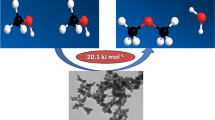
Abstract
Highly porous nanocrystalline alumina was synthesized using two different precipitation processes and precipitating agents, which were characterized by powder X-ray diffraction (XRD), scanning electron microscopy (SEM) and porosimetry analyses. Different precipitating agents yielded nanocrystalline alumina catalysts with different morphologies and textural properties. Batch precipitation using sodium bicarbonate at constant pH resulted in a highly porous nanocrystalline γ-alumina catalyst, having surface area of 351.47 m2 g−1, total pore volume of 1.68 cm3 g−1 and mean pore diameter of 19.17 nm. The mean crystallite size was also determined to be 3.8 nm, based on the XRD results. Catalytic performance of the synthesized catalysts was investigated in a slurry reactor for dehydration of methanol to dimethyl ether (DME). Commercial alumina was also investigated in order to compare results with those of synthesized materials. The aforementioned γ-alumina synthesized catalyst exhibited a much better catalytic performance than other synthesized catalysts in this research as well as the commercial alumina. Ultimately, repetition of experiments emphasized the reasonable stability of this catalyst.




Similar content being viewed by others
References
F. Yaripour, F. Baghaei, I. Schmidt, J. Perregaard, Catal. Commun. 6, 147–152 (2005)
S. Jiang, J.S. Hwang, T. Jin, T. Cai, W. Cho, Y.S. Baek, S.E. Park, Bull Korean Chem. Soc. 25, 185–189 (2004)
C.W. Seo, K.D. Jung, K. Lee, K.S. Yoo, Ind. Eng. Chem. Res. 47, 6573–6578 (2008)
G. Ertl, H. Knözinger, F. Schüth, J. Weitkamp (eds.), Handbook of Heterogeneous Catalysis, 2nd edn. (WILEY-VCH, Weinheim, 2008)
Q. Sun, Y. Fu, H. Yang, A. Auroux, J. Shen, J. Mol. Catal. A: Chem. 275, 183–193 (2007)
S.D. Kim, S.C. Baek, Y.J. Lee, K.W. Jun, M.J. Kim, I.S. Yoo, Appl. Catal. A 309, 139–143 (2006)
J. Khom-in, P. Praserthdam, J. Panpranot, O. Mekasuwandumrong, Catal. Commun. 9, 1955–1958 (2008)
V. Siva Kumar, A.H. Padmasri, C.V.V. Satyanarayana, I. Ajit Kumar Reddy, B. David Raju, K.S. Rama Rao, Catal. Commun. 7, 745–751 (2006)
F. Yaripour, M. Mollavali, Sh Mohammadi Jam, H. Atashi, Energy Fuels 23, 1896–1900 (2009)
Y. Fu, T. Hong, J. Chen, A. Auroux, J. Shen, Thermochim. Acta. 434, 22–26 (2005)
H.S. Potdar, K.W. Jun, J.W. Bae, S.M. Kim, Y.J. Lee, Appl. Catal. A. 321, 109–116 (2007)
S. Wang, X. Li, S. Wang, Y. Li, Y. Zhai, Mater. Lett. 62, 3552–3554 (2008)
L.D. Mitchell, P.S. Whitfield, J. Margeson, J.J. Beaudoin, J. Mater. Sci. Lett. 21, 1773–1775 (2002)
Q. Yang, Y. Deng, W. Hu, Ceram. Int. 35, 531–535 (2009)
C. Lesaint, G. Kleppa, D. Arla, W.R. Glomm, G. Øye, Microporous Mesoporous Mater. 119, 245–251 (2009)
L. Wana, H. Fu, K. Shi, X. Tian, Microporous Mesoporous Mater. 115, 301–307 (2008)
L. Zhong, Y. Zhang, F. Chen, Y. Zhang, Microporous Mesoporous Mater. 142, 740–744 (2011)
Y. Rozita, R. Brydson, A.J. Scott, J. Phys.: Conf. Ser. 241, 1–4 (2010)
J.B. Condon, Surface Area and Porosity Determinations by Physisorption: Measurements and Theory, 1st edn. (Elsevier, Amsterdam, 2006)
O. Mekasuwandumrong, P.L. Silveston, P. Praserthdam, M. Inoue, V. Pavarajarn, W. Tanakulrungsank, Inorg. Chem. Commun. 6, 930–934 (2003)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zaherian, A., Kazemeini, M., Aghaziarati, M. et al. Synthesis of highly porous nanocrystalline alumina as a robust catalyst for dehydration of methanol to dimethyl ether. J Porous Mater 20, 151–157 (2013). https://doi.org/10.1007/s10934-012-9584-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-012-9584-z