Abstract
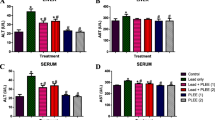
Porphyridium purpureum biomass was assessed for its acute and subchronic safety in albino Wistar rats and for its use as animal feed/human food application. P. purpureum contained 29.9 % protein, 7.99 % lipid, and 2.98 % phycoerythrin on dry basis. Polyunsaturated fatty acids constituted >50 % of the total fatty acids (53.9 % w/w), with eicosapentaenoic acid as the predominant ω3 (21.9 %) and arachidonic acid as the predominant ω6 fatty acid (15.7 %) with an overall ω6/ω3 ratio of 0.42. The essential amino acids (EAAs) constituted 37.97 % (w/w) of proteins with leucine as the predominant amino acid (10.99 %). The EAA index was similar to casein and soybean proteins. Water absorption capacity (WAC; 3.86 ± 0.01 g water g−1 biomass) and oil absorption capacity (OAC; 3.57 ± 0.04 g oil g−1 biomass) were six times higher compared to the basal diet (WAC 0.66 ± 0.007 g water g−1 feed and OAC 0.57 ± 0.02 g oil g−1feed). The acute toxicity study showed that the biomass was safe at acute doses and LD50 exceeded 5.0 g kg−1 body weight, the highest dose used in the study. In a subchronic study, rats were fed a diet containing 0, 2.5, 5.0, 7.5, and 10.0 % levels (w/w) for a period of 13 weeks. The biomass fortification did not induce any untoward changes in physiology such as body weight gain, relative organ weights, histopathology, and hematological and serum biochemical indices. However, supplementation of P. purpureum biomass resulted in decreased serum cholesterol and triglyceride levels compared to control groups indicating its high nutritional value.



Similar content being viewed by others
References
Association of Official Analytical Chemists (AOAC) (1997). Official methods of analysis, 16th edn, 3rd revision. Association of Official Analytical Chemists, Washington DC, USA. 1115 pp
Association of Official Analytical Chemists (AOAC) (2006). Method 975.03 B (b): Metals in plants and pet foods atomic absorption spectrophotometric method. Association of Official Analytical Chemists, Washington, DC
Baethgen WE, Alley MM (1989) A manual colorimetric procedure for measuring ammonium nitrogen in soil and plant Kjeldahl digests. Commun Soil Sci Plant Anal 20:961–969
Becker EW (1994) Microalgae: biotechnology and microbiology. Cambridge University Press, Cambridge, 293 pp
Becker W (2004) Microalgae in human and animal nutrition. In: Richmond A (ed) Handbook of microalgal culture: biotechnology and applied phycology. Blackwell Science, Oxford, pp 312–351
Belay A (2002) The potential application of Spirulina (Arthrospira) as a nutritional and therapeutic supplement in health management. J Am Nutraceut Assoc 5:27–48
Belay A, Kato T, Ota Y (1996) Spirulina (Arthrospira): potential application as an animal feed supplement. J Appl Phycol 8:303–311
Bharucha C, Meyer H, Mody A, Carman RH (1976) Hematology. Handbook of medical laboratory technique. Wesley Press, Mysore, pp 53–59
Bidlingmeyer BA, Cohen SA, Tarvin TL (1984) Rapid analysis of amino acids using pre-column derivatization. J Chromatogr 336:93–104
Borowitzka MA (1988) Vitamins and fine chemicals from micro-algae. In: Borowitzka MA, Borowitzka LJ (eds) Micro-algal biotechnology. Cambridge University Press, Cambridge, pp 153–196
Borowitzka MA (2013) High-value products from microalgae—their development and commercialisation. J Appl Phycol 25:743–756
Christie WW (1982) Lipid analysis, 2nd edn. Pergamon Press, New York, p 338
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Dvir I, Chayoth R, Sod-Moriah U, Shany S, Nyska A, Stark AH, Madar Z, Arad SM (2000) Soluble polysaccharide and biomass of red microalga Porphyridium sp. alter intestinal morphology and reduce serum cholesterol in rats. Br J Nutr 84:469–476
Fevrier C, Seve B (1975) Incorporation of a Spirulina (Spirulina maxima) in swine food. Ann Nutr Aliment 29:625–650
Gellenbeck K (2012) Utilization of algal materials for nutraceutical and cosmeceutical applications—what do manufacturers need to know? J Appl Phycol 24:309–313
Geresh S, Mamontov A, Weinstein J (2002) Sulfation of extracellular polysaccharides of red microalgae: preparation, characterization and properties. J Biochem Biophys Methods 50:179–187
Ginzberg A, Cohen M, Sod-Moriah UA, Shany S, Rosenshtrauch A, Arad (Malis) S (2000) Chickens fed with biomass of the red microalga Porphyridium sp. have reduced blood cholesterol level and modified fatty acid composition in egg yolk. J Appl Phycol 12:325–330
Gouveia L, Batista AP, Sousa I, Raymundo A, Bandarra NM (2008) Microalgae in novel food products. In: Papadoupoulos K (ed) Food Chemistry Research Developments. Nova, New York, pp 75–112
Grobbelaar JU (2003) Quality control and assurance: crucial for the sustainability of the applied phycology industry. J Appl Phycol 15:209–215
Guedes AC, Amaro HM, Barbosa CR, Pereira RD, Malcata FX (2011) Fatty acid composition of several wild microalgae and cyanobacteria, with a focus on eicosapentaenoic, docosahexaenoic and α-linolenic acids for eventual dietary uses. Food Res Int 44:2721–2729
Guil-Guerrero JL, Navaro-Juarez R, Lopez-Martinez JC, Campara-Madrid P, Rebolloso-Fuentes MM (2004) Functional properties of the biomass of three microalgal species. J Food Eng 65:511–517
Hills C, Nakamura H (1978) Food from sunlight; planetary survival for hungry people. University of the Trees Press, Boulder Creek
Hintz HF, Heitman H (1967) Sewage-grown algae as a protein supplement for swine. J Anim Prod 9:135–140
Imafidon GI, Sosulski FW (1990) Nucleic acid nitrogen of animal and plant foods. J Agric Food Chem 38:118–120
Isaacs R, Roneker KR, Huntley M, Lei XG (2011) A partial replacement of soybean meal by whole or defatted algal meal in diet for weanling pigs does not affect their plasma biochemical indicators. J Anim Sci 89:723–729
Iwata K, Inayama T, Kato T (1990) Effects of Spirulina platensis on plasma lipoprotein lipase activity in fructose-induced hyperlipidemia in rats. J Nutr Sci Vitaminol 36:165–171
Kavitha MD, Anila N, Ravishankar GA, Sarada R (2011) Effect of metabolic inhibitors on growth and carotenoids production in Dunaliella bardawil. J Food Sci Technol 50:1130–1136
Kennedy MJ, Reader SL, Davies RJ (1993) Fatty acid production characteristics of fungi with particular emphasis on gamma linolenic acid production. Biotechnol Bioeng 42:625–634
Lacaz-Ruiz R, Mos EN (1990) Produçao de biomass adem Spirulina maxima paraalimentaçao humana e animal. Rev Microbiol 21:85–97
Leng X, Hsu KN, Austic RE, Lei XG (2014) Effect of dietary defatted diatom biomass on egg production and quality of laying hens. J Anim Sci Biotechnol 5:3–10
Lichtenstein AH, Appel LJ, Brands M, Carnethon M, Daniels S, Franch HA (2006) Diet and lifestyle recommendations revision 2006—a scientific statement from the American Heart Association Nutrition Committee. Circulation 114:82–96
Lichtenthaler HK (1987) Chlorophylls and carotenoids: the pigments of photosynthetic biomembranes. In: Douce R, Packer L (eds) Methods in enzymology 148. Academic Press, New York, pp 350–382
Lum KK, Kim J, Lei XG (2013) Dual potential of microalgae as a sustainable biofuel feedstock and animal feed. J Anim Sci Biotechnol 4:53
Merritt RJ, Auestad N, Kruger C, Buchanan S (2003) Safety evaluation of sources of docosahexaenoic acid and arachidonic acid for use in infant formulas in newborn piglets. Food Chem Toxicol 41:897–904
Moreira LM, Behling BS, Rodrigues RS, Costa JAV, Soares AS (2013) Spirulina as a protein source in the nutritional recovery of Wistar rats. Braz Arch Biol Technol 56:447–456
Neori A (2011) “Green water” microalgae: the leading sector in world aquaculture. J Appl Phycol 23:143–149
Plaza M, Cifuentes A, Ibanez E (2008) In the search of new functional food ingredients from algae. Trends Food Sci Technol 19:31–39
Priyadarshani I, Rath B (2012) Commercial and industrial applications of micro algae. J Algal Biomass Utln 3:89–100
Pulz O, Gross W (2004) Valuable products from biotechnology of microalgae. Appl Microbiol Biotechnol 65:635–648
Quinn JR, Paton D (1979) A practical measurement of water hydration capacity of protein materials. Cereal Chem 56:38–40
Reeves PG, Nielsen FH, Fahey GCJ (1993) AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J Nutr 123:1939–1951
Rodríguez-Cruz M, Tovar AR, del Prado M, Torres N (2005) Molecular mechanisms of action and health benefits of polyunsaturated fatty acids. Rev Invest Clin 57:457–472
Roman RB, Alvarez-Pez JM, Fernandez FGA, Molina Grima E (2002) Recovery of pure B-phycoerythrin from the microalga Porphyridium cruentum. J Biotechnol 93:73–85
Ross E, Dominy W (1990) The nutritional value of dehydrated, blue-green algae (Spirulina plantensis) for poultry. Poult Sci 69:794–800
Sadasivam S, Manickam A (2008) Biochemical methods, 8th edn. New Age International Ltd. Publishers, New Delhi
Samarakoon K, Jeon Y-J (2012) Bio-functionalities of proteins derived from marine algae. Food Res Int 48:948–960
Schilter B, Andersson C, Anton R, Constable A, Kleiner J, O’Brien J, Renwick AG, Korver O, Smit F, Walker R (2003) Guidance for the safety assessment of botanicals and botanical preparations for use in food and food supplements. Food Chem Toxicol 41:1625–1649
Sekar S, Chandramohan M (2008) Phycobiliproteins as a commodity: trends in applied research, patents and commercialization. J Appl Phycol 20:113–136
Sellers RS, Morton D, Michael B, Roome N, Johnson JK, Yano BL, Perry R, Schafer K (2007) Society of toxicologic pathology position paper: organ weight recommendations for toxicology studies. Toxicol Pathol 35:751–755
Servel MO, Claire C, Derrien A, Coiard L, De Roeck-Holtzhauer Y (1994) Fatty acid composition of some marine microalgae. Phytochemistry 36:691–693
Shaish A, Ben-Amotz A, Avron M (1992) Biosynthesis of β-carotene in Dunaliella. Methods Enzymol 213:439–444
Shields RJ, Lupatsch I (2012) Algae for aquaculture and animal feeds. J Anim Sci 21:23–37
Simopoulos AP (2008) The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp Biol Med 233:674–688
Sosulski FW, Humbert ES, Bui K, Jones JD (1976) Functional properties of rapeseed flours, concentrates and isolates. J Food Sci 41:1349–1352
SPSS Inc. (1998) SPSS base 8.0 for Windows user’s guide. SPSS, Chicago IL
Tao Y, Barnett SM (2004) Effect of light quality on production of extracellular polysaccharides and growth rate of Porphyridium cruentum. Biochem Eng J 19:251–258
Vidyashankar S, VenuGopal KS, Chauhan VS, Muthukumar SP, Sarada R (2014) Characterization of defatted Scenedesmus dimorphus algal biomass as animal feed. J Appl Phycol. doi:10.1007/s10811-014-0498-9
Yap TN, Wu JF, Pond WG, Krook L (1982) Feasibility of feeding Spirulina maxima, or Chlorella sp. to pigs weaned to a dry diet at 4 to 8 days of age. Nutr Rep Int 25:543–552
Acknowledgments
KMD acknowledges ICMR, Govt. of India, New Delhi, for Senior Research Fellowship. The authors also thank the Director, CSIR-CFTRI, for providing research grant (MLP 0122) to carry out this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kavitha, M.D., Seema Shree, M.H., Vidyashankar, S. et al. Acute and subchronic safety assessment of Porphyridium purpureum biomass in the rat model. J Appl Phycol 28, 1071–1083 (2016). https://doi.org/10.1007/s10811-015-0655-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-015-0655-9