Abstract
Purpose
Whether there are differences in the pregnancy outcomes of blastocysts cryopreserved during different developmental stages remains under debate because the results among studies are inconsistent. We analyzed blastocyst quality and pregnancy outcomes by considering blastocyst euploidy and investigated the differences in the development potential between blastocysts of different developmental stages (frozen-thawed day 5 [D5] and day 6 [D6] cycles) and their relationship with clinical pregnancy outcomes.
Methods
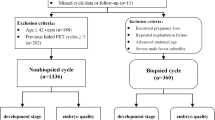
In total, 1374 D5 and 255 D6 frozen-thawed blastocyst transfer cycles were retrospectively analyzed. Additionally, the chromosome euploidy and clinical pregnancy rates of 237 blastocysts from 50 pre-implantation genetic diagnosis (PGS) cycles were statistically analyzed. The corresponding euploidy rate and pregnancy outcomes of the D5 and D6 blastocyst transfers were also compared.
Results
The clinical pregnancy rate (47.2 vs 40.0 %; P = 0.04) and implantation rate (34.2 vs 28.8 %; P = 0.03) of the D5 blastocysts were higher than were those of the D6 blastocysts. However, the clinical pregnancy rate (52.4 vs 52.6 %; P = 0.97) and implantation rate (38.9 vs 35.6 %; P = 0.39) of the high-quality D5 blastocysts did not significantly differ from those of the high-quality D6 blastocysts. Analysis of blastocyst euploidy in 237 blastocysts examined in 50 PGS cycles showed that the euploidy rates of the D5 and D6 blastocysts were both 48.1 % (P = 0.99). The clinical pregnancy rate of the D5 blastocysts (48.5 vs 17.6 %; P = 0.03) was higher than that of the D6 blastocysts. The euploidy rates (55.2 vs 55.3 %; P = 0.99) and clinical pregnancy rates (60.0 vs 42.9 %; P = 0.77) of the high-quality D5 and D6 blastocysts did not differ. The euploidy rate (55.3 vs 41.5 %, P = 0.03) and clinical pregnancy rate (54.5 vs 25.0 %, P = 0.03) of the high-quality blastocysts were higher than were those of the poor-quality blastocysts.
Conclusions
The euploidy rates between the D5 and D6 blastocysts did not differ. High-quality D6 blastocysts in frozen-thawed cycles had similar developmental potential and pregnancy outcomes compared to those of high-quality D5 blastocysts. The quality of the blastocysts was an important factor that affected the pregnancy outcomes of the frozen-thawed cycles.



Similar content being viewed by others
References
Glujovsky D, Blake D, Farquhar C, Bardach A. Cleavage stage versus blastocyst stage embryo transfer in assisted reproductive technology. Cochrane Database Syst Rev. 2012;7:CD002118.
Sullivan EA, Wang YA, Hayward I, Chambers GM, Illingworth P, McBain J, et al. Single embryo transfer reduces the risk of perinatal mortality, a population study. Hum Reprod. 2012;27:3609–15.
Maheshwari A, Griffiths S, Bhattacharya S. Global variations in the uptake of single embryo transfer. Hum Reprod Update. 2011;17(1):107–20.
Barrenetxea G, Lopez de Larruzea A, Ganzabal T, Jimenez R, Carbonero K, Mandiola M. Blastocyst culture after repeated failure of cleavage-stage embryo transfers: a comparison of day 5 and day 6 transfers. Fertil Steril. 2005;83:49–53.
Utsunomiya T, Ito H, Nagaki M, Sato J. A prospective, randomized study: day 3 versus hatching blastocyst stage. Hum Reprod. 2004;19:1598–603.
Shapiro B, Daneshmand S, Garner F, Aguirre M, Ross R. Contrasting patterns in in vitro fertilization pregnancy rates among fresh autologous, fresh oocyte donor, and cryopreserved cycles with the use of day 5 or day 6 blastocysts may reflect differences in embryo-endometrium synchrony. Fertil Steril. 2008;89:20–6.
Van Voorhis BJ, Dokras A. Delayed blastocyst transfer: is the window shutting? Fertil Steril. 2008;89(1):31–2.
Marek DM, Langley MT, McKean C, et al. Frozen embryo transfer (FET) of day 5 blastocyst embryos compared to transfer of day 6 blastocyst embryos. Fertil Steril. 2000;74(1):S52–3.
Kovalevsky G, Carney SM, Morrison LS, et al. Should embryos developing to blastocysts on day 7 be cryopreserved and transferred: an analysis of pregnancy and implantation rates. Fertil Steril. 2013;100:1008–12.
Behr B, Gebhardt J, Lyon J, Milki A. Factors relating to a successful cryopreserved blastocyst transfer program. Fertil Steril. 2002;77:697–9.
Hiraoka K, Hiraoka K, Miyazaki M, Fukunaga E, Horiuchi T. Perinatal outcomes following transfer of human blastocysts vitrified at day 5, 6 and 7. J Exp Clin Assist Reprod. 2009;6:4.
Levens E, Whitcomb B, Hennessy S, James A, Yauger B, Larsen F. Blastocyst development rate impacts outcome in cryopreserved blastocyst transfer cycles. Fertil Steril. 2008;90:2138–43.
Gardner DK, Lane M, Stevens J, et al. Blastocyst score affects implantation and pregnancy outcome: towards a single blastocyst transfer. Fertil Steril. 2000;73(6):1155–8.
Mukaida T, Goto OC, et al. Artificial shrinkage of blastocoels using either a micro-needle or a laser pulse prior to the cooling steps of vitrification improves survival rate and pregnancy outcome of vitrified human blastocysts. Hum Repord. 2006;21(12):3246–52.
Kuwayama M, Vajta G, Kato O, et al. Highly efficient vitrification method for cryopreservation of human oocytes. Reprod Biomed Online. 2005;11(3):300–8.
Li G, He N, Jin H, Liu Y, et al. The influence of single nucleotide polymorphism microarray-based molecular karyotype on preimplantation embryonic development potential. PLoS One. 2015;10(9):e0138234.
Stanger J, Wong J, Conceicao J, Yovich J. Vitrification of human embryos previously cryostored by either slow freezing or vitrification results in high pregnancy rates. Reprod Biomed Online. 2012;24(3):314–20.
Liebermann J. Vitrification of human blastocysts: an update. Reprod Biomed Online. 2009;19 Suppl 4:4328.
Liebermann J, Tucker M. Comparison of vitrification and conventional cryopreservation of day 5 and 6 blastocysts during clinical application. Fertil Steril. 2006;86:20–6.
El-Toukhy T, Wharf E, Walavalkar R, Singh A, Bolton V, Khalaf Y, et al. Delayed blastocyst development does not influence the outcome of frozen thawed transfer cycles. BJOG. 2011;118:1551–6.
Sunkara S, Siozos A, Bolton V, Khalaf Y, Braude P, El-Toukhy T. The influence of delayed blastocyst formation on the outcome of frozen-thawed blastocyst transfer: a systematic review and meta-analysis. Hum Reprod. 2010;25(8):1906–15.
Capalbo A, Rienzi L, Cimadomo D, Maggiulli R, Elliott T, Wright G, et al. Correlation between standard blastocyst morphology, euploidy and implantation: an observational study in two centers involving 956 screened blastocysts. Hum Reprod. 2014;29(6):1173–81.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule The euploidy rates between the D5 and D6 blastocysts did not differ. High-quality D5 and D6 blastocyst transfers had similar pregnancy outcomes.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
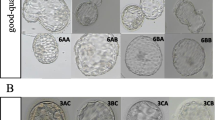
Different grades of blastocysts. A-O represent blastocysts of 3AA,3AB, 3BB,4AA,4AB,4BB,5BB,6BB,3 AC,3BC,4 AC,4BC,4CB, 4CC and 5BC, respectively. Bar = 50 μm. (DOCX 1118 kb)
Rights and permissions
About this article
Cite this article
Yang, H., Yang, Q., Dai, S. et al. Comparison of differences in development potentials between frozen-thawed D5 and D6 blastocysts and their relationship with pregnancy outcomes. J Assist Reprod Genet 33, 865–872 (2016). https://doi.org/10.1007/s10815-016-0712-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-016-0712-6