Abstract
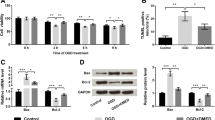
Ischemic stroke has been confirmed to cause neuronal injury due to its insufficient supply of glucose and oxygen to brain tissue. Previous research has shown that oxidative stress, a result of excessive accumulation of reactive oxygen species (ROS), relates to pathophysiology of ischemic stroke, and causes oxidative damage to biomolecules, eventually leading to programmed cell death. Meanwhile, apigenin has been shown to exhibit antioxidant, anti-inflammatory, anti-cancer properties and neuroprotective action. Hence, this study was to investigate the potential mechanisms underlying the neural protection of apigenin on oxygen and glucose deprivation/reperfusion (OGD/R) induced neuronal injury in differentiated PC12 cells. Cells were pretreated with apigenin for 6 h, and then subjected to OGD for 12 h followed by reperfusion for 24 h. The results showed that OGD/R significantly decreased cell viability, mitochondrial membrane potential, mRNA levels of antioxidant and detoxifying enzymes and Nrf2 protein expression, while elevated the release of LDH, cell apoptosis, intracellular ROS level, P53 protein expression and upregulated its downstream genes in PC12 cells. However, apigenin effectively inhibited these undesirable changes induced by OGD/R. Our findings demonstrate that this compound attenuates OGD/R induced neuronal injury mainly by virtue of its anti-apoptosis and antioxidative properties via affecting the expression of Nrf2 and P53, and their downstream target gene transcription.







Similar content being viewed by others
References
Clemens JA (2000) Cerebral ischemia: gene activation, neuronal injury, and the protective role of antioxidants. Free Radic Biol Med 28:1526–1531
Janardhan V, Qureshi AI (2004) Mechanisms of ischemic brain injury. Curr Cardiol Rep 6:117–123
Rothwell PM, Coull AJ, Giles MF, Howard SC, Silver LE, Bull LM, Gutnikoy SA, Edwards P, Mant D, Sackley CM, Farmer A, Sandercock PA, Dennis MS, Warlow CP, Bamford JM, Anslow P, Oxford Vascular Study (2004) Change in stroke incidence, mortality, case-fatality, severity, and risk factors in Oxfordshire, UK from 1981 to 2004 (Oxford Vascular Study). Lancet 363:1925–1933
Dirnagl U, Iadecola C, Moskowitz MA (1999) Pathobiology of ischaemic stroke: an integrated view. Trends Neurosci 22:391–397
Lipton P (1999) Ischemic cell death in brain neurons. Physiol Rev 79:1431–1568
Lo EH, Dalkara T, Moskowitz MA (2003) Mechanisms, challenges and opportunities in stroke. Nat Rev Neurosci 4:399–415
Almeida A, Delgado-Esteban M, Bolanos JP, Medina JM (2002) Oxygen and glucose deprivation induces mitochondrial dysfunction and oxidative stress in neurones but not in astrocytes in primary culture. J Neurochem 81:207–217
Turrens JF (2003) Mitochondrial formation of reactive oxygen species. J Physiol 552:335–344
Shen CC, Huang HM, Ou HC, Chen HL, Chen WC, Jeng KC (2004) Protective effect of nicotinamide on neuronal cells under oxygen and glucose deprivation and hypoxia/reoxygenation. J Biomed Sci 11:472–481
Kontos HA (2001) Oxygen radicals in cerebral ischemia: the 2001 Willis lecture. Stroke 32:2712–2716
Li C, Jackson RM (2002) Reactive species mechanisms of cellular hypoxia-reoxygenation injury. Am J Physiol Cell Physiol 282:C227–C241
Sugawara T, Chan PH (2003) Reactive oxygen radicals and pathogenesis of neuronal death after cerebral ischemia. Antioxid Redox Signal 5:597–607
Gilgun-Sherki Y, Rosenbaum Z, Melamed E, Offen D (2002) Antioxidant therapy in acute central nervous system injury: current state. Pharmacol Rev 54:271–284
Mehta SL, Manhas N, Raghubir R (2007) Molecular targets in cerebral ischemia for developing novel therapeutics. Brain Res Rev 54:34–66
Dichter MA, Tischler AS, Greene LA (1977) Nerve growth factor-induced increase in electrical excitability and acetylcholine sensitivity of a rat pheochromocytoma cell line. Nature 268:501–504
Buxser S, Decker D, Ruppel P (1990) Relationship among types of nerve growth factor receptors on PC12 cells. J Biol Chem 265:12701–12710
Reimann-Philipp U, Ovase R, Weigel PH, Grammas P (2001) Mechanisms of cell death in primary cortical neurons and PC12 cells. J Neurosci Res 64:654–660
Tabakman R, Jiang H, Levine RA, Kohen R, Lazarovici P (2004) Apoptotic characteristics of cell death and the neuroprotective effect of homocarnosine on pheochromocytoma PC12 cells exposed to ischemia. J Neurosci Res 75:499–507
Singh G, Siddiqui MA, Khanna VK, Kashyap MP, Yadav S, Gupta YK, Pant KK, Pant AB (2009) Oxygen glucose deprivation model of cerebral stroke in PC-12 cells: glucose as a limiting factor. Toxicol Mech Methods 19:154–160
Jiang H, Koubi D, Zhang L, Kuo J, Rodriguez AI, Hunter TJ, Gautam SC, Levine RA (2005) Inhibitors of iNOS protects PC12 cells against the apoptosis induced by oxygen and glucose deprivation. Neurosci Lett 375:59–63
Zhou XQ, Zeng XN, Kong H, Sun XL (2008) Neuroprotective effects of berberine on stroke models in vitro and in vivo. Neurosci Lett 447:31–36
Manach C, Scalbert A, Morand C, Remesy C, Jimenez L (2004) Polyphenols: food sources and bioavailability. Am J Clin Nutr 79:727–747
Choi AY, Choi JH, Lee JY, Yoon KS, Choe W, Ha J, Yeo EJ, Kang I (2010) Apigenin protects HT22 murine hippocampal neuronal cells against endoplasmic reticulum stress-induced apoptosis. Neurochem Int 57:143–152
Lin M, Lu SS, Wang AX, Qi XY, Zhao D, Wang ZH, Man MQ, Tu CX (2011) Apigenin attenuates dopamine-induced apoptosis in melanocytes via oxidative stress-related p38, c-Jun NH2-terminal kinase and Akt signaling. J Dermatol Sci 63:10–16
Yano S, Umeda D, Yamashita S, Yamada K, Tachibana H (2009) Dietary apigenin attenuates the development of atopic dermatitis-like skin lesions in NC/Nga mice. J Nutr Biochem 20:876–881
Shukla S, Gupta S (2010) Apigenin: a promising molecule for cancer prevention. Pharm Res 27:962–978
Liu R, Zhang T, Yang H, Lan X, Ying J, Du G (2011) The flavonoid apigenin protects brain neurovascular coupling against amyloid-beta (2)(5)(-)(3)(5)-induced toxicity in mice. J Alzheimers Dis 24:85–100
Zhao L, Wang JL, Wang YR, Fa XZ (2013) Apigenin attenuates copper-mediated beta-amyloid neurotoxicity through antioxidation, mitochondrion protection and MAPK signal inactivation in an AD cell model. Brain Res 1492:33–45
Ha SK, Lee P, Park JA, Oh HR, Lee SY, Park JH, Lee EH, Ryu JH, Lee KR, Kim SY (2008) Apigenin inhibits the production of NO and PGE2 in microglia and inhibits neuronal cell death in a middle cerebral artery occlusion-induced focal ischemia mice model. Neurochem Int 52:878–886
Fan L, Dang X, Shi Z, Zhang C, Wang K (2011) Hydroxysafflor yellow A protects PC12 cells against the apoptosis induced by oxygen and glucose deprivation. Cell Mol Neurobiol 31:1187–1194
Kong SZ, Xian YF, Ip SP, Lai XP, Shi XG, Lin ZX, Su ZR (2013) Protective effects of hydroxysafflor yellow A on beta-amyloid-induced neurotoxicity in PC12 cells. Neurochem Res 38:951–960
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C (T)) Method. Methods 25:402–408
Xing G, Dong M, Li X, Zou Y, Fan L, Wang X, Cai D, Li C, Zhou L, Liu J, Niu Y (2011) Neuroprotective effects of puerarin against beta-amyloid-induced neurotoxicity in PC12 cells via a PI3K-dependent signaling pathway. Brain Res Bull 85:212–218
Chan PH (2001) Reactive oxygen radicals in signaling and damage in the ischemic brain. J Cereb Blood Flow Metab 21:2–14
Mitsios N, Gaffney J, Kumar P, Krupinski J, Kumar S, Slevin M (2006) Pathophysiology of acute ischaemic stroke: an analysis of common signalling mechanisms and identification of new molecular targets. Pathobiology 73:159–175
Hougee S, Sanders A, Faber J, Graus YM, van den Berg WB, Garssen J, Smit HF, Hoijer MA (2005) Decreased pro-inflammatory cytokine production by LPS-stimulated PBMC upon in vitro incubation with the flavonoids apigenin, luteolin or chrysin, due to selective elimination of monocytes/macrophages. Biochem Pharmacol 69:241–248
Fiskum G, Rosenthal RE, Vereczki V, Martin E, Hoffman GE, Chinopoulos C, Kowaltowski A (2004) Protection against ischemic brain injury by inhibition of mitochondrial oxidative stress. J Bioenerg Biomembr 36:347–352
Liu Y, Song XD, Liu W, Zhang TY, Zuo J (2003) Glucose deprivation induces mitochondrial dysfunction and oxidative stress in PC12 cell line. J Cell Mol Med 7:49–56
Niizuma K, Endo H, Chan PH (2009) Oxidative stress and mitochondrial dysfunction as determinants of ischemic neuronal death and survival. J Neurochem 109(Suppl 1):133–138
Dawson VL, Dawson TM, London ED, Bredt DS, Snyder SH (1991) Nitric oxide mediates glutamate neurotoxicity in primary cortical cultures. Proc Natl Acad Sci USA 8:6368–6371
Afsharzadeh M, Tayarani-Najaran Z, Zare A, Mousavi SH (2012) Protective effect of scutellaria litwinowii extract on serum/glucose-deprived cultured PC12 Cells and determining the role of reactive oxygen species. J Toxicol 2012:413279
Taguchi K, Motohashi H, Yamamoto M (2011) Molecular mechanisms of the Keap1-Nrf2 pathway in stress response and cancer evolution. Genes Cells 16:123–140
Itoh K, Chiba T, Takahashi S, Ishii T, Igarashi K, Katoh Y, Oyake T, Hayashi N, Satoh K, Hatayama I, Yamamoto M, Nabeshima Y (1997) An Nrf2/small Maf heterodimer mediates the induction of phase II detoxifying enzyme genes through antioxidant response elements. Biochem Biophys Res Commun 236:313–322
Wu KC, Cui JY, Klaassen CD (2011) Beneficial role of Nrf2 in regulating NADPH generation and consumption. Toxicol Sci 123:590–600
Zhang M, An C, Gao Y, Leak RK, Chen J, Zhang F (2013) Emerging roles of Nrf2 and phase II antioxidant enzymes in neuroprotection. Prog Neurobiol 100:30–47
Chai YS, Hu J, Lei F, Wang YG, Yuan ZY, Lu X, Wang XP, Du F, Zhang D, Xing DM, Du LJ (2013) Effect of berberine on cell cycle arrest and cell survival during cerebral ischemia and reperfusion and correlations with p53/cyclin D1 and PI3K/Akt. Eur J Pharmacol 708:44–55
Crompton M (2000) Mitochondrial intermembrane junctional complexes and their role in cell death. J Physiol 529(Pt 1):11–21
Christophe M, Nicolas S (2006) Mitochondria: a target for neuroprotective interventions in cerebral ischemia-reperfusion. Curr Pharm Des 12:739–757
Li J, Li B, Zhou WJ, Zhao FG, Li DJ, Wang WJ (2010) Medicated rat serum containing Gengnianchun decoction reduces apoptosis of pheochromocytoma cells insulted by amyloid beta protein. Zhong Xi Yi Jie He Xue Bao 8:472–479
Sablina AA, Budanov AV, Ilyinskaya GV, Agapova LS, Kravchenko JE, Chumakov PM (2005) The antioxidant function of the p53 tumor suppressor. Nat Med 11:1306–1313
Liu D, Xu Y (2011) p53, oxidative stress, and aging. Antioxid Redox Signal 15:1669–1678
el-Deiry WS, Harper JW, O’Connor PM, Velculescu VE, Canman CE, Jackman J, Pietenpol JA, Burrell M, Hill DE, Wang Y (1994) WAF1/CIP1 is induced in p53-mediated G1 arrest and apoptosis. Cancer Res 54:1169–1174
Nakano K, Vousden KH (2001) PUMA, a novel proapoptotic gene, is induced by p53. Mol Cell 7:683–694
Liu Z, Lu H, Shi H, Du Y, Yu J, Gu S, Chen X, Liu KJ, Hu CA (2005) PUMA overexpression induces reactive oxygen species generation and proteasome-mediated stathmin degradation in colorectal cancer cells. Cancer Res 65:1647–1654
Terao J (2009) Dietary flavonoids as antioxidants. Forum Nutr 61:87–94
Han RM, Tian YX, Liu Y, Chen CH, Ai XC, Zhang JP, Skibsted LH (2009) Comparison of flavonoids and isoflavonoids as antioxidants. J Agric Food Chem 57:3780–3785
Zhang D, Liu Y, Chu L, Wei Y, Wang D, Cai S, Zhou F, Ji B (2013) Relationship between the structures of flavonoids and oxygen radical absorbance capacity values: a quantum chemical analysis. J Phys Chem A 117:1784–1794
Faraonio R, Vergara P, Di Marzo D, Pierantoni MG, Napolitano M, Russo T, Cimino F (2006) p53 suppresses the Nrf2-dependent transcription of antioxidant response genes. J Biol Chem 281:39776–39784
Pani G, Bedogni B, Anzevino R, Colavitti R, Palazzotti B, Borrello S, Galeotti T (2000) Deregulated manganese superoxide dismutase expression and resistance to oxidative injury in p53-deficient cells. Cancer Res 60:4654–4660
Afrazi S, Esmaeili-Mahani S, Sheibani V, Abbasnejad M (2014) Neurosteroid allopregnanolone attenuates high glucose-induced apoptosis and prevents experimental diabetic neuropathic pain: in vitro and in vivo studies. J Steroid Biochem Mol Biol 139:98–103
Acknowledgments
The work was supported by Guangdong Provincial International Cooperation Project Funding of People’s Republic of China (No. 2011B050300021).
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Huizhen Guo and Songzhi Kong have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Guo, H., Kong, S., Chen, W. et al. Apigenin Mediated Protection of OGD-Evoked Neuron-Like Injury in Differentiated PC12 Cells. Neurochem Res 39, 2197–2210 (2014). https://doi.org/10.1007/s11064-014-1421-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-014-1421-0