Abstract
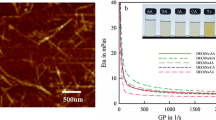
Previously, we reported the preparation of high-crystalline hydrogels from α-chitin nanofibers by a simple NaOH treatment at low temperatures. In the present study, mild conditions were applied to both α-chitin powder and nanofibers to make hydrogels using calcium chloride dehydrate-saturated methanol (Ca solvent). Interestingly, nanofibrillation of powder occurred during the treatment. Compared with the powder, here, both wet chitin nanofiber (WChNF) and methanol-treated chitin nanofiber (MChNF) sheets could easily form into hydrogels at room temperature without the reflux conditions. In this system, water was an important factor in limiting the destruction of chitin crystalline structures caused by calcium ions. This was evident in the WChNF-based hydrogel, which exhibited higher crystallinity and retained its original continuous nanofiber network structure. Finally, we examined the tensile properties of samples prepared from NaOH solution and Ca solvent in the wet state. Although the hydrogel made from Ca solvent was not formed via interdigitation, the WChNF-based one exhibited considerably high tensile strength at around 1.3 MPa, which was close to the NaOH/ethanol-treated sample (tensile strength: 1.8 MPa).






Similar content being viewed by others
References
Abe K, Yano H (2012) Cellulose nanofiber-based hydrogels with high mechanical strength. Cellulose 19:1907–1912
Abe K, Iwamoto S, Yano H (2007) Obtaining cellulose nanofibers with a uniform width of 15 nm from wood. Biomacromolecules 8:3276–3278
Chakrabarty T, Shahi VK (2014) Modified chitosan-based, pH-responsive membrane for protein separation. RSC Adv 4:53245–53252
Chen CC, Li DG, Abe K, Yano H (2014) Dissolution and gelation of α-chitin nanofibers using a simple NaOH treatment at low temperatures. Cellulose 21:3339–3346
Fan Y, Saito T, Isogai A (2008) Preparation of chitin nanofibers from squid pen β-chitin by simple mechanical treatment under acid conditions. Biomacromolecules 9:1919–1923
Hirano S (2002) Some functional biofibers based on chitin and chitosan. Bioindustry 19:62–70
Hu X, Du Y, Tang Y, Wang Q, Feng T, Yang J et al (2007) Solubility and property of chitin in NaOH/urea aqueous solution. Carbohydr Polym 70:451–458
Huang ZH, Dong YS, Chen CL, Lin PH (2008) Electrochemistry assisted reacting deposition of hydroxyapatite in porous chitosan scaffolds. Mater Lett 62:3376–3378
Ifuku S, Saimoto H (2012) Chitin nanofibers: preparations, modifications, and applications. Nanoscale 4:3308–3318
Ifuku S, Nogi M, Abe K, Yoshioka M, Morimoto M, Saimoto H et al (2009) Preparation of chitin nanofibers with a uniform width as α-chitin from crab shells. Biomacromolecules 10:1584–1588
Iwamoto S, Nakagaito A, Yano H (2007) Nano-fibrillation of pulp fibers for the processing of transparent nanocomposites. Appl Phys A 89:461–466
Kim SS, Lee YM (1995) Synthesis and properties of semi-interpenetrating polymer networks composed of β-chitin and poly(ethylene glycol) macromer. Polymer 36:4497–4501
Lavall RL, Assis OBG, Campana-Filho SP (2007) Beta-Chitin from the pens of Loligo sp.: extraction and characterization. Bioresour Technol 98:2465–2472
Lee YM, Kim S, Kim SH (1997) Synthesis and properties of poly(ethylene glycol) macro/β-chitosan hydrogels. J Mater Sci Mater Med 8:537–541
Nagahama H, New N, Jayakumar R, Furuike T, Tamura H (2008) Preparation of chitinous compound/gelatin composite and their biological application. Macromol Symp 264:8–12
Nata I, Wang S, Wu T, Lee C (2012) Carbonaceous hydrogels based on hydrothermal carbonization of glucose with chitin nanofibers. Soft Matter 8:3522–3525
Tamura H, Nagahama H, Tokura S (2006a) Preparation of chitin hydrogel under mild conditions. Cellulose 13:357–364
Tamura H, Sawada M, Nagahama H, Higuchi T, Tokura S (2006b) Influence of amide content on the crystal structure of chitin. Holzforschung 60:480–484
Tamura H, Furuike T, Nair SV, Jayakumar R (2011) Biomedical applications of chitin hydrogel membranes and scaffolds. Carbohydr Polym 84:820–824
Tokura S, Nishi N, Noguchi J (1979) Studies on chitin. III. Preparation of chitin fibers. Polym J 11:781–786
Tokura S, Nishimura S, Sakairi N, Nishi N (1996) Biological activities of biodegradable polysaccharide. Macromol Symp 101:389–396
Wada M, Saito Y (2001) Lateral thermal expansion of chitin crystals. J Polym Sci Part B Polym Phys 39:168–174
Wu J, Liang S, Dai H et al (2010) Structure and properties of cellulose/chitin blended hydrogel membranes fabricated via a solution pre-gelation technique. Carbohydr Polym 79:677–684
Acknowledgments
This work was financially supported by National Natural Science Foundation of China (NSFC 31170514, 31370557), Doctorate Fellowship Foundation of Nanjing Forestry University, Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), Graduate Cultivation Innovative Project of Jiangsu Province and Doctorate Tutor Foundation of Department of Education (20113204110011).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chen, C., Yano, H., Li, D. et al. Preparation of high-strength α-chitin nanofiber-based hydrogels under mild conditions. Cellulose 22, 2543–2550 (2015). https://doi.org/10.1007/s10570-015-0654-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-015-0654-7