Abstract
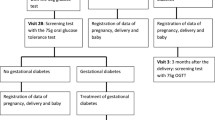
To test the feasibility of conducting a pragmatic randomized controlled trial (RCT) comparing the International Association of Diabetes in Pregnancy Study Groups (IADPSG) versus Carpenter–Coustan diagnostic criteria for gestational diabetes (GDM), and to examine patient and provider views on GDM screening. A single-blinded pragmatic pilot RCT. Participants with a singleton pregnancy between 24 and 28 weeks gestation received a 50 g oral glucose challenge test and if the value was <200 mg/dL were randomized to either the 2 h 75 g OGTT using the IADPSG criteria or the 3 h 100 g OGTT using the Carpenter–Coustan criteria. Primary outcome was the feasibility of randomization and screening. Secondary outcomes included patient and provider views (or preferences) on GDM testing. Sixty-eight women were recruited, 48 (71 %) enrolled and 47 (69 %) were randomized. Participants in both study arms identified the main challenges to GDM testing to be: drinking the glucola, fasting prior to testing, waiting to have blood drawn, and multiple venipuntures. Women in both study arms would prefer the 2 h 75 g OGTT or whichever test is recommended by their doctor in a future pregnancy. Physicians and nurse midwives endorsed screening and were comfortable with being blinded to the GDM testing strategy and results values. Both pregnant women and providers value GDM screening, and pregnant women can be recruited to a blinded, randomized GDM screening trial with minimal attrition and missing data.

Similar content being viewed by others
References
Hod, M., Merlob, P., Friedman, S., Schoenfeld, A., & Ovadia, J. (1991). Gestational diabetes mellitus: A survey of perinatal complications in the 1980s. Diabetes, 40(Suppl 2), 74–78.
Yogev, Y., Xenakis, E. M., & Langer, O. (2004). The association between preeclampsia and the severity of gestational diabetes: The impact of glycemic control. American Journal of Obstetrics and Gynecology, 191(5), 1655–1660.
Practice Bulletin No. 137. (2013). Gestational diabetes mellitus. Obstetrics and Gynecology, 122(2 Pt 1), 406–416.
Metzger, B. E., Lowe, L. P., Dyer, A. R., Trimble, E. R., Chaovarindr, U., Coustan, D. R., et al. (2008). Hyperglycemia and adverse pregnancy outcomes. New England Journal of Medicine, 358(19), 1991–2002.
Metzger, B. E., Gabbe, S. G., Persson, B., Buchanan, T. A., Catalano, P. A., Damm, P., et al. (2010). International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care, 33(3), 676–682.
Gradmark, A., Pomeroy, J., Renstrom, F., Steiginga, S., Persson, M., Wright, A., et al. (2011). Physical activity, sedentary behaviors, and estimated insulin sensitivity and secretion in pregnant and non-pregnant women. BMC Pregnancy Childbirth, 11, 44.
Sacks, D. A., Hadden, D. R., Maresh, M., Deerochanawong, C., Dyer, A. R., Metzger, B. E., et al. (2012). Frequency of gestational diabetes mellitus at collaborating centers based on IADPSG consensus panel-recommended criteria: The Hyperglycemia and Adverse Pregnancy Outcome (HAPO) Study. Diabetes Care, 35(3), 526–528.
Carpenter, M. W., & Coustan, D. R. (1982). Criteria for screening tests for gestational diabetes. American Journal of Obstetrics and Gynecology, 144(7), 768–773.
Alexander, G. R., Himes, J. H., Kaufman, R. B., Mor, J., & Kogan, M. (1996). A United States national reference for fetal growth. Obstetrics and Gynecology, 87(2), 163–168.
ACOG Practice Bulletin. (2002). Diagnosis and management of preeclampsia and eclampsia. Number 33, January 2002. Obstetrics and Gynecology, 99(1), 159–167.
Rasmussen, K. M., Yaktine, A. L., Commitee to Re-examine IOM Pregnancy Weight Guidelines, Institue of Medicine, National Research Council. (2009). Weight gain during pregnancy: Reexamining the guidelines. The National Academies Press.
Landon, M. B., Spong, C. Y., Thom, E., Carpenter, M. W., Ramin, S. M., Casey, B., et al. (2009). A multicenter, randomized trial of treatment for mild gestational diabetes. The New England journal of medicine, 361(14), 1339–1348.
Crowther, C. A., Hiller, J. E., Moss, J. R., McPhee, A. J., Jeffries, W. S., & Robinson, J. S. (2005). Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. The New England journal of medicine, 352(24), 2477–2486.
Moses, R. G., Webb, A. J., Comber, C. D., Walton, J. G., Coleman, K. J., Davis, W. S., et al. (2003). Gestational diabetes mellitus: Compliance with testing. The Australian & New Zealand Journal of Obstetrics & Gynaecology, 43(6), 469–470.
Agarwal, M. M., Punnose, J., & Dhatt, G. S. (2004). Gestational diabetes: Problems associated with the oral glucose tolerance test. Diabetes Research and Clinical Practice, 63(1), 73–74.
Meltzer, S. J., Snyder, J., Penrod, J. R., Nudi, M., & Morin, L. (2010). Gestational diabetes mellitus screening and diagnosis: A prospective randomised controlled trial comparing costs of one-step and two-step methods. BJOG: An International Journal of Obstetrics and Gynaecology, 117(4), 407–415.
Acknowledgments
This work was supported by funding from Agency for Healthcare Research and Quality Comparative Effectiveness Grant # AHRQ K12 HS019461 and via internal grants/funds received from the University of Pittsburgh Departments of Medicine and Obstetrics, Gynecology, and Reproductive Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Scifres, C.M., Abebe, K.Z., Jones, K.A. et al. Gestational Diabetes Diagnostic Methods (GD2M) Pilot Randomized Trial. Matern Child Health J 19, 1472–1480 (2015). https://doi.org/10.1007/s10995-014-1651-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10995-014-1651-4