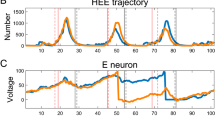
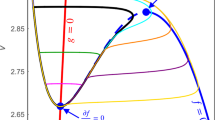
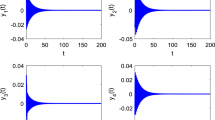
Abstract
This article communicates results on regular depolarization cascades in periodically kicked feedforward chains of excitable two-dimensional FitzHugh–Nagumo systems driven by sufficiently strong excitatory forcing at the front node. The study documents a parameter exploration by way of changes to the forcing period, upon which the dynamics undergoes a transition from simple depolarization to more complex behavior, including the emergence of mixed-mode oscillations. Both rigorous studies and careful numerical observations are presented. In particular, we provide rigorous proofs for existence and stability of periodic traveling waves of depolarization, as well as the existence and propagation of a simple mixed-mode oscillation that features depolarization and refraction in alternating fashion. Detailed numerical investigation reveals a mechanism for the emergence of complex mixed-mode oscillations featuring a potentially high number of large amplitude voltage spikes interspersed by an occasional small amplitude reset that fails to cross threshold. Further careful numerical investigation provides insights into the propagation of this complex phenomenology in the downstream, where we see an effective filtration property of the network; the latter amounts to a successive reduction in the complexity of mixed-mode oscillations down the chain.
























Similar content being viewed by others
Availability of data and materials
Not Applicable.
Notes
Of interest for the feedforward chain network studied here, how do MMOs propagate downstream (do they regularize or become more irregular?), and are there regimes in which wave propagation/signal transmission downstream is altogether blocked?
See our discussion of the state of affairs for \(\alpha \) only slightly smaller than \(\alpha _2\) in Remark 11.
References
Hodgkin, A.L., Huxley, A.F.: A quantitative description of membrane current and its application to conduction and excitation in nerve. J. Physiol. 117(4), 500–544 (1952). https://doi.org/10.1113/jphysiol.1952.sp004764
FitzHugh, R.: Impulses and physiological states in theoretical models of nerve membrane. Biophys. J . 1(6), 445–466 (1961). https://doi.org/10.1016/s0006-3495(61)86902-6
Nagumo, J., Arimoto, S., Yoshizawa, S.: An active pulse transmission line simulating nerve axon. Proc. IRE 50(10), 2061–2070 (1962). https://doi.org/10.1109/jrproc.1962.288235
Fenton, F., Cherry, E.: Models of cardiac cell. Scholarpedia 3(8), 1868 (2008). https://doi.org/10.4249/scholarpedia.1868
Hastings, S.P.: Some mathematical problems from neurobiology. Am. Math. Mon. 82(9), 881–895 (1975). https://doi.org/10.1080/00029890.1975.11993972
Hastings, S.P.: On the existence of of homoclinic and periodic orbits for the Fitzhugh–Nagumo equations. Q. J. Math. 27(1), 123–134 (1976). https://doi.org/10.1093/qmath/27.1.123
McKean, H.P.: Nagumo’s equation. Adv. Math. 4(3), 209–223 (1970). https://doi.org/10.1016/0001-8708(70)90023-x
Jones, C.K.R.T.: Stability of the travelling wave solution of the FitzHugh–Nagumo system. Trans. Am. Math. Soc. 286(2), 431–431 (1984). https://doi.org/10.1090/s0002-9947-1984-0760971-6
Jones, C., Kopell, N., Langer, R.: Construction of the Fitzhugh–Nagumo pulse using differential forms. In: Aris, R., Aronson, D.G., Swinney, H.L. (eds.) Patterns and Dynamics in Reactive Media, pp. 101–115. Springer, New York (1991)
Rauch, J., Smoller, J.: Qualitative theory of the FitzHugh–Nagumo equations. Adv. Math. 27(1), 12–44 (1978). https://doi.org/10.1016/0001-8708(78)90075-0
Rinzel, J., Keaner, J.P.: Hopf bifurcation to repetitive activity in nerve. SIAM J. Appl. Math. 43(4), 907–922 (1983). https://doi.org/10.1137/0143058
Ambrosio, B.: Hopf bifurcation in an oscillatory-excitable reaction–diffusion model with spatial heterogeneity. Int. J. Bifurc. Chaos 27(05), 1750065 (2017). https://doi.org/10.1142/s0218127417500651
Ambrosio, B., Francoise, J.-P.: Propagation of bursting oscillations. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences 367(1908), 4863–4875 (2009). https://doi.org/10.1098/rsta.2009.0143
Maia, P.D., Kutz, J.N.: Identifying critical regions for spike propagation in axon segments. J. Comput. Neurosci. 36(2), 141–155 (2013). https://doi.org/10.1007/s10827-013-0459-3
Jean-René, C., Fernandez, B.: Dynamics of Coupled Map Lattices and of Related Spatially Extended Systems. Springer, Berlin (2005). https://doi.org/10.1007/b103930
Afraimovich, V.: Some Topological Properties of Lattice Dynamical Systems, pp. 153–179. Springer, Berlin, Heidelberg (2005). https://doi.org/10.1007/11360810_7
Baesens, C.: Spatially extended systems with monotone dynamics: continuous time. In: Lecture Notes in Physics, pp. 241–263. Springer, Berlin, Heidelberg (2005)
Floría, L.M., Baesens, C., Gómez-Gardeñes, J.: The Frenkel–Kontorova model. In: Lecture Notes in Physics, pp. 209–240. Springer, Berlin, Heidelberg (2005). https://doi.org/10.1007/11360810_9
Ermentrout, G.B., Terman, D.H.: Mathematical Foundations of Neuroscience. Springer, New York, London (2010). https://doi.org/10.1007/978-0-387-87708-2
Izhikevich, E.M.: Dynamical Systems in Neuroscience. Computational neuroscience Dynamical systems in neuroscience. MIT Press, Cambridge (2007). https://books.google.com/books?id=kVjM6DFk-twC
Chariker, L., Shapley, R., Young, L.-S.: Orientation selectivity from very sparse LGN inputs in a comprehensive model of macaque v1 cortex. J. Neurosci. 36(49), 12368–12384 (2016). https://doi.org/10.1523/jneurosci.2603-16.2016
Chariker, L., Young, L.-S.: Emergent spike patterns in neuronal populations. J. Comput. Neurosci. 38(1), 203–220 (2014). https://doi.org/10.1007/s10827-014-0534-4
Gerstner, W., Kistler, W.M.: Spiking Neuron Models: Single Neurons, Populations, Plasticity. Cambridge University Press, Cambridge, New York (2002). https://books.google.com/books?id=Rs4oc7HfxIUC
Keener, J., Sneyd, J.: Mathematical Physiology. Springer, New York (2009). https://doi.org/10.1007/978-0-387-75847-3
Fenichel, N.: Geometric singular perturbation theory for ordinary differential equations. J. Differ. Equ. 31(1), 53–98 (1979). https://doi.org/10.1016/0022-0396(79)90152-9
Jones, C.K.R.T.: Geometric singular perturbation theory. In: Dynamical Systems, pp. 44–118. Springer, Berlin, Heidelberg (1995). https://doi.org/10.1007/bfb0095239
Krupa, M., Szmolyan, P.: Extending geometric singular perturbation theory to nonhyperbolic points–fold and canard points in two dimensions. SIAM J. Math. Anal. 33(2), 286–314 (2001). https://doi.org/10.1137/s0036141099360919
Broens, M., Bar-Eli, K.: Canard explosion and excitation in a model of the Belousov–Zhabotinskii reaction. J. Phys. Chem. 95(22), 8706–8713 (1991). https://doi.org/10.1021/j100175a053
Rotstein, H.G., Kopell, N., Zhabotinsky, A.M., Epstein, I.R.: Canard phenomenon and localization of oscillations in the Belousov–Zhabotinsky reaction with global feedback. J. Chem. Phys. 119(17), 8824–8832 (2003). https://doi.org/10.1063/1.1614752
Rotstein, H.G., Wechselberger, M., Kopell, N.: Canard induced mixed-mode oscillations in a medial entorhinal cortex layer II stellate cell model. SIAM J. Appl. Dyn. Syst. 7(4), 1582–1611 (2008). https://doi.org/10.1137/070699093
V-Ghaffari, B., Kouhnavard, M., Elbasiouny, S.M.: Mixed-mode oscillations in pyramidal neurons under antiepileptic drug conditions. PLOS ONE 12(6), 0178244 (2017). https://doi.org/10.1371/journal.pone.0178244
Desroches, M., Guckenheimer, J., Krauskopf, B., Kuehn, C., Osinga, H.M., Wechselberger, M.: Mixed-mode oscillations with multiple time scales. SIAM Rev. 54(2), 211–288 (2012). https://doi.org/10.1137/100791233
Wechselberger, M.: Canards. Scholarpedia 2(4), 1356 (2007). https://doi.org/10.4249/scholarpedia.1356
Benoît, E., Callot, J.F., Diener, F., Diener, M.: Chasse au canard. Collect. Math. 32, 37–119 (1981)
Ambrosio, B., Aziz-Alaoui, M.A., Yafia, R.: Canard phenomenon in a slow-fast modified Leslie–Gower model. Math. Biosci. 295, 48–54 (2018). https://doi.org/10.1016/j.mbs.2017.11.003
Dumortier, F., Roussarie, R.: Canard cycles and center manifolds. Mem. Am. Math. Soc. 121, 577 (1996). https://doi.org/10.1090/memo/0577
Eckhaus, W.: Relaxation oscillations including a standard chase on french ducks. In: Lecture Notes in Mathematics, pp. 449–497. Springer, Berlin, Heidelberg (1983). https://doi.org/10.1007/bfb0062381
Brøns, M., Krupa, M., Wechselberger, M.: Mixed mode oscillations due to the generalized Canard phenomenon. Am. Math. Soc. (2006). https://doi.org/10.1371/journal.pone.0178244
Krupa, M., Ambrosio, B., Aziz-Alaoui, M.A.: Weakly coupled two-slow-two-fast systems, folded singularities and mixed mode oscillations. Nonlinearity 27(7), 1555–1574 (2014). https://doi.org/10.1088/0951-7715/27/7/1555
Fernandez, B., Mintchev, S.M.: Wave generation in unidirectional chains of idealized neural oscillators. J. Math. Neurosci. (2016). https://doi.org/10.1186/s13408-016-0037-x
Lanford, O.E., Mintchev, S.M.: Stability of a family of travelling wave solutions in a feedforward chain of phase oscillators. Nonlinearity 28(1), 237–261 (2014). https://doi.org/10.1088/0951-7715/28/1/237
Perko, L.: Differential Equations and Dynamical Systems. Springer, New York (2001). https://doi.org/10.1007/978-1-4613-0003-8
Andronov, A.A.: Theory of Bifurcations of Dynamic Systems on a Plane. Wiley, New York (1973)
Krupa, M., Popović, N., Kopell, N.: Mixed-mode oscillations in three time-scale systems: A prototypical example 7(2), 361–420 (2008). https://doi.org/10.1137/070688912
Krupa, M., Popović, N., Kopell, N., Rotstein, H.G.: Mixed-mode oscillations in a three time-scale model for the dopaminergic neuron 18(1), 015106 (2008). https://doi.org/10.1063/1.2779859
Maesschalck, P.D., Kutafina, E., Popović, N.: Three time-scales in an extended bonhoeffer-van der pol oscillator 26(4), 955–987 (2014). https://doi.org/10.1007/s10884-014-9356-3
Ambrosio, B., Aziz-Alaoui, M.A., Mondal, A., Mondal, A., Sanjeev, Sharma, K., Upadhyay, R.K.: Non trivial dynamics dynamics in the fizhugh-rinzel model and in non-homogeneous oscillatory-excitable reaction-diffusion systems: a few highlights. https://doi.org/10.48550/arXiv.2205.04311
Acknowledgements
The research of Benjamin Ambrosio (BA) on Neuroscience inspired modeling is partially funded by CNRS (IEA00134). More generally BA wants to thank Le Havre Normandie University, LMAH, Région Normandie, ISCN, the Courant Institute of Mathematical Sciences and New York University, the Hudson School of Mathematics, New York, USA, for material and/or financial support for research in Dynamical Systems, Complex Systems and its applications.
Funding
The research of BA has been partially funded by CNRS (IEA00134), LMAH, Région Normandie and the Hudson School of Mathematics.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no competing interests
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ambrosio, B., Mintchev, S.M. Periodically kicked feedforward chains of simple excitable FitzHugh–Nagumo neurons. Nonlinear Dyn 110, 2805–2829 (2022). https://doi.org/10.1007/s11071-022-07757-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11071-022-07757-0