Abstract
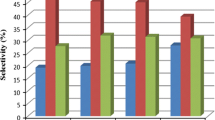
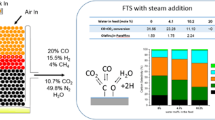
The effect of 1.0 ppm H2S in the syngas feed on initial activity and selectivity of a 0.5%Pt–25%Co/Al2O3 catalyst was studied by comparing the catalyst performance under H2S and sulfur free conditions. The reaction tests were performed using a 1-L slurry phase reactor for 141–212 h under constant reaction conditions: 220 °C, 2.0 MPa, H2/CO = 2.0 and 6.0 Nl/g-cat/h. In the H2S poisoning test, an H2S in N2 gas mixture was added to the syngas feed (1.0 ppm) after running the Fischer–Tropsch synthesis (FTS) reaction for ca. 6.0 h; as such, the impact of H2S on the initial deactivation of the cobalt catalyst (unsteady state) was successfully assessed. The used catalysts were characterized by XANES to investigate if Co–S surface species were formed during the deactivation. The initial deactivation under 1.0 ppm H2S condition was significantly higher (by 2.0–2.4 times) than that under clean conditions. CH4 selectivity increased substantially and C5+ selectivity decreased greatly with time regardless of whether H2S was added or not; however, the addition of H2S accelerated the changes in the hydrocarbon selectivities, which were likely caused by the sharp deactivation of the catalyst in the presence of H2S. After co-feeding the sulfur for 141 h, a comparison was made at similar conversions by adjusting space velocity; the sulfur pretreated cobalt catalysts favored heavier hydrocarbons (C5+) formation and suppressed lower hydrocarbon formation. The addition of H2S to the feed increased CO2 selectivity and the secondary reaction of 1-olefins. The XANES results revealed that surface species involving Co–S bonding formed on the cobalt catalyst after exposure to H2S during FTS. This was likely the primary reason for the abnormal selectivity trends observed during and after the deactivation of the catalyst by sulfur. This study points out a possible approach to increase the selectivity to heavy hydrocarbons by properly sulfiding the cobalt catalyst prior to the FTS reaction.
Graphical Abstract





Similar content being viewed by others
References
Borg O, Hammer N, Enger BC, Myrstad R, Lindv OA, Eri S, Skagseth TH, Rytter E (2011) J Catal 279:163
Shultz JF, Hofer LJE, Karn FS, Anderson RB (1962) J Phys Chem 66:501
Bartholomew CH, Bowman RM (1985) Appl Catal 15:59
Stenger HG Jr, Satterfield CN (1985) Ind Eng Chem Proc Des Dev 24:415
Agrawal PK, Fitzharris WD, Katzer JR (1980) Stud Surf Sci Catal 6:179
Sparks DE, Jacobs G, Gnanamani MK, Pendyala VRR, Ma W, Kang J, Shafer WD, Keogh RA, Graham UM, Gao P, Davis BH (2013) Catal Today 215:67
Visconti CG, Lietti L, Forzatti P, Zennaro R (2007) Appl Catal 330:49
Hadadzadeh H, Mirzaei AA, Morshedi M, Raeisi A, Feyzi M, Rostamizadeh N (2010) Petr Chem 50:78
Ma W, Jacobs G, Sparks DE, Shafer WD, Hamdeh HH, Hopps SD, Pendyala VRR, Hu YF, Xiao QF, Davis BH (2016) Appl Catal A: Gen 513:127
Barrientos J, Montes V, Boutonnet M, Jaras S (2015) Catal Today. doi:10.1016/j.cattod.2015.10.039
Jermwongratanachai T, Jacobs G, Ma W, Shafer WD, Gnanamani MK, Gao P, Kitiyanan B, Davis BH, Klettlinger JLS, Yen CH, Cronauer DC, Kropf AJ, Marshall CL (2013) Appl Catal A: Gen 165:464
Ressler T (1998) J Synchrotron Rad 5:118
Iglesias E (1997) Appl Catal A: Gen 161:59
Ma WP, Jacobs G, Todic B, Bukur DB, Davis BH (2013) Fischer–Tropsch synthesis: influence of process conditions on deactivation of Ru and Re promoted 25%Co/Al2O3 catalysts 23rd North American catalysis society meeting, The Galt House Hotel & Suites, Louisville, Kentucky, June 2–7
Ma WP, Graham UM, Jacobs G, Todic B, Bukur DB, Davis BH (2016) Fischer-Tropsch synthesis, catalysts, and catalysis: advances and applications. In: B.H. Davis, M.L. Occelli (eds). Taylor & Francis Group, Boca Raton, pp 117
Ma W, Jacobs G, Keogh RA, Bukur DB, Davis BH (2012) Appl Catal A Gen 437–438:1
Jacobs G, Ma W, Davis BH (2014) Catalysts 4(1):49
Jacobs G, Ma W, Gao P, Todic B, Bhatelia T, Bukur DB, Khalid S, Davis BH (2012) Topics Catal 55:811
Jacobs G, Das TK, Zhang Y, Li J, Racoillet G, Davis BH (2002) Appl Catal A: General 233:263
Jacobs G, Ji Y, Davis BH, Cronauer DC, Kropf AJ, Marshall CL (2007) Appl Catal A: General 333:177
Jacobs G, Ma W, Gao P, Todic B, Bhatelia T, Bukur DB, Davis BH (2013) Catal Today 214:100
Saib AM, Borgna A, van de Loosdrecht J, van Berge PJ, Niemantsverdriet JW (2006) Appl Catal A: General 312:12
Jacobs G, Chaney JA, Patterson PM, Das TK, Maillot JC, Davis BH (2004) J Synchrotron Rad 11:414
Acknowledgments
This work was made possible by the financial support from DOE Contract Number of DE-FC26-08NT0006368, and the Commonwealth of Kentucky. The XANES research described in this paper was performed in part at the Canadian Light Source, which is funded by the Canada Foundation for Innovation, the Natural Sciences and Engineering Research Council of Canada, the National Research Council Canada, the Canadian Institutes of Health Research, the Government of Saskatchewan, Western Economic Diversification Canada, and the University of Saskatchewan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ma, W., Jacobs, G., Shafer, W.D. et al. Effect of H2S in Syngas on the Fischer–Tropsch Synthesis Performance of a 0.5%Pt–25%Co–Al2O3 Catalyst. Catal Lett 146, 1204–1212 (2016). https://doi.org/10.1007/s10562-016-1747-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-016-1747-0