\text{ }}k_{{\text{R}}} {\left( t \right)} = k_{{{\text{R0}}}} \) and χ(t) = 0, the integrals in Eq. 35 can be further simplified to
The reticulocytes at time t = 0 are at steady-state, therefore Eq. 4 implies that the first and second term in Eq. 36 cancel each other out. If the first integral is combined with the second, and the third with the fourth, then one obtains
which is the exact form of Eq. 17. Assuming the following relationships between the time parameters
they can be ordered as follows \( T_{{\text{0}}} < T_{{\text{0}}} + T_{{{\text{RET0}}}} < T_{{\text{0}}} + T_{{{\text{RET}}}} < T_{{\text{1}}} < T_{{\text{1}}} + T_{{{\text{RET0}}}} < T_{{\text{1}}} + T_{{{\text{RET}}}} < T_{{\text{2}}} < T_{{\text{2}}} + T_{{{\text{RET0}}}} \). Consequently, an arbitrary t value must fall into one of the following intervals:
t ≤ T0, then
T0 < t ≤ T0 + TRET0, then
T0 + TRET0 < t ≤ T0 + TRET, then
T0 + TRET < t ≤ T1, then
T1 < t ≤ T1 + TRET0, then
T1 + TRET0 < t ≤ T1 + TRET, then
T1 + TRET < t ≤ T2, then
T2 < t ≤ T2 + TRET0, then
T2 + TRET0 ≤ t, then
Arranging terms in Eqs. 39, 40, 41, 42, 43, 44, 45, 46, 47 yields Eq. 10. A similar derivation holds for Eq. 20, except that one needs to consider the cases determined by the intervals T 0 < T 1 < T 2 < T RBC and use Eq. 8.
APPENDIX C
NONMEM control stream and data file for estimation of T RET0 from Eq. 12
$PROB Estimation of TRET
$INPUT ID TIME CMT MDV DV RET RBC DOSE
$DATA C:\data_1041.csv IGNORE=#
$PRED
TRET = THETA(1)*EXP(ETA(1))
"OPEN(2,FILE=’C:\nmv\run\fdata’)
"REWIND 2
"DO WHILE (.NOT.EOF(2))
" READ(2,*) X1,X2,X3,X4,X5,X6,X7,X8
" IF(X1.EQ.ID.AND.CMT.EQ.1) THEN
" IF (X2.EQ.0) RT0=X6
" IF (X2.EQ.2) RT2=X6
" IF (X2.EQ.3) RT3=X6
" IF (X2.EQ.4) RT4=X6
" IF (X2.EQ.5) RT5=X6
" IF (X2.EQ.6) RT6=X6
" IF (X2.EQ.7) RT7=X6
" IF (X2.EQ.8) RT8=X6
" IF (X2.EQ.9) RT9=X6
" IF (X2.EQ.11) RT11=X6
" IF (X2.EQ.13) RT13=X6
" IF (X2.EQ.15) RT15=X6
" IF (X2.EQ.17) RT17=X6
" IF (X2.EQ.19) RT19=X6
" IF (X2.EQ.21) RT21=X6
" IF (X2.EQ.23) RT23=X6
" IF (X2.EQ.25) RT25=X6
" IF (X2.EQ.27) RT27=X6
" IF (X2.EQ.28) RT28=X6
" IF (X2.EQ.31) RT31=X6
" IF (X2.EQ.32) RT32=X6
" IF (X2.EQ.34) RT34=X6
" IF (X2.EQ.35) RT35=X6
" IF (X2.EQ.40) RT40=X6
" IF (X2.EQ.42) RT42=X6
" IF (X2.EQ.0) RBC0=X7
"ENDIF
"ENDDO
"CLOSE(2)
" RB=RBC0
" IF(DOSE.GT.0) THEN
" I=0
" DO WHILE (TIME−I*TRET.GE.0.0)
" X=TIME−I*TRET
" IF (0.LT.X.AND.X.LE.2) RB=RB+RT0+(RT2−RT0)/(2−0)*(X−0)−RT0
" IF (2.LT.X.AND.X.LE.3) RB=RB+RT2+(RT3−RT2)/(3−2)*(X−2)−RT0
" IF (3.LT.X.AND.X.LE.4) RB=RB+RT3+(RT4−RT3)/(4−3)*(X−3)−RT0
" IF (4.LT.X.AND.X.LE.5) RB=RB+RT4+(RT5−RT4)/(5−4)*(X−4)−RT0
" IF (5.LT.X.AND.X.LE.6) RB=RB+RT5+(RT6−RT5)/(6−5)*(X−5)−RT0
" IF (6.LT.X.AND.X.LE.7) RB=RB+RT6+(RT7−RT6)/(7−6)*(X−6)−RT0
" IF (7.LT.X.AND.X.LE.8) RB=RB+RT7+(RT8−RT7)/(8−7)*(X−7)−RT0
" IF (8.LT.X.AND.X.LE.9) RB=RB+RT8+(RT9−RT8)/(9−8)*(X−8)−RT0
" IF (9.LT.X.AND.X.LE.11) RB=RB+RT9+(RT11−RT9)/(11−9)*(X−9)−RT0
" IF (11.LT.X.AND.X.LE.13) RB=RB+RT11+(RT13−RT11)/(13−11)*(X−11)−RT0
" IF (13.LT.X.AND.X.LE.15) RB=RB+RT13+(RT15−RT13)/(15−13)*(X−13)−RT0
" IF (15.LT.X.AND.X.LE.17) RB=RB+RT15+(RT17−RT15)/(17−15)*(X−15)−RT0
" IF (17.LT.X.AND.X.LE.19) RB=RB+RT17+(RT19−RT17)/(19−17)*(X−17)−RT0
" IF (19.LT.X.AND.X.LE.21) RB=RB+RT19+(RT21−RT19)/(21−19)*(X−19)−RT0
" IF (21.LT.X.AND.X.LE.23) RB=RB+RT21+(RT23−RT21)/(23−21)*(X−21)−RT0
" IF (23.LT.X.AND.X.LE.25) RB=RB+RT23+(RT25−RT23)/(25−23)*(X−23)−RT0
" IF (25.LT.X.AND.X.LE.27) RB=RB+RT25+(RT27−RT25)/(27−25)*(X−25)−RT0
" IF (27.LT.X.AND.X.LE.28) RB=RB+RT27+(RT28−RT27)/(28−27)*(X−27)−RT0
" IF (28.LT.X.AND.X.LE.31) RB=RB+RT28+(RT31−RT28)/(31−28)*(X−28)−RT0
" IF (31.LT.X.AND.X.LE.32) RB=RB+RT31+(RT32−RT31)/(32−31)*(X−31)−RT0
" IF (32.LT.X.AND.X.LE.34) RB=RB+RT32+(RT34−RT32)/(34−32)*(X−32)−RT0
" IF (34.LT.X.AND.X.LE.35) RB=RB+RT34+(RT35−RT34)/(35−34)*(X−34)−RT0
" IF (35.LT.X.AND.X.LE.40) RB=RB+RT35+(RT40−RT35)/(40−35)*(X−35)−RT0
" IF (40.LT.X.AND.X.LE.42) RB=RB+RT40+(RT42−RT40)/(42−40)*(X−40)−RT0
" I=I+1
" ENDDO
" ENDIF
IPRED=RB+ERR(1)
Y = IPRED
IRES = DV − IPRED
$THETA (0,,15) ;TRET
$OMEGA 0.00 FIX
$SIGMA 0.02
$EST NSIGDIG=3 MAX=9999 PRINT=1 NOABORT POSTHOC METHOD=1
$COV PRINT=E
$TABLE ID TIME CMT IPRED IRES DOSE RET
NOPRINT FILE=C:\1041.txt
The following file data_1041.txt contains records for Subject 1041. The missing values for RET were calculated by the linear interpolation of the neighboring reticulocyte measurements
#ID | TIME | CMT | MDV | DV | RET | RBC | DOSE |
|---|---|---|---|---|---|---|---|
1041 | 0 | 1 | 0 | 5.145 | 0.054 | 5.145 | 40 |
1041 | 2 | 1 | 0 | 5.55 | 0.072 | 5.55 | 40 |
1041 | 3 | 1 | 0 | 5.49 | 0.135 | 5.49 | 40 |
1041 | 4 | 1 | 0 | 5.36 | 0.192 | 5.36 | 40 |
1041 | 5 | 1 | 0 | 5.2 | 0.253 | 5.2 | 40 |
1041 | 6 | 1 | 0 | 5.26 | 0.245 | 5.26 | 40 |
1041 | 7 | 1 | 0 | 5.5 | 0.291 | 5.5 | 40 |
1041 | 8 | 1 | 0 | 5.4 | 0.349 | 5.4 | 40 |
1041 | 9 | 1 | 0 | 5.69 | 0.218 | 5.69 | 40 |
1041 | 11 | 1 | 0 | 5.44 | 0.132 | 5.44 | 40 |
1041 | 13 | 1 | 0 | 5.37 | 0.103 | 5.37 | 40 |
1041 | 15 | 1 | 0 | 5.27 | 0.092 | 5.27 | 40 |
1041 | 17 | 1 | 0 | 5.45 | 0.070 | 5.45 | 40 |
1041 | 19 | 1 | 0 | 5.55 | 0.093 | 5.55 | 40 |
1041 | 21 | 1 | 0 | 5.39 | 0.072 | 5.39 | 40 |
1041 | 23 | 1 | 0 | 5.49 | 0.083 | 5.49 | 40 |
1041 | 25 | 1 | 0 | 5.55 | 0.084 | 5.55 | 40 |
1041 | 27 | 1 | 1 | 0 | 0.108 | 0 | 40 |
1041 | 28 | 1 | 0 | 5.49 | 0.120 | 5.49 | 40 |
1041 | 31 | 1 | 1 | 0 | 0.111 | 0 | 40 |
1041 | 32 | 1 | 1 | 0 | 0.108 | 0 | 40 |
1041 | 34 | 1 | 1 | 0 | 0.101 | 0 | 40 |
1041 | 35 | 1 | 1 | 0 | 0.098 | 0 | 40 |
1041 | 40 | 1 | 1 | 0 | 0.082 | 0 | 40 |
1041 | 42 | 1 | 0 | 5.22 | 0.076 | 5.22 | 40 |
APPENDIX D
NONMEM control stream for estimation of T 0, ΔT, ΔT 1, Δk R1, and Δk R2
$PROB RHUEPO EFFECT ON RET AND RBC
$INPUT ID TIME CMT MDV DV RET RBC DOSE
$DATA C:\data_1034.csv IGNORE = #
$PRED
T0 = THETA(1)*EXP(ETA(1))
DT1 = THETA(2)
DT = THETA(3)
DT2 = THETA(4)
DKR1 = THETA(5)
DKR2 = THETA(6)
"OPEN(2,FILE=’C:\nmv\run\fdata’)
"REWIND 2
"DO WHILE (.NOT.EOF(2))
" READ(2,*) X1,X2,X3,X4,X5,X6,X7,X8
" IF(X1.EQ.ID.AND.CMT.EQ.1) THEN
" IF (X2.EQ.0) THEN
" RT0=X6
" RBC0=X7
" TT=RT0/(RBC0−RT0)*120.0
" ENDIF
" ENDIF
"ENDDO
" CLOSE(2)
KR0=RT0/TT
KR1=KR0+DKR1
KR2=KR0−DKR2
T1=T0+DT1
T2=T1+DT2
TR=TT+DT
CH0=0
CH1=1
CH2=0
X=TIME
IT1=0
IT2=0
IT3=0
IT4=0
IF (X.LE.T0) IT1=KR0*X
IF (X.GT.T0.AND.X.LE.T1) IT1=KR1*(X−T0)+KR0*T0
IF (X.GT.T1.AND.X.LE.T2) IT1=KR2*(X−T1)+KR1*(T1−T0)+KR0*T0
IF (X.GT.T2) IT1=KR0*(X−T2)+KR2*(T2−T1)+KR1*(T1−T0)+KR0*T0
X=TIME−TT
IF (X.LE.T0) IT2=KR0*X
IF (X.GT.T0.AND.X.LE.T1) IT2=KR1*(X−T0)+KR0*T0
IF (X.GT.T1.AND.X.LE.T2) IT2=KR2*(X−T1)+KR1*(T1−T0)+KR0*T0
IF (X.GT.T2) IT2=KR0*(X−T2)+KR2*(T2−T1)+KR1*(T1−T0)+KR0*T0
X=TIME−TT
IF (X.LE.T0) IT3=KR0*CH0*X
IF (X.GT.T0.AND.X.LE.T1) IT3=KR1*CH1*(X−T0)+KR0*CH0*T0
IF (X.GT.T1.AND.X.LE.T2) THEN
IT3=KR2*CH2*(X−T1)+KR1*CH1*(T1−T0)+KR0*CH0*T0
ENDIF
IF (X.GT.T2) THEN
IT3=KR0*CH0*(X−T2)+KR2*CH2*(T2−T1)+KR1*CH1*(T1−T0)+KR0*CH0*T0
ENDIF
X=TIME−TR
IF (X.LE.T0) IT4=KR0*CH0*X
IF (X.GT.T0.AND.X.LE.T1) IT4=KR1*CH1*(X−T0)+KR0*CH0*T0
IF (X.GT.T1.AND.X.LE.T2) THEN
IT4=KR2*CH2*(X−T1)+KR1*CH1*(T1−T0)+KR0*CH0*T0
ENDIF
IF (X.GT.T2) THEN
IT4=KR0*CH0*(X−T2)+KR2*CH2*(T2−T1)+KR1*CH1*(T1−T0)+KR0*CH0*T0
ENDIF
R=IT1−IT2+IT3−IT4
RB=IT1−KR0*TIME+RBC0
IPRED=0
IF(CMT.EQ.1) IPRED=RB+ERR(1)
IF(CMT.EQ.2) IPRED=R+ERR(2)
Y = IPRED
IRES = DV − IPRED
$THETA
(0,1.5,5) ;T0
(0,7.0 ,20) ;DT1
(0,2,20) ;DT
42 FIX ;DT2
(0,0.04, 0.1) ;DKR1
(0,0.01,0.05) ;DKR2
$OMEGA 0.0 FIX
$SIGMA
0.01 ; RBC
0.01 ; RET
$ESTIMATION NSIGDIG=3 MAX=999 PRINT=1 NOABORT POSTHOC METHOD=1
$COV PRINT=E
$TABLE ID TIME CMT IPRED DOSE RET RBC
NOPRINT
FILE=C:\1034.txt
#ID | TIME | CMT | MDV | DV | RET | RBC | DOSE |
|---|---|---|---|---|---|---|---|
1034 | 0 | 1 | 0 | 4.925 | 0.079 | 4.925 | 160 |
1034 | 0 | 2 | 0 | 0.079 | 0.079 | 4.925 | 160 |
1034 | 2 | 1 | 0 | 5.38 | 0.126 | 5.38 | 160 |
1034 | 2 | 2 | 0 | 0.126 | 0.126 | 5.38 | 160 |
1034 | 3 | 1 | 0 | 5.59 | 0.149 | 5.59 | 160 |
1034 | 3 | 2 | 0 | 0.149 | 0.149 | 5.59 | 160 |
1034 | 4 | 1 | 0 | 5.49 | 0.189 | 5.49 | 160 |
1034 | 4 | 2 | 0 | 0.189 | 0.189 | 5.49 | 160 |
Rights and permissions
About this article
Cite this article
Krzyzanski, W., Perez-Ruixo, J.J. An Assessment of Recombinant Human Erythropoietin Effect on Reticulocyte Production Rate and Lifespan Distribution in Healthy Subjects. Pharm Res 24, 758–772 (2007). https://doi.org/10.1007/s11095-006-9195-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-006-9195-y