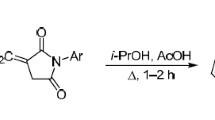
We studied the condensation of 1,2-diamino-4-phenylimidazole with N-arylmaleimides and established that this reaction occurred upon brief refluxing of reactants in isopropanol in the presence of a catalytic amount of acetic acid and produced substituted 7-amino-N-aryl-2-oxo-5-phenyl-1,2,3,4-tetrahydroimidazo[1,5-b]pyridazine-4-carboxamides. Performing this reaction at room temperature led to the acyclic intermediates N-aryl-3-(1,2-diamino-4-phenylimidazol-5-yl)pyrrolidine-2,5-diones.


Similar content being viewed by others
References
(a) Huang, W.-S.; Metcalf, C. A.; Sundaramoorthi, R.; Wang, Y.; Zou, D.; Thomas, R. M.; Zhu, X.; Cai, L.; Wen, D.; Liu, S.; Romero, J.; Qi, J.; Chen, I.; Banda, G.; Lentini, S. P.; Das, S.; Xu, Q.; Keats, J.; Wang, F.; Wardwell, S.; Ning, Y.; Snodgrass, J. T.; Broudy, M. I.; Russian, K.; Zhou, T.; Commodore, L.; Narasimhan, N. I.; Mohemmad, Q. K.; Iuliucci, J.; Rivera, V. M.; Dalgarno, D. C.; Sawyer, T. K.; Clackson, T.; Shakespeare, W. C. J. Med. Chem. 2010, 53, 4701. (b) Peterson, E. A.; Boezio, A. A.; Andrewa, P. S.; Boezio, C. M.; Bush, T. L.; Cheng, A. C.; Choquette, D.; Coats, J. R.; Colletti, A. E.; Copeland, K. W.; DuPont, M.; Graceffa, R.; Grubinska, B.; Kim, J. L.; Lewis, R. T.; Liu, J.; Mullady, E. L.; Potashman, M. H.; Romero, K.; Shaffer, P. L.; Stanton, M. K.; Stellwagen, J. C.; Teffera, Y.; Yi, S.; Cai, T.; La, D. S. Bioorg. Med. Chem. Lett. 2012, 22, 4967. (c) Miller, G. D.; Woessner, D. W.; Sirch, M. J.; Lim C. S., Mol. Pharmaceutics 2013, 10, 3475. (d) Foulks, J. M.; Carpenter, K. J.; Luo, B.; Xu, Y.; Senina, A.; Nix, R.; Chan, A.; Clifford, A.; Wilkes, M.; Vollmer, D.; Brenning, B.; Merx, S.; Lai, S.; McCullar, M. V.; Ho, K.-K.; Albertson, D. J.; Call, L. T.; Bearss, J. J.; Tripp, S.; Liu, T.; Stephens, B. J.; Mollard, A.; Warner, S. L.; Bearss, D. J.; Kanner, S. B. Neoplasia 2014, 16, 403.
(a) Moreau, S.; Coudert, P.; Rubat, C.; Vallee-Goyet, D.; Gardette, D.; Gramain, J.-C.; Couquelet, J., Bioorg. Med. Chem. 1998, 6, 983. (b) Rimoli, M. G.; Russo, E.; Cataldi, M.; Citraro, R.; Ambrosino, P.; Melisi, D.; Curcio, A.; De Lucia, S.; Patrignani, P.; De Sarro, G.; Abignente, E. Neuropharmacology 2009, 56, 637.
Chapman, T. M.; Osborne, S. A.; Bouloc, N.; Large, J. M.; Wallace, C.; Birchall, K.; Ansell, K. H.; Jones, H. M.; Taylor, D.; Clough, B.; Green, J. L.; Holder, A. A. Bioorg. Med. Chem. Lett. 2013, 23, 306
Roberts, L. R.; Bradley, P. A.; Bunnage, M. E.; England, K. S.; Fairman, D.; Fobian, Y. M.; Fox, D. N. A.; Gymer, G. E.; Heasley, S. E.; Molette, J.; Smith, G. L.; Schmidt, M. A.; Tones, M. A.; Dack, K. N. Bioorg. Med. Chem. Lett. 2011, 21, 6515.
Livermore, D. G. H.; Bethell, R. C.; Cammack, N.; Hancock, A. P.; Harm, M. M.; Green, D. V. S.; Lamont, R. B.; Noble, S. A.; Orr, D. C.; Payne, J. J.; Ramsay, M. V. J.; Shingler, A. H.; Smith, C.; Storer, R.; Williamson, C.; Willson, T. J. Med. Chem. 1993, 36, 3784.
Knight, D. J.; Scopes, D. I. C.; Storer, R.; Holman, S. DE 3541358. (Chem. Abstr. 1986, 105, 173001r).
(a) Shih, M.-H. Tetrahedron, 2002, 58, 10437. (b) Costanzo, A.; Bruni, F.; Auzzi, G.; Selleri, S.; Pecori Vettori, L. J. Heterocycl. Chem. 1990, 27, 695. (c) Grandberg, I. I.; Py, D. V.; Kost, A. N. Zh. Obshch. Khim., 1961, 31, 2311. (d) Rudenko, R. V.; Komykhov, S. A.; Musatov, V. I.; Konovalova, I. A.; Shishkin, O. V.; Desenko, S. M. J. Heterocycl. Chem. 2011, 48, 888. (e) Kovygi, Yu. A.; Krylski, D. V.; Zorina, A. V.; Shikhaliev, Kh. S. Chem. Heterocycl. Compd. 2004, 40, 1222. [Khim. Geterotsikl. Soedin., 2004, 1404.] (f) Zorina, A. V.; Shikhaliev, Kh. S.; Kovygin, Yu. A. Vestnik VGU. Seriya: Khim. Biol. Farm., 2005, 1, 39.
Rudenko, R. V.; Komykhov, S. A.; Desenko, S. M.; Musatov, V. I.; Shishkin, O. V.; Konovalova, I. A.; Vashchenko, E. V.; Chebanov, V. A. Synthesis, 2011, 783.
(a) Vandyshev, D. Yu.; Shikalyev, Kh. S.; Potapov, A. Yu.; Krysin, M. Yu. Chem. Heterocycl. Compd. 2015, 51, 573 [Khim. Geterotsikl. Soedin. 2015, 51, 573.] (b) Kolos, N. N.; Orlov, V. D.; Paponov, B. V.; Shishkin, O. V. Chem. Heterocycl. Compd. 1999, 35, 1207. [Khim. Geterotsikl. Soedin. 1999, 1388.] (c) Kolos, N. N.; Orlov, V. D.; Paponov, B. V.; Baumer, V. N. Chem. Heterocycl. Compd. 1998, 34, 1189. [Khim. Geterotsikl. Soedin. 1998, 1397.] (d) Orlov, V. D.; Papiashvili, I. Z.; Povstyanoi, M. V.; Kruglenko, V. P. Chem. Heterocycl. Compd. 1984, 20, 1152. [Khim. Geterotsikl. Soedin. 1984, 1396.] (e) Brückner, R.; Lavergne, J.-P.; Viallefont, P. Liebigs Ann. Chem. 1979, 639. (f) Plaskon, A. S.; Ryabukhin, S. V.; Volochnyuk, D. M.; Shivanyuk, A. N.; Tolmachev, A. A. Heterocycles 2008, 75, 1765. (g) Kolos, N. N.; Beryozkina, T. V.; Orlov, V. D. Mendeleev Commun. 2002, 12, 91. (h) Kolos, N. N.; Kovalenko, L. Yu.; Shishkina, S. V.; Shishkin, O. V.; Konovalova, I. S. Chem. Heterocycl. Compd. 2007, 43, 1397. [Khim. Geterotsikl. Soedin. 2007, 1646.] (i) Lipson, V. V.; Svetlichnaya, N. V.; Shishkina, S. V.; Shishkin, O. V. Mendeleev Commun. 2008, 18, 141. (j) Lipson, V. V.; Svetlichnaya, N. V.; Shirobokov, M. G.; Musatov, V. I.; Shishkin, O. V.; Shishkina, S. V. Russ. J. Org. Chem. 2012, 48, 273. [Zh. Org. Khim. 2012, 48, 281.] (k) Vandyshev, D. Yu.; Shikhaliev, Kh. S.; Potapov, A. Yu.; Krysin, M. Yu. Chem. Heterocycl. Compd. 2014, 50, 1316. [Khim. Geterotsikl. Soedin. 2014, 1428.]
Filimonov, S. I.; Korsakov, M. K.; Chirkova, Zh. V.; Abramov, I. G.; Stashina, G. A.; Firgang, S. I.; Kovygin, Yu. A.; Shikhaliev, Kh. S. Chem. Heterocycl. Compd. 2013, 49, 993. [Khim. Geterotsikl. Soedin. 2013, 1065.]
Lipson, V. V.; Svetlichnaya, N. V.; Shishkina, S. V.; Shishkin, O. V. Mendeleev Commun. 2008, 18, 141.
Ivashchenko, A. V.; Lazareva, V. T.; Prudnikova, E. K.; Ivashchenko, S. P.; Rumyantsev, V. G. Chem. Heterocycl. Compd. 1982, 18, 185. [Khim. Geterotsikl. Soedin. 1982, 236.]
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2015, 51(9), 829–833
Rights and permissions
About this article
Cite this article
Vandyshev, D.Y., Shikhaliev, K.S., Potapov, A.Y. et al. Condensation of 1,2-diamino-4-phenylimidazole and N-arylmaleimides with the formation of new tetrahydroimidazo[1,5-b]pyridazines. Chem Heterocycl Comp 51, 829–833 (2015). https://doi.org/10.1007/s10593-015-1782-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-015-1782-6