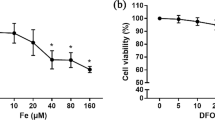
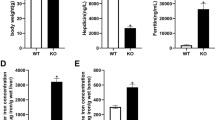
Abstract
Hepcidin is a key player in the regulation of mammalian iron homeostasis. Because iron overload may be one of the causes of osteoporosis, hepcidin may have therapeutic potential for osteoporosis patients. However, the effects of hepcidin on bone metabolism are not fully clear. We recently found that hepcidin can increase intracellular iron and calcium levels and promote mineralization in osteoblasts. The present study was designed to evaluate the effects of hepcidin on osteoclasts. Our results showed that mouse hepcidin 1 (MH1) can increase the number of TRAP-positive MNCs concomitant in both bone marrow-derived macrophages (BMMs) and RAW264.7 cells and upregulate mRNA levels of TRAP, cathepsin K, and MMP-9 and increase TRAP-5b protein secretion in RAW264.7 cells. Moreover, MH1 can downregulate the level of FPN1 protein and increase intracellular iron in RAW 264.7 cells. Therefore, we conclude that MH1 can significantly facilitate osteoclast differentiation in vitro. The mechanism behind accelerated differentiation may be associated with increased levels of intracellular iron. These findings may facilitate understanding of the effects of hepcidin on bone metabolism.






Similar content being viewed by others
Abbreviations
- MH1:
-
Mouse hepcidin 1
- BMMs:
-
Bone marrow-derived macrophages
- M-CSF:
-
Macrophage colony-stimulating factor
- RANKL:
-
Receptor Activator of Nuclear Factor κ B Ligand
- TRAP:
-
Tartrate-resistant acid phosphatase
- CTK:
-
Cathepsin K
- MMP-9:
-
Matrix metalloproteinase 9
- RT-PCR:
-
Reverse transcriptase polymerase chain reaction
- FPN1:
-
Ferroportin 1
- CLSM:
-
Confocal laser scanning microscope
References
Pietrangelo, A. 2011. Hepcidin in human iron disorders: therapeutic implications. Journal of Hepatology 54: 173–181. doi:10.1016/j.jhep.2010.08.004.
Parrow, N.L., S. Gardenghi, and S. Rivella. 2011. Prospects for a hepcidin mimic to treat beta-thalassemia and hemochromatosis. Expert Review of Hematology 4: 233–235. doi:10.1586/ehm.11.22.
Weinberg, E.D. 2006. Iron loading: a risk factor for osteoporosis. Biometals: an international journal on the role of metal ions in biology, biochemistry, and medicine 19: 633–635. doi:10.1007/s10534-006-9000-8.
Weinberg, E.D. 2008. Role of iron in osteoporosis. Pediatric Endocrinology Reviews 6(Suppl 1): 81–85.
Jian, J., E. Pelle, and X. Huang. 2009. Iron and menopause: does increased iron affect the health of postmenopausal women? Antioxidants & Redox Signaling 11: 2939–2943. doi:10.1089/ARS.2009.2576.
Li, G.F., Y.Z. Pan, P. Sirois, K. Li, and Y.J. Xu. 2012. Iron homeostasis in osteoporosis and its clinical implications. Osteoporosis international: a journal established as result of cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA. doi:10.1007/s00198-012-1982-1.
Xi Huang (2010) Treatment of osteoporosis in peri- and post-menopausal women with hepcidin. United States Patent Application Publication. No: US 2010/0204122 A1
Lou, D.Q., G. Nicolas, J.C. Lesbordes, L. Viatte, G. Grimber, M.F. Szajnert, A. Kahn, and S. Vaulont. 2004. Functional differences between hepcidin 1 and 2 in transgenic mice. Blood 103: 2816–2821. doi:10.1182/blood-2003-07-2524 2003-07-2524.
Ganz, T. 2005. Cellular iron: ferroportin is the only way out. Cell Metabolism 1: 155–157. doi:10.1016/j.cmet.2005.02.005.
Rice, A.E., M.J. Mendez, C.A. Hokanson, D.C. Rees, and P.J. Bjorkman. 2009. Investigation of the biophysical and cell biological properties of ferroportin, a multipass integral membrane protein iron exporter. Journal of Molecular Biology 386: 717–732. doi:10.1016/j.jmb.2008.12.063.
Nemeth, E., M.S. Tuttle, J. Powelson, M.B. Vaughn, A. Donovan, D.M. Ward, T. Ganz, and J. Kaplan. 2004. Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science 306: 2090–2093. doi:10.1126/science.1104742.
Abboud, S., and D.J. Haile. 2000. A novel mammalian iron-regulated protein involved in intracellular iron metabolism. The Journal of Biological Chemistry 275: 19906–19912. doi:10.1074/jbc.M000713200.
Donovan, A., A. Brownlie, Y. Zhou, J. Shepard, S.J. Pratt, J. Moynihan, B.H. Paw, A. Drejer, B. Barut, A. Zapata, T.C. Law, C. Brugnara, S.E. Lux, G.S. Pinkus, J.L. Pinkus, P.D. Kingsley, J. Palis, M.D. Fleming, N.C. Andrews, and L.I. Zon. 2000. Positional cloning of zebrafish ferroportin1 identifies a conserved vertebrate iron exporter. Nature 403: 776–781. doi:10.1038/35001596.
McKie, A.T., P. Marciani, A. Rolfs, K. Brennan, K. Wehr, D. Barrow, S. Miret, A. Bomford, T.J. Peters, F. Farzaneh, M.A. Hediger, M.W. Hentze, and R.J. Simpson. 2000. A novel duodenal iron-regulated transporter, IREG1, implicated in the basolateral transfer of iron to the circulation. Molecular Cell 5: 299–309.
Ge, X.H., Q. Wang, Z.M. Qian, L. Zhu, F. Du, W.H. Yung, L. Yang, and Y. Ke. 2009. The iron regulatory hormone hepcidin reduces ferroportin 1 content and iron release in H9C2 cardiomyocytes. The Journal of Nutritional Biochemistry 20: 860–865. doi:10.1016/j.jnutbio.2008.07.014.
Xu, Y., W. Zhang, P. Zhang, L. Xiao, A. Wang, P. Sirois, and K. Li. 2012. Downregulation of ferroportin 1 expression in hFOB1.19 osteoblasts by hepcidin. Inflammation 35: 1058–1061. doi:10.1007/s10753-011-9411-8.
Zhang, P., Y.J. Xu, D.Y. Zhao, Y. Ma, L. Xiao, Y.S. Feng, B.C. Du, Z.M. Qian, and K. Li. 2010. Increased intracellular iron and mineralization of cultured hFOB 1.19 cells following hepcidin activation through ferroportin-1. Saudi Medical Journal 31: 1303–1308.
Li, G.F., Y.J. Xu, Y.F. He, B.C. Du, P. Zhang, D.Y. Zhao, C. Yu, C.H. Qin, and K. Li. 2012. Effect of hepcidin on intracellular calcium in human osteoblasts. Molecular and Cellular Biochemistry 366: 169–174. doi:10.1007/s11010-012-1294-y.
Xu, Y., G. Li, B. Du, P. Zhang, L. Xiao, P. Sirois, and K. Li. 2011. Hepcidin increases intracellular Ca2+ of osteoblast hFOB1.19 through L-type Ca2+ channels. Regulatory Peptides 172: 58–61. doi:10.1016/j.regpep.2011.08.009.
Huh, Y.J., J.M. Kim, H. Kim, H. Song, H. So, S.Y. Lee, S.B. Kwon, H.J. Kim, H.H. Kim, S.H. Lee, Y. Choi, S.C. Chung, D.W. Jeong, and B.M. Min. 2006. Regulation of osteoclast differentiation by the redox-dependent modulation of nuclear import of transcription factors. Cell Death and Differentiation 13(7): 1138–1146. doi:10.1038/sj.cdd.4401793.
Das, S.K., R. Ren, T. Hashimoto, and K. Kanazawa. 2010. Fucoxanthin induces apoptosis in osteoclast-like cells differentiated from RAW264.7 cells. Journal of Agricultural and Food Chemistry 58: 6090–6095. doi:10.1021/jf100303k.
Kirstein, B., T.J. Chambers, and K. Fuller. 2006. Secretion of tartrate-resistant acid phosphatase by osteoclasts correlates with resorptive behavior. Journal of Cellular Biochemistry 98: 1085–1094. doi:10.1002/jcb.20835.
Linsuwanont, B., Y. Takagi, K. Ohya, and H. Shimokawa. 2002. Localization of cathepsin K in bovine odontoclasts during deciduous tooth resorption. Calcified Tissue International 70: 127–133. doi:10.1007/s002230010018.
Linsuwanont, B., Y. Takagi, K. Ohya, and H. Shimokawa. 2002. Expression of matrix metalloproteinase-9 mRNA and protein during deciduous tooth resorption in bovine odontoclasts. Bone 31: 472–478.
Roodman, G.D. 2009. Osteoclasts pump iron. Cell Metabolism 9: 405–406. doi:10.1016/j.cmet.2009.04.005.
Ishii, K.A., T. Fumoto, K. Iwai, S. Takeshita, M. Ito, N. Shimohata, H. Aburatani, S. Taketani, C.J. Lelliott, A. Vidal-Puig, and K. Ikeda. 2009. Coordination of PGC-1beta and iron uptake in mitochondrial biogenesis and osteoclast activation. Nature Medicine 15: 259–266. doi:10.1038/nm.1910.
Sonoda, J., I.R. Mehl, L.W. Chong, R.R. Nofsinger, and R.M. Evans. 2007. PGC-1beta controls mitochondrial metabolism to modulate circadian activity, adaptive thermogenesis, and hepatic steatosis. Proceedings of the National Academy of Sciences of the United States of America 104: 5223–5228. doi:10.1073/pnas.0611623104.
Vianna, C.R., M. Huntgeburth, R. Coppari, C.S. Choi, J. Lin, S. Krauss, G. Barbatelli, I. Tzameli, Y.B. Kim, S. Cinti, G.I. Shulman, B.M. Spiegelman, and B.B. Lowell. 2006. Hypomorphic mutation of PGC-1beta causes mitochondrial dysfunction and liver insulin resistance. Cell Metabolism 4: 453–464. doi:10.1016/j.cmet.2006.11.003.
Acknowledgments
This work was partially supported by the National Natural Science Foundation of China (No. 81273090), Jiangsu provincial grant (No. BK2012608), Social Development Fund of Jiangsu Province (No. BE2011605), and Science and Technology Support Project of Zhenjiang City (social development 2014, to Guo-yang Zhao).
Conflict of Interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, Gy., Di, Dh., Wang, B. et al. Effects of Mouse Hepcidin 1 Treatment on Osteoclast Differentiation and Intracellular Iron Concentration. Inflammation 38, 718–727 (2015). https://doi.org/10.1007/s10753-014-9982-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-014-9982-2