Abstract
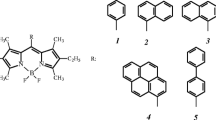
Three boron-dipyrrine (BODIPY) based dyes with bulky substituents in 8-position of dipyrrin ligand have been synthesized and characterized. Photophysical properties of the obtained compounds have been investigated in different individual solvents and solvent mixtures. Investigated compounds was found to be intensive fluorescent molecular rotors. The influence of different solvent parameters and the substituent nature on rotor characteristics have been observed and discussed. Minor changes in the nature of 8-substituent does not influence the spectral properties, but the presence of nitrogen donor atom in the phenyl substituent could be used for the sensing of the donor-acceptor interactions with solvent or dissolved compounds. The new approach of spectral properties correlation with solvent parameters was proposed, the viscosity parameter should be taken into account in case of BODIPYs with bulky substituents. The intensity of fluorescence molecular rotor properties decrease gradually with the viscosity increase above 1 cP.








Similar content being viewed by others
References
Wang LY, Fang GP, Cao DR (2015) A novel phenol-based BODIPY chemosensor for selective detection Fe3+ with colorimetric and fluorometric dual-mode. Sensors Actuators B Chem 207:849–857
He Y, Feng RK, Yi YR, Liu ZX (2014) Recent progress in the research of borondipyrromethene-based fluorescent ion chemosensor. Chinese J Org Chem 34:2236–2248
Madhu S, Kalaiyarasi R, Basu SK, Jadhavb S, Ravikanth M (2014) A boron-dipyrrin–mercury (II) complex as a fluorescence turn-on sensor for chloride and applications towards logic gates. J Mater Chem C 2:2534–2544
Liu J, He XX, Zhang J, He T, Huang LQ, Shen JQ, Li D, Qiu HY, Yin SC (2015) A BODIPY derivative for colorimetric and fluorometric sensing of fluoride ion and its logic gates behavior. Sensors Actuators B Chem 208:538–545
Gareis T, Huber C, Wolfbeis OS, Daub J (1997) Phenol/phenolate-dependent on/off switching of the luminescence of 4,4-difluoro-4-Bora-3a,4a-diaza-S-indacenes. Chem Commun 18:1717–1718
Werner T, Huber C, Heinl S, Kollmannsberger M, Daub J, Wolfbeis OS (1997) Novel optical Ph-sensor based on a boradiaza-indacene derivative. Fresenius J Anal Chem 359:150–154
Sui B, Tang S, Liu T (2014) Novel BODIPY-based fluorescence turn-on sensor for Fe3+ and its bioimaging application in living cells. ACS Appl Mater Interfaces 21:18408–18412
Andrea B, Costero M, Salvador AG (2014) Chromo-fluorogenic BODIPY-complexes for selective detection of V-type nerve agent surrogates. Chem Commun 50:13289–13291
Fengling S, Yingying X, Xu W, Jingyun W, Xiaoqing X, Xiaojun P (2014) Ratiometric fluorescent probe based on novel red-emission BODIPY for determination of bovine serum albumin. Chem Res Chin Univ 30:738–742
Ma D, Kim D, Seo E, Lee S, Ahn KH (2015) Ratiometric fluorescence detection of cysteine and homocysteine with a BODIPY dye by mimicking the native chemical ligation. Analyst 140:422–427
Bahaidarah E, Harriman A, Stachelek P, Rihn S, Heyerb E, Ziessel R (2014) Fluorescent molecular rotors based on the BODIPY motif: effect of remote substituents. Photochem Photobiol Sci 10:1397–1401
Marfin YS, Merkushev DA, Levshanov GA, Rumyantsev EV (2014) Fluorescent properties of 8-phenylBODIPY in ethanol – ethylene glycol mixed solutions. J Fluoresc 24:1613–1619
Rongrong H, Lager E, Aguilar-Aguilar A, Liu J, Lam JWY, Sung HHY, Williams ID (2009) Twisted intramolecular charge transfer and aggregation-induced emission of BODIPY derivatives. J Phys Chem C 113:15845–15853
Dal Molin M, Verolet Q, Soleimanpour S, Matile S (2015) Mechanosensitive membrane probes. Chemistry 21:6012–6021
Haidekker M, Theodorakis E (2010) Environment-sensitive behavior of fluorescent molecular rotors. J Biol Eng 4:11–25
Benniston AC (2014) Monitoring rheological properties in biological systems by fluorescence spectroscopy using borondipyrromethene (Bodipy) dyes: a mini review. J Anal Bioanal Tech 5:1–11
Forster T, Hoffmann G (1971) Effect of viscosity on the fluorescence quantum yield of some dye systems. Z Phys Chem 75:63–76 (in German)
Weissberger A, Proskauer ES, Riddick JA, Toops EE (1955) Organic solvents. Physical properties and methods of purification. Interscience Publishers Inc, New York
Bruttel P, Schlink R (2003) Water determination by Karl Fischer titration. Metrohm Monogr 8(5003):2003–2009.
Wolfbeis OS (2008) Standardization and quality assurance in fluorescence measurements techniques. Springer Ser Fluoresc. doi:10.1007/978-3-540-75207-3
Marfin YS, Rumyantsev EV, Ya SF, Antina EV (2012) Relationship between the spectral properties of solutions of borofluoride complex of alkylated dipyrromethene and the physicochemical parameters of solvents. Russ J Phys Chem A 86:1068–1072
Arbeloa TL (1999) Correlations between photophysics and lasing properties of dipyrromethene–bf2 dyes in solution. Chem Phys Lett 299:315–321
Volchkov VV, Khimich MN, Ya MM, Uzhinov BM (2013) A fluorescence study of the excited-state dynamics of boron dipyrrin molecular rotors. High Energy Chem 47:224–229
Jeyanthy S, Lichlyter D, Wright NE, Dakanali M, Haidekker M, Theodorakis E (2010) Molecular rotors: synthesis and evaluation as viscosity sensors. Tetrahedron 66:2582–2588
Giulio M, Martinelli E, Ruggeri G, Galli G, Pucci A (2015) Julolidine fluorescent molecular rotors as vapour sensing probes in polystyrene films. Dye Pigment 113:47–54
Ling KH, Kirmaier C, Yu L, Thamyongkit P, Youngblood WJ, Calder ME, Ramos L et al (2005) Structural control of the photodynamics of boron - dipyrrin complexes. J Phys Chem B 109:20433–20443
Gordon H, Ruseckas A, Harriman A, Samuel IDW (2011) Conformational effects on the dynamics of internal conversion in boron dipyrromethene dyes in solution. Angew Chem Int Ed 50:6634–6637
Rezende LC, Vaidergorn M, Moraes JCB, da Silva Emery F (2014) Synthesis, photophysical properties and solvatochromism of meso-substituted tetramethyl BODIPY dyes. J Fluoresc 24:257–266
Banuelos J, Lopez Arbeloa F, Arbeloa T, Salleres S, Vilas JL, Amat-Guerri F, Liras M, Lopez Arbeloa I (2008) Photophysical characterization of new 3-amino and 3-acetamido BODIPY dyes with solvent sensitive properties. J Fluoresc 18:899–907
Lager E, Liu J, Aguilar-Aguilar A, Zhong Tang B, Pena-Cabrera E (2009) Novel meso-polyarylamine-BODIPY hybrids: synthesis and study of their optical properties. J Org Chem 74:2053–2058
Banuelos PJ, Lopez Arbeloa F, Martinez Martinez V, Lopez Arbeloa T, Lopez Arbeloa I (2004) Photophysical properties of the pyrromethene 597 dye: solvent effect. J Phys Chem A 108:5503–5508
Marfin YS, Rumyantsev EV (2014) Analysis of solvation and structural contributions in spectral characteristics of dipyrrin Zn(II) complexes. Spectrochim Acta A Mol Biomol Spectrosc 130:423–428
Chaudhuri T (2010) Photophysical properties of the 8-phenyl analogue of PM567: a theoretical rationalization. Spectrochim Acta A Mol Biomol Spectrosc 75:739–744
Alamiry MAH, Benniston AC, Copley G, Elliott KJ, Harriman A, Stewart B, Zhi YG (2008) A molecular rotor based on an unhindered boron dipyrromethene (Bodipy) dye. Chem Mater 20:4024–4032
Krestov GA, Afanas’yev VN, Yefremova LS (1988) Physical and chemical properties of binary solvents: handbook. Khimiya, Leningrad, p 688. (in Russian)
Acknowledgments
The work was supported by the grants of the Russian Foundation for Basic Research (No 14-03-31888, No 15-33-20002) and the grant of the President of the Russian Federation for young scientists and graduate students engaged in advanced research and development in priority directions of modernization of the Russian economics (2013 – 2015) (Grant No. SP-1742.2013.1).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marfin, Y.S., Merkushev, D.A., Usoltsev, S.D. et al. Fluorescent Properties of 8-Substituted BODIPY Dyes: Influence of Solvent Effects. J Fluoresc 25, 1517–1526 (2015). https://doi.org/10.1007/s10895-015-1643-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-015-1643-9