Summary
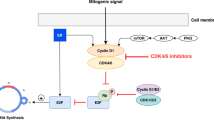
Purpose, Low molecular weight cyclin E (LMW-E) isoforms, overexpressed in a majority (~70 %) of triple-negative breast cancers (TNBC), were found in preclinical models to mediate tumorigenesis through binding and activation of CDK2. CDK1/CDK2 inhibitors, such as dinaciclib, combined with anthracyclines, were synergistic in decreasing viability of TNBC cell lines. Based on this data, a phase 1 study was conducted to determine the maximum tolerated dose of dinaciclib in combination with epirubicin in patients with metastatic TNBC. Methods, Cohorts of at least 2 patients were treated with escalating doses of dinaciclib given on day 1 followed by standard dose of epirubicin given on day 2 of a 21 day cycle. No intra-patient dose escalation was allowed. An adaptive accrual design based upon toxicity during cycle 1 determined entry into therapy cohorts. The target acceptable dose limiting toxicity (DLT) to advance to the next treatment level was 30 %. Results, Between 9/18/2012 and 7/18/2013, 9 patients were enrolled and treated at MD Anderson Cancer Center. DLTs included febrile neutropenia (grade 3, n = 2), syncope (grade 3, n = 2) and vomiting (grade 3, n = 1). Dose escalation did not proceed past the second cohort due to toxicity. After further accrual, the first dose level was also found to be too toxic. No treatment responses were noted, median time to progression was 5.5 weeks (range 3–12 weeks). Thus, accrual was stopped rather than explore the −1 dose level. Conclusion, The combination of dinaciclib and epirubicin is associated with substantial toxicities and does not appear to be an effective treatment option for TNBC.


Similar content being viewed by others
References
Lobbezoo DJ et al (2013) Prognosis of metastatic breast cancer subtypes: the hormone receptor/HER2-positive subtype is associated with the most favorable outcome. Breast Cancer Res Treat 141(3):507–14
Weide R et al (2014) Metastatic breast cancer: prolongation of survival in routine care is restricted to hormone-receptor- and Her2-positive tumors. Springerplus 3:535
Keyomarsi K et al (2002) Cyclin E and survival in patients with breast cancer. N Engl J Med 347(20):1566–75
Wingate H et al (2005) The tumor-specific hyperactive forms of cyclin E are resistant to inhibition by p21 and p27. J Biol Chem 280(15):15148–57
Porter DC et al (2001) Tumor-specific proteolytic processing of cyclin E generates hyperactive lower-molecular-weight forms. Mol Cell Biol 21(18):6254–69
Corin I et al (2006) Tumor-specific hyperactive low-molecular-weight cyclin E isoforms detection and characterization in non-metastatic colorectal tumors. Cancer Biol Ther 5(2):198–203
Bedrosian I et al (2004) Cyclin E deregulation alters the biologic properties of ovarian cancer cells. Oncogene 23(15):2648–57
Akli S et al (2004) Tumor-specific low molecular weight forms of cyclin E induce genomic instability and resistance to p21, p27, and antiestrogens in breast cancer. Cancer Res 64(9):3198–208
Akli S et al (2007) Overexpression of the low molecular weight cyclin E in transgenic mice induces metastatic mammary carcinomas through the disruption of the ARF-p53 pathway. Cancer Res 67(15):7212–22
Nanos-Webb A et al (2012) Targeting low molecular weight cyclin E (LMW-E) in breast cancer. Breast Cancer Res Treat 132(2):575–88
Nemunaitis JJ et al (2013) A first-in-human, phase 1, dose-escalation study of dinaciclib, a novel cyclin-dependent kinase inhibitor, administered weekly in subjects with advanced malignancies. J Transl Med 11:259
Ortiz-Ferron G et al (2008) Roscovitine sensitizes breast cancer cells to TRAIL-induced apoptosis through a pleiotropic mechanism. Cell Res 18(6):664–76
Paruch K et al (2010) Discovery of dinaciclib (SCH 727965): a potent and selective inhibitor of cyclin-dependent kinases. ACS Med Chem Lett 1(5):204–8
Parry D et al (2010) Dinaciclib (SCH 727965), a novel and potent cyclin-dependent kinase inhibitor. Mol Cancer Ther 9(8):2344–53
Mita MM et al (2014) Randomized phase II trial of the cyclin-dependent kinase inhibitor dinaciclib (MK-7965) versus capecitabine in patients with advanced breast cancer. Clin Breast Cancer 14(3):169–76
Ji Y, Li Y, Nebiyou Bekele B (2007) Dose-finding in phase I clinical trials based on toxicity probability intervals. Clin Trials 4(3):235–44
Mita, M.M., et al. (2011) A phase I study of the CDK inhibitor dinaciclib (SCH 727965) administered every 3 weeks in patients (pts) with advanced malignancies: Final results. J Clin Oncol 29(15)
Fabre C et al (2014) Clinical study of the novel cyclin-dependent kinase inhibitor dinaciclib in combination with rituximab in relapsed/refractory chronic lymphocytic leukemia patients. Cancer Chemother Pharmacol 74(5):1057–64
Acknowledgments
This study was supported by R01CA152228, Dr. Khandan Keyomarsi is the PI of the R01 CA15228 and Cancer Center Support Grant CA45809 from the National Cancer Institute
Financial Disclosures: none declared
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mitri, Z., Karakas, C., Wei, C. et al. A phase 1 study with dose expansion of the CDK inhibitor dinaciclib (SCH 727965) in combination with epirubicin in patients with metastatic triple negative breast cancer. Invest New Drugs 33, 890–894 (2015). https://doi.org/10.1007/s10637-015-0244-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-015-0244-4