Abstract
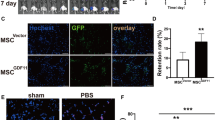
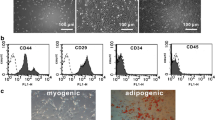
Gene and stem-cell therapies hold promise for the treatment of ischemic cardiovascular disease. Combined stem cell, chemokine, and angiogenic growth factor gene therapy could augment angiogenesis, and better improve heart function in the infarcted myocardium. In order to prove this action, we established the animal model of myocardial infarction (MI) was by occlusion of the left anterior descending artery in rats. Seven days after surgery, 5.0 × 106 Ad-EGFP–MSC, 5.0 × 106 Ad-SDF-1–MSC, 5.0 × 106 Ad-VEGF–MSC, or 5.0 × 106 Ad-SDF–VEGF–MSC (Ad-SDF-1–VEGF–MSC) suspension in 0.2 ml of serum-free medium was injected into four sites in the infarcted hearts. Results showed that MSCs transfected with Ad-VEGF and Ad-SDF-1 produced more SDF-1 and VEGF protein than MSCs alone, the increased protein levels of VEGF and SDF-1 activated Akt in MSCs transfected with Ad-VEGF and Ad-SDF-1, and improved the survival capability of the MSCs in vitro and in vivo. These transplanted cells showed that the characteristic phenotype of cardiomyocyte (e.g., cTnt) and endothelial cells (e.g., CD31). Four weeks after transplantation, reduced infarct size and fibrosis, greater vascular density, and a thicker left ventricle wall were observed in Ad-SDF–VEGF–MSC group. Measurement of hemodynamic parameters showed an improvement in left ventricular performance in Ad-SDF–VEGF–MSC group compared with other groups. These results demonstrated that combination of chemokine and angiogenic factor gene and stem cells could enhance angiogenesis and improves cardiac function after acute myocardial infarction in rats.






Similar content being viewed by others
Abbreviations
- MSCs:
-
Mesenchymal stem cells
- VEGF:
-
Vascular endothelial growth factor
- SDF-1α:
-
Stromal cell-derived factor-1 alpha
- EGFP:
-
Enhanced green fluorescent protein
- MI:
-
Myocardial infarction
- Ad:
-
Adenoviral
- LVSP:
-
Left ventricle systolic pressure
- LVEDP:
-
Left ventricle end-diastolic pressure
- +dP/dt max and −dP/dt max :
-
Rate of rise and fall of ventricular pressure
References
Schuleri KH, Boyle AJ, Hare JM (2007) Mesenchymal stem cells for cardiac regenerative therapy. Handb Exp Pharmacol 180:195–218
Roobrouck VD, Ulloa-Montoya F, Verfaillie CM (2008) Self-renewal and differentiation capacity of young and aged stem cells. Exp Cell Res 314:1937–1944
Yang J, Zhou W, Zheng W et al (2007) Effects of myocardial transplantation of marrow mesenchymal stem cells transfected with vascular endothelial growth factor for the improvement of heart function and angiogenesis after myocardial infarction. Cardiology 107:17–29
Mangi AA, Noiseux N, Kong D et al (2003) Mesenchymal stem cells modified with Akt prevent remodeling and restore performance of infarcted hearts. Nat Med 9:1195–1201
Zentilin L, Tafuro S, Zacchigna S et al (2006) Bone marrow mononuclear cells are recruited to the sites of VEGF-induced neovascularization but are not incorporated into the newly formed vessels. Blood 107:3546–3554
Carmeliet P (2000) VEGF gene therapy: stimulating angiogenesis or angioma-genesis? Nat Med 6:1102–1113
Grunewald M, Avraham I, Dor Y et al (2006) VEGF-induced adult neovascularization: recruitment, retention, and role of accessory cells. Cell 124:175–189
Tang JM, Xie QY, Pan GD et al (2006) Mesenchymal stem cells participate in angiogenesis and improve heart function in rat model of myocardial ischemia with reperfusion. Eur J Cardiothorac Surg 30:353–361
Tang JM, Wang JN, Guo LY et al (2010) Mesenchymal stem cells modified with stromal cell-derived factor 1α improve cardiac remodeling via paracrine activation of hepatocyte growth factor in a rat model of myocardial infarction. Mol Cells 29:1–10
Tang J, Wang J, Kong X et al (2009) Vascular endothelial growth factor promotes cardiac stem cell migration via the PI3 K/Akt pathway. Exp Cell Res 315:3521–3531
Hochman JS, Choo H (1987) Limitation of myocardial infarct expansion by reperfusion independent of myocardial salvage. Circulation 75:299–306
Chen HK, Hung HF, Shyu KG et al (2005) Combined cord blood stem cells and gene therapy enhances angiogenesis and improves cardiac performance in mouse after acute myocardial infarction. Eur J Clin Invest 35:677–686
Moore MA, Hattori K, Heissig B et al (2001) Mobilization of endothelial and hematopoietic stem and progenitor cells by adenovector-mediated elevation of serum levels of SDF-1, VEGF, and angiopoietin-1. Ann N Y Acad Sci 938:36–45 (discussion 45–47)
Abbott JD, Huang Y, Liu D et al (2004) Stromal cell-derived factor-1alpha plays a critical role in stem cell recruitment to the heart after myocardial infarction but is not sufficient to induce homing in the absence of injury. Circulation 110:3300–3305
Kryczek I, Lange A, Mottram P et al (2005) CXCL12 and vascular endothelial growth factor synergistically induce neoangiogenesis in human ovarian cancers. Cancer Res 65:465–472
Shyu KG, Wang BW, Hung HF et al (2006) Mesenchymal stem cells are superior to angiogenic growth factor genes for improving myocardial performance in the mouse model of acute myocardial infarction. J Biomed Sci 13:47–58
Byrne AM, Bouchier-Hayes DJ, Harmey JH et al (2005) Angiogenic and cell survival functions of vascular endothelial growth factor (VEGF). J Cell Mol Med 9:777–794
Pasha Z, Wang Y, Sheikh R et al (2008) Preconditioning enhances cell survival and differentiation of stem cells during transplantation in infarcted myocardium. Cardiovasc Res 77:134–142
Chen M, Xie HQ, Deng L et al (2008) Stromal cell-derived factor-1 promotes bone marrow-derived cells differentiation to cardiomyocyte phenotypes in vitro. Cell Prolif 41:336–347
Zhu Y, Liu T, Song K et al (2009) ADSCs differentiated into cardiomyocytes in cardiac microenvironment. Mol Cell Biochem 24:117–129
Gnecchi M, He H, Noiseux N et al (2006) Evidence supporting paracrine hypothesis for Akt-modified mesenchymal stem cell-mediated cardiac protection and functional improvement. FASEB J 20:661–669
Kinnaird T, Stabile E, Burnett MS et al (2004) Marrow-derived stromal cells express genes encoding a broad spectrum of arteriogenic cytokines and promote in vitro and in vivo arteriogenesis through paracrine mechanisms. Circ Res 94:678–685
Zhang D, Zhang F, Zhang Y et al (2007) Erythropoietin enhances the angiogenic potency of autologous bone marrow stromal cells in a rat model of myocardial infarction. Cardiology 108:228–236
Urbanek K, Rota M, Cascapera S et al (2005) Cardiac stem cells possess growth factor-receptor systems that after activation regenerate the infarcted myocardium, improving ventricular function and long-term survival. Circ Res 97:663–673
Tang YL, Zhu W, Cheng M et al (2009) Hypoxic preconditioning enhances the benefit of cardiac progenitor cell therapy for treatment of myocardial infarction by inducing CXCR4 expression. Circ Res 104:1209–1216
Moore KA, Lemischka IR (2006) Stem cells and their niches. Science 311:1880–1885
Jin DK, Shido K, Kopp HG et al (2006) Cytokine-mediated deployment of SDF-1 induces revascularization through recruitment of CXCR4+ hemangiocytes. Nat Med 12:557–567
Tang J, Wang J, Yang J et al (2009) Mesenchymal stem cells over-expressing SDF-1 promote angiogenesis and improve heart function in experimental myocardial infarction in rats. Eur J Cardiothorac Surg 36:644–650
Zhang M, Mal N, Kiedrowski M et al (2007) SDF-1 expression by mesenchymal stem cells results in trophic support of cardiac myocytes after myocardial infarction. FASEB J 21:3197–3207
Hiasa K, Ishibashi M, Ohtani K et al (2004) Gene transfer of stromal cell-derived factor-1alpha enhances ischemic vasculogenesis and angiogenesis via vascular endothelial growth factor/endothelial nitric oxide synthase-related pathway: next-generation chemokine therapy for therapeutic neovascularization. Circulation 109:2454–2461
Ackah E, Yu J, Zoellner S et al (2005) Akt1/protein kinase B alpha is critical for ischemic and VEGF-mediated angiogenesis. J Clin Invest 115:2119–2127
Unzek S, Zhang M, Mal N et al (2007) SDF-1 recruits cardiac stem cell-like cells that depolarize in vivo. Cell Transplant 16:879–886
Li RK, Mickle DA, Weisel RD et al (2001) Optimal time for cardiomyocyte transplantation to maximize myocardial function after left ventricular injury. Ann Thorac Surg 72:1957–1963
Kissel CK, Lehmann R, Assmus B et al (2007) Selective functional exhaustion of hematopoietic progenitor cells in the bone marrow of patients with postinfarction heart failure. J Am Coll Cardiol 49:2341–2349
Dimmeler S, Leri A (2008) Aging and disease as modifiers of efficacy of cell therapy. Circ Res 102:1319–1330
Torella D, Ellison GM, Méndez-Ferrer S et al (2006) Resident human cardiac stem cells: role in cardiac cellular homeostasis and potential for myocardial regeneration. Nat Clin Pract Cardiovasc Med 3(Suppl 1):S8–S13
Acknowledgments
We thank Professor Yongsheng Ren for his helpful instruction in heart function evaluation. We are grateful to Long Chen for help with immunofluorescence staining. We thank international science editing corporation for his helpful instruction in the article edition. This study was supported by grants from the National Natural Science Foundation of China (30700306), Hubei Natural Science Foundation (2005ABA079), Hubei Health Department Science Foundation (JX3B29), Hubei Education Department Science Foundation (Q200524003, B200624006), Yun Yang Medical College Science Foundation (2005QDJ01), and Shiyan Renmin Hospital Science Foundation, China.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tang, J., Wang, J., Zheng, F. et al. Combination of chemokine and angiogenic factor genes and mesenchymal stem cells could enhance angiogenesis and improve cardiac function after acute myocardial infarction in rats. Mol Cell Biochem 339, 107–118 (2010). https://doi.org/10.1007/s11010-009-0374-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-009-0374-0