Abstract
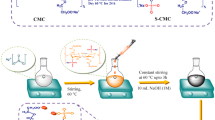
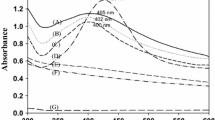
In order to improve the antimicrobial activity of bacterial cellulose (BC), the silver nanoparticles (Ag NPs) were in situ fabricated on the BC membranes, affording BC and Ag hybrid antimicrobial materials, BC + Ag, which possesses excellent antimicrobial performance. Typically, carboxyl groups were firstly introduced into BC by TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl radical)-mediated oxidation. Then, the carboxyl-functionalized BC was performed with ion-exchange reaction to change the sodium ions into Ag+ by immersing in AgNO3 aqueous solution, generating Ag+ anchored BC. Finally, two types of distinct reductive reagents including NaBH4 and sodium citrate were employed to transform Ag+ into Ag NPs to fabricate BC + Ag. The diameters of Ag NPs were determined to be 3.8 nm for NaBH4-reduced BC + Ag, and 22.0 nm for sodium citrate-reduced one, respectively. The silver content of BC + Ag were determined to be 1.944 and 2.895 wt% for NaBH4-reduced sample and sodium citrate-reduced one, respectively. Two types of BC + Ag both showed a slow and persistent Ag+ release profile, but the NaBH4-reduced one released much more Ag+ than that of sodium citrate under the same measurement condition. In-depth antibacterial analysis via the disc diffusion and colony forming count method disclosed that BC + Ag exhibited strong bactericidal effects against both Escherichia coli and Staphylococcus aureus. And the antibacterial activity of NaBH4-reduced BC + Ag was higher than the sodium citrate-reduced one. Overall, this study would further improve the antibacterial efficiency of BC + Ag.







Similar content being viewed by others
Abbreviations
- BC:
-
Bacterial cellulose
- TEMPO:
-
2,2,6,6- tetramethylpiperidine-1-oxyl radical
- BC + Ag:
-
Silver nanoparticles on carboxyl-functionalized BC composites
- ROS:
-
Reactive oxygen species
- DP:
-
Degree of polymerization
- E. coli :
-
Escherichia coli
- S. aureus :
-
Staphylococcus aureus
- SH:
-
Schenk and Hildebrandt medium
- SEM:
-
Scanning electron microscopy
- XRD:
-
X-ray diffraction
- ICP-MS:
-
Inductively coupled plasma mass spectrometry
- FTIR:
-
Fourier transform infrared
- TEM:
-
Transmission electron microscopy
- LB:
-
Luria–Bertani medium
- SPR:
-
Surface plasmon resonance
References
Barud HS, Regiani T, Marques RF, Lustri WR, Messaddeq Y, Ribeiro SJ (2011) Antimicrobial bacterial cellulose-silver nanoparticles composite membranes. J Nanomater 2011:10
Cao X, Ding B, Yu J, Al-Deyab SS (2013) In situ growth of silver nanoparticles on TEMPO-oxidized jute fibers by microwave heating. Carbohydr Polym 92:571–576
Carr HS, Wlodkowski TJ, Rosenkranz HS (1973) Silver sulfadiazine: in vitro antibacterial activity. Antimicrob Agents Chemother 4:585–587
Chaloupka K, Malam Y, Seifalian AM (2010) Nanosilver as a new generation of nanoproduct in biomedical applications. Trends Biotechnol 28:580–588
Choi O, Deng KK, Kim N-J, Ross L Jr, Surampalli RY, Hu Z (2008) The inhibitory effects of silver nanoparticles, silver ions, and silver chloride colloids on microbial growth. Water Res 42:3066–3074
Creighton JA, Blatchford CG, Albrecht MG (1979) Plasma resonance enhancement of Raman scattering by pyridine adsorbed on silver or gold sol particles of size comparable to the excitation wavelength. J Chem Soc Faraday Trans 2 Mol Chem Phys 75:790–798
Czaja W, Krystynowicz A, Bielecki S, Brown RM Jr (2006) Microbial cellulose—the natural power to heal wounds. Biomaterials 27:145–151
Czaja WK, Young DJ, Kawecki M, Brown RM (2007) The future prospects of microbial cellulose in biomedical applications. Biomacromolecules 8:1–12
French AD (2014) Idealized powder diffraction patterns for cellulose polymorphs. Cellulose 21:885–896
He D, Hu B, Yao Q-F, Wang K, Yu S-H (2009) Large-scale synthesis of flexible free-standing SERS substrates with high sensitivity: electrospun PVA nanofibers embedded with controlled alignment of silver nanoparticles. ACS Nano 3:3993–4002
Holladay RJ, Christensen H, Moeller WD (2006) Treatment of humans with colloidal silver composition. US Patents
Ifuku S, Tsuji M, Morimoto M, Saimoto H, Yano H (2009) Synthesis of silver nanoparticles templated by TEMPO-mediated oxidized bacterial cellulose nanofibers. Biomacromolecules 10:2714–2717
Isogai A, Saito T, Fukuzumi H (2011a) TEMPO-oxidized cellulose nanofibers. Nanoscale 3:71–85
Isogai T, Saito T, Isogai A (2011b) Wood cellulose nanofibrils prepared by TEMPO electro-mediated oxidation. Cellulose 18:421–431
Kittler S, Greulich C, Diendorf J, Koller M, Epple M (2010) Toxicity of silver nanoparticles increases during storage because of slow dissolution under release of silver ions. Chem Mater 22:4548–4554
Kong H, Jang J (2006) One-step fabrication of silver nanoparticle embedded polymer nanofibers by radical-mediated dispersion polymerization. Chem Commun 28:3010–3012
Lee P, Meisel D (1982) Adsorption and surface-enhanced Raman of dyes on silver and gold sols. J Phys Chem 86:3391–3395
Li W-R, Xie X-B, Shi Q-S, Zeng H-Y, Ouyang Y-S, Chen Y-B (2010) Antibacterial activity and mechanism of silver nanoparticles on Escherichia coli. Appl Microbiol Biotechnol 85:1115–1122
Li W-R, Xie X-B, Shi Q-S, Duan S-S, Ouyang Y-S, Chen Y-B (2011) Antibacterial effect of silver nanoparticles on Staphylococcus aureus. Biometals 24:135–141
Liu J, Hurt RH (2010) Ion release kinetics and particle persistence in aqueous nano-silver colloids. Environ Sci Technol 44:2169–2175
Lu X, Rycenga M, Skrabalak SE, Wiley B, Xia Y (2009) Chemical synthesis of novel plasmonic nanoparticles. Annu Rev Phys Chem 60:167–192
Maneerung T, Tokura S, Rujiravanit R (2008) Impregnation of silver nanoparticles into bacterial cellulose for antimicrobial wound dressing. Carbohydr Polym 72:43–51
Marambio-Jones C, Hoek EM (2010) A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. J Nanopart Res 12:1531–1551
Martinez-Castanon G, Nino-Martinez N, Martinez-Gutierrez F, Martinez-Mendoza J, Ruiz F (2008) Synthesis and antibacterial activity of silver nanoparticles with different sizes. J Nanopart Res 10:1343–1348
Morones JR, Elechiguerra JL, Camacho A, Holt K, Kouri JB, Ramírez JT, Yacaman MJ (2005) The bactericidal effect of silver nanoparticles. Nanotechnology 16:2346
Nam S, Condon BD (2014) Internally dispersed synthesis of uniform silver nanoparticles via in situ reduction of [Ag (NH3) 2]+ along natural microfibrillar substructures of cotton fiber. Cellulose. doi:10.1007/s10570-014-0270-y
Ouyang Y, Xie Y, Tan S, Shi Q, Chen Y (2009) Structure and antibacterial activity of Ce3+ exchanged montmorillonites. J Rare Earths 27:858–863
Pal S, Tak YK, Song JM (2007) Does the antibacterial activity of silver nanoparticles depend on the shape of the nanoparticle? A study of the gram-negative bacterium Escherichia coli. Appl Environ Microbiol 73:1712–1720
Panáček A, Kvitek L, Prucek R, Kolar M, Vecerova R, Pizurova N, Sharma VK, Nevečná TJ, Zboril R (2006) Silver colloid nanoparticles: synthesis, characterization, and their antibacterial activity. J Phys Chem B 110:16248–16253
Petersen N, Gatenholm P (2011) Bacterial cellulose-based materials and medical devices: current state and perspectives. Appl Microbiol Biotechnol 91:1277–1286
Saito T, Isogai A (2004) TEMPO-Mediated oxidation of native cellulose. The effect of oxidation conditions on chemical and crystal structures of the water-insoluble fractions. Biomacromolecules 5:1983–1989
Saito T, Shibata I, Isogai A, Suguri N, Sumikawa N (2005) Distribution of carboxylate groups introduced into cotton linters by the TEMPO-mediated oxidation. Carbohydr Polym 61:414–419
Saito T, Okita Y, Nge T, Sugiyama J, Isogai A (2006) TEMPO-mediated oxidation of native cellulose: microscopic analysis of fibrous fractions in the oxidized products. Carbohydr Polym 65:435–440
Saito T, Hirota M, Tamura N, Kimura S, Fukuzumi H, Heux L, Isogai A (2009) Individualization of nano-sized plant cellulose fibrils by direct surface carboxylation using TEMPO catalyst under neutral conditions. Biomacromolecules 10:1992–1996
Saito T, Hirota M, Tamura N, Isogai A (2010) Oxidation of bleached wood pulp by TEMPO/NaClO/NaClO2 system: effect of the oxidation conditions on carboxylate content and degree of polymerization. J Wood Sci 56:227–232
Sanpui P, Murugadoss A, Prasad P, Ghosh SS, Chattopadhyay A (2008) The antibacterial properties of a novel chitosan–Ag-nanoparticle composite. Int J Food Microbiol 124:142–146
Su H-L, Chou C-C, Hung D-J, Lin S-H, Pao I, Lin J-H, Huang F-L, Dong R-X, Lin J-J (2009) The disruption of bacterial membrane integrity through ROS generation induced by nanohybrids of silver and clay. Biomaterials 30:5979–5987
Yang G, Xie J, Hong F, Cao Z, Yang X (2012) Antimicrobial activity of silver nanoparticle impregnated bacterial cellulose membrane: effect of fermentation carbon sources of bacterial cellulose. Carbohydr Polym 87:839–845
Acknowledgments
This work was funded by the Cooperation Projects of Foshan City and Chinese Academy (No. 2012HY100115), Strategic Cooperation Projects Guangdong Province and Chinese Academy (No. 2011B090300018), the Scientific and Technological Project of Guangdong Province (No. 2011B010400039, 2011B070500020) and the Scientific and Technological Project of Guangzhou City (No. 11A24060559).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Feng, J., Shi, Q., Li, W. et al. Antimicrobial activity of silver nanoparticles in situ growth on TEMPO-mediated oxidized bacterial cellulose. Cellulose 21, 4557–4567 (2014). https://doi.org/10.1007/s10570-014-0449-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-014-0449-2