Abstract
Purpose
This study aimed to induce follicular wave emergence (FWE) using pharmacological (recombinant hCG administration) or mechanical (aspiration of dominant follicle) interventions in infertile women.
Methods
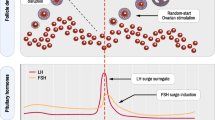
Sixteen infertile women (≤35 years) with indications for in vitro fertilization due to tubal and/or male factor infertility were randomized into three groups: control (n = 6), pharmacological (n = 5) and mechanical (n = 5) groups. Women in both experimental groups underwent serial transvaginal sonograms (TVS) from menstrual cycle day 10 until identification of a dominant follicle ≥15 mm. Women in the pharmacological group received 250 μg of recombinant-hCG to induce ovulation, and resumed serial TVS 2 days later. In the mechanical group, dominant and subordinate follicles ≥10 mm were aspirated, and daily TVS was resumed on the following day. An increased pool of follicles ≥5 and ≤9 mm after interventions characterized FWE. Women in the control group underwent ovulation induction (OI) with 150 IU/day of recombinant follicle-stimulating hormone started on menstrual cycle day 3 (D3). OI was started on the day of FWE in the experimental groups. Endometrial asynchrony with development of the embryo was expected in the experimental groups. Therefore, all viable embryos were cryopreserved and transferred in an endometrial-stimulated cycle.
Results
The number of follicles ≥5 and ≤9 mm increased after the interventions in both experimental groups (p < .001), indicating induction of FWE. OI outcomes were similar among the groups.
Conclusions
The pharmacological and mechanical interventions are efficient in inducing FWE; outcomes of OI synchronized with FWE should be further investigated.


Similar content being viewed by others
References
Macklon NS, Stouffer RL, Giudice LC, Fauser BC. The science behind 25 years of ovarian stimulation for in vitro fertilization. Endocr Rev. 2006;27(2):170–207.
Gougeon A. Dynamics of follicular growth in the human: a model from preliminary results. Hum Reprod. 1986;1(2):81–7.
Gougeon A. Human ovarian follicular development: from activation of resting follicles to preovulatory maturation. Ann Endocrinol (Paris). 2010;71(3):132–43.
Baerwald AR, Adams GP, Pierson RA. Ovarian antral folliculogenesis during the human menstrual cycle: a review. Hum Reprod Update. 2012;18(1):73–91.
Fauser BC, Van Heusden AM. Manipulation of human ovarian function: physiological concepts and clinical consequences. Endocr Rev. 1997;18(1):71–106.
Ginther OJ. Ultrasonic imaging of equine ovarian follicles and corpora lutea. Vet Clin North Am Equine Pract. 1988;4(2):197–213.
Nasser LF, Adams GP, Bo GA, Mapletoft RJ. Ovarian superstimulatory response relative to follicular wave emergence in heifers. Theriogenology. 1993;40(4):713–24.
Menchaca A, Pinczak A, Rubianes E. Follicular recruitment and ovulatory response to FSH treatment initiated on day 0 or day 3 postovulation in goats. Theriogenology. 2002;58(9):1713–21.
Mapletoft RJ, Bó GA, Baruselli PS. Control of ovarian function for assisted reproductive technologies in cattle. 2009.
Bó GA, Guerrero DC, Tríbulo A, Tríbulo H, Tríbulo R, Rogan D, et al. New approaches to superovulation in the cow. Reprod Fertil Dev. 2010;22(1):106–12.
Menchaca A, Vilariño M, Crispo M, de Castro T, Rubianes E. New approaches to superovulation and embryo transfer in small ruminants. Reprod Fertil Dev. 2010;22(1):113–8.
Baerwald AR, Adams GP, Pierson RA. A new model for ovarian follicular development during the human menstrual cycle. Fertil Steril. 2003;80(1):116–22.
Baerwald AR, Adams GP, Pierson RA. Characterization of ovarian follicular wave dynamics in women. Biol Reprod. 2003;69(3):1023–31.
de Mello Bianchi PH, Serafini P, Monteiro da Rocha A, Assad Hassun P, da Motta EL A, Sampaio Baruselli P, et al. Review: follicular waves in the human ovary: a new physiological paradigm for novel ovarian stimulation protocols. Reprod Sci. 2010;17(12):1067–76.
Ginther OJ, Gastal EL, Gastal MO, Bergfelt DR, Baerwald AR, Pierson RA. Comparative study of the dynamics of follicular waves in mares and women. Biol Reprod. 2004;71(4):1195–201.
Adams GP, Singh J, Baerwald AR. Large animal models for the study of ovarian follicular dynamics in women. Theriogenology. 2012.
Serafini P, Yadid I, Motta EL, Alegretti JR, Fioravanti J, Coslovsky M. Ovarian stimulation with daily late follicular phase administration of low-dose human chorionic gonadotropin for in vitro fertilization: a prospective, randomized trial. Fertil Steril. 2006;86(4):830–8.
Embryology ASiRMaESIGo. The Istanbul consensus workshop on embryo assessment: proceedings of an expert meeting. Hum Reprod. 2011;26(6):1270–83.
Kirkwood BR, Sterne JAC. Essential medical statistics. 2nd ed. Massachusetts: Blackwell Science; 2006.
Neter J, Kutner MH, Nachtsheim CJ, Wasserman W. Applied linear statistical models. 4th ed. Illinois; 1996.
Baerwald A, Anderson P, Yuzpe A, Case A, Fluker M. Synchronization of ovarian stimulation with follicle wave emergence in patients undergoing in vitro fertilization with a prior suboptimal response: a randomized, controlled trial. Fertil Steril. 2012.
Fischer RA, Nakajima ST, Gibson M, Brumsted JR. Ovulation after intravenous and intramuscular human chorionic gonadotropin. Fertil Steril. 1993;60(3):418–22.
Ludwig M, Doody KJ, Doody KM. Use of recombinant human chorionic gonadotropin in ovulation induction. Fertil Steril. 2003;79(5):1051–9.
Andersen AG, Als-Nielsen B, Hornnes PJ, Franch AL. Time interval from human chorionic gonadotrophin (HCG) injection to follicular rupture. Hum Reprod. 1995;10(12):3202–5.
Cakmak H, Katz A, Cedars MI, Rosen MP. Effective method for emergency fertility preservation: random-start controlled ovarian stimulation. Fertil Steril. 2013;100(6):1673–80.
Bergfelt DR, Bo GA, Mapletoft RJ, Adams GP. Superovulatory response following ablation-induced follicular wave emergence at random stages of the oestrous cycle in cattle. Anim Reprod Sci. 1997;49(1):1–12.
Baracaldo MI, Martinez MF, Adams GP, Mapletoft RJ. Superovulatory response following transvaginal follicle ablation in cattle. Theriogenology. 2000;53(6):1239–50.
Andersen AN, Witjes H, Gordon K, Mannaerts B, investigators X. Predictive factors of ovarian response and clinical outcome after IVF/ICSI following a rFSH/GnRH antagonist protocol with or without oral contraceptive pre-treatment. Hum Reprod. 2011;26(12):3413–23.
Cédrin-Durnerin I, Bständig B, Parneix I, Bied-Damon V, Avril C, Decanter C, et al. Effects of oral contraceptive, synthetic progestogen or natural estrogen pre-treatments on the hormonal profile and the antral follicle cohort before GnRH antagonist protocol. Hum Reprod. 2007;22(1):109–16.
Garcia-Velasco JA, Bermejo A, Ruiz F, Martinez-Salazar J, Requena A, Pellicer A. Cycle scheduling with oral contraceptive pills in the GnRH antagonist protocol vs the long protocol: a randomized, controlled trial. Fertil Steril. 2011;96(3):590–3.
Humm KC, Sakkas D. Role of increased male age in IVF and egg donation: is sperm DNA fragmentation responsible? Fertil Steril. 2013;99(1):30–6.
French DB, Sabanegh ES, Goldfarb J, Desai N. Does severe teratozoospermia affect blastocyst formation, live birth rate, and other clinical outcome parameters in ICSI cycles? Fertil Steril. 2010;93(4):1097–103.
von Wolff M, Thaler CJ, Frambach T, Zeeb C, Lawrenz B, Popovici RM, et al. Ovarian stimulation to cryopreserve fertilized oocytes in cancer patients can be started in the luteal phase. Fertil Steril. 2009;92(4):1360–5.
Nasser LF, Sá Filho MF, Reis EL, Rezende CR, Mapletoft RJ, Bó GA, et al. Exogenous progesterone enhances ova and embryo quality following superstimulation of the first follicular wave in Nelore (Bos indicus) donors. Theriogenology. 2011;76(2):320–7.
Kuang Y, Hong Q, Chen Q, Lyu Q, Ai A, Fu Y, et al. Luteal-phase ovarian stimulation is feasible for producing competent oocytes in women undergoing in vitro fertilization/intracytoplasmic sperm injection treatment, with optimal pregnancy outcomes in frozen-thawed embryo transfer cycles. Fertil Steril. 2014;101(1):105–11.
Acknowledgments
The authors would like to thank the staff of Centro de Reprodução Humana do Hospital das Clínicas da Faculdade de Medicina da USP for their assistance during this study. The authors would like also to thank Merck Serono for donating all recombinant FSH, hCG and cetrorelix acetate used in this study, and acknowledge the valuable contribution made by Tatiana Bonetti (PhD) in the revision of the manuscript. The first author received a grant from CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior)
This study has been registered at ClinicalTrials.gov as NCT 01668056.
Conflict of interest
The first author received a grant from CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior).
Merck Serono (São Paulo, Brazil) donated the medication used in the study.
Compliance with ethical standards
Our hospital’s Ethics Committee approved this study, and women were only enrolled after signing a written informed consent form.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule A follicular wave emergence can be elicited through pharmacological and mechanical interventions, but the outcomes of OI synchronized with FWE should be further evaluated
Rights and permissions
About this article
Cite this article
Bianchi, P.H.M., Viera, L.M., Gouveia, G.R.F.C.A. et al. Study of two strategies to induce follicular wave emergence for assisted reproductive treatments (ART)—a preliminary trial. J Assist Reprod Genet 32, 543–549 (2015). https://doi.org/10.1007/s10815-015-0432-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-015-0432-3